2017 Volume 40 Issue 11 Pages 1873-1882
2017 Volume 40 Issue 11 Pages 1873-1882
In this study, we used Drosophila as a model species to examine the effects of vitamin or energy-drink and theses ingredients on behavioral activity, life-span, and survivorship. Behavioral assays were performed to analyze total activity during the subjective daytime and nighttime and the lifespan assay was performed to investigate the influence of the drink ingredients. Quantitative RT-PCR and enzyme activity analyses were applied to analyze the mutual relationship of neural pathways and anti-oxidant activities. Caffeine and taurine treatments resulted in significant differences between the control and ascorbic acid groups with respect to subjective daytime and nighttime activity (p<0.05). Additionally, the lifespan and survival on individual flies significantly decreased with 1.6% taurine, and 0.025 and 0.05% caffeine treatment compared to the normal group (p<0.05). These results are related to the transcript levels of neuromodulator (p<0.05). In addition, ascorbic acid treatments significantly increased the activity of antioxidant-related enzymes (p<0.05). We successfully demonstrated that 0.5 and 1.0% ascorbic acid increases the lifespan of fruit flies to a greater extent than 1.6% taurine, and 0.025 and 0.05% caffeine, and that this effect is driven by changes in gene expression and the activity of oxidative stress-related enzyme. In summary, these findings support the use of ascorbic acid as a drink ingredient to enhance body function. Use of the fruit fly in combination with behavior activities and biological processes is recommended for validating the effects of functional substances used by the drink and food industry.
In world markets, functional beverages represent, a fast-growing segment in the beverage industry, and are divided into nutraceutical, energy, and sport drinks.1) Nutraceutical drinks contain bioactive compounds, such as vitamins, minerals, polyphenols, and extracts of fruits, teas, herbs, and vegetables. In addition, energy drinks include to beverages that contain taurine, caffeine, herbal extracts, and vitamin B, and sport drinks are designed to prevent dehydration and provide carbohydrates and electrolytes.1) Functional ingredients in many popular beverages are ascorbic acid (vitamin C), taurine, and caffeine and these ingredients have been used with the purpose of maintaining continuous physical activity and mental alertness.
Ascorbic acid is an important water-soluble antioxidant and an essential micronutrient that is associated with several biochemical and biological functions.2) Ever since the free radical theory of ageing proposed forward by Harman almost 60 years ago,3) there has been speculation on the possible role of ascorbic acid in the prevention of age-related oxidative damage, and in the use of antioxidant dietary factors to prolong lifespan.4) Two common ingredients found in energy drinks such as Red Bull and Monster are taurine and caffeine. Taurine is a sulfhydryl amino acid and a γ-aminobutyric acid (GABA) receptor agonist, which has various physiological actions, such as affecting cellular membrane stability and intracellular calcium levels, and inhibiting of neuronal firing.5) In addition, caffeine is an active ingredient due to its stimulatory effect on the central nervous system, and caffeine administration has been widely studied its cellular and behavioral effects in animal species.6)
In healthy adults, ≤400 mg of caffeine can be consumed daily without any adverse effects and the average daily human intake of taurine is between 40 and 400 mg.7,8) The stimulant effects are short lasting, and at high doses, caffeine can lead to heart disease, gastric acid secretion, and kidney malfunctioning.9) Furthermore, there have been few reports on the intake of large quantities of taurine in combination with other major ingredients of energy drink, such as caffeine.
Although the use of taurine and caffeine is considered to be safe and beneficial, the effect of their continuous ingestion on locomotor activity, lifespan, and biomodulation is still unclear. In the present study, we aimed to investigate the individual and combined effects of these ingredients on locomotor activity, lifespan, and antioxidant activities induced by the consumption of commercial vitamin and energy drinks, or their active ingredients such as ascorbic acid, taurine, and caffeine. The non-invertebrate model using Drosophila melanogaster was used for the analysis of these mutual relationships induced individually and multiplicatively by active ingredients.
Wild-type D. melanogaster Canton-S strain flies were obtained from the Bloomington Drosophila Stock Center at Indiana University (Bloomington, IN, U.S.A.). The flies were raised on standard medium (sucrose, cornmeal, dried yeast, agar, propionic acid, and p-hydroxybenzoic acid methyl ester solution) at 25±1°C in 60–70% relative humidity and a 12 : 12 h light : dark cycle for both control and treatment groups. Prior to treatment, each sample of 2–5 d old male flies were collected under anesthesia, using CO2.
Locomotor Activity AssayCommercial drinks, ascorbic acid drink (0.5% ascorbic acid; 0.0012% vitamin B2; 0.0005% hyaluronic acid; 0.01% taurine, K Pharmaceutical Co., Ltd., Seoul, Republic of Korea), taurine drink (0.83% taurine, 4.2% vitamin B2; 0.025% caffeine; 0.004% thiamine nitrate; 0.004% pyridoxine hydrochloride; 0.017% nicotinic acid amide; 0.04% inositol, D Pharmaceutical Co., Ltd., Seoul, Republic of Korea) and caffeine drink (0.024% caffeine, 0.4% taurine, L Beverage Co., Ltd., Seoul, Republic of Korea) were purchased from a local market and mixed in standard medium. In addition, single treatments of ascorbic acid (0.25, 0.5, and 1.0%), taurine (0.4, 0.8, and 1.6%) and caffeine (0.0125, 0.025, and 0.05%) (Sigma-Aldrich, St. Louis, MO, U.S.A.) were dissolved in distilled water and mixed in sucrose-agar media (5% sucrose and 2% agar) to validate long-term effects of drink ingredients.10) Male flies, 2–5 d-old, were kept in individual glass tubes and the activity of individual flies was assessed by the Drosophila Activity Monitoring system (DAM; TriKinetics, Waltham, MA, U.S.A.) for the single treatment conditions. All recordings were performed every 30 min for 3 (single treatments of drink ingredients with sucrose agar medium)-5 (Commercial drinks with standard medium) days under constant darkness (DD) at 24±1°C and the experiments were performed in triplicate (10 flies per replicate). Data were collected with DAM System Collection software (TriKinetics) and the raw binary data were processed using DAM Filescan110X (TriKinetics). On the actogram of all groups, Actogram J software was used for visualization and black/white bars on top of the x axis representing a 24 h period (from 22:00 to 10:00 of the next day) and the y axis demonstrates successive days of continuous recording. Locomotor activity was collected at 1-min intervals to measure the total number of movements.
Climbing Activity AssayAfter 7 d of each treatment, twenty male adult flies were placed in empty polystyrene vials (9.5 cm tall×2.4 cm diameter) to verify the locomotion patterns with a negative geotaxis assay. Flies were gently tapped to the bottom of the vial and left to allow them to climb to the top of the vial.11) Distance zones from the bottom of the plastic vials were divided into six zones, 0, 0–2, 2–3, 3–5, 5–7, and over 7 cm from the bottom to the top. The number of flies migrating from the bottom to each zone was counted at 1 min observation time point after tapping. For each experiment, the climbing index was calculated as the percentage of those climbing relative to the total number of test flies and replicated five times at 15-min intervals.
Lifespan AssayExperimental groups of 100 male flies distributed in 10 vials were incubated in standard medium supplemented with ascorbic acid (0.25, 0.5, and 1.0%), taurine (0.4, 0.8, and 1.6%) and caffeine (0.0125, 0.025, and 0.05%). Survivors from each group were scored when transferring to fresh medium with ascorbic acid, taurine and caffeine every 3 d. Mean (the sum of all values divided by the number of values added) and median (the central point of a sample set) values were presented to compare results.
SurvivorshipFor the survival rate test, adult male flies (1–2 d old) were transferred to fly vials containing 2 mL of standard medium. Flies were divided into 10 groups (normal and each treatment, by dose). In each group, 50 newly enclosed male flies were transferred to fly vials (10 flies per each vial) and placed in new medium after every 3 d for 7 d. After rearing on standard medium for 1 week, the flies were transferred to new fly vials containing 5% sucrose in 10% H2O2 solution with filter paper, where they remained for 48 h. Survival of flies was recorded every 2 h for 48 h. Data were collected and displayed by survival curve and mean lifespan (MLS).
mRNA ExpressionThe experiment was performed under a 12 : 12 h light:dark cycle for 1 week. Total RNA was extracted from whole bodies of 17–20 d old flies using TRIzol® reagent (Invitrogen, CA, U.S.A.) according to the manufacturer’s protocol. All the experiments were performed in triplicate (50 flies per replicate). Quality controlled RNA samples with high optical density ratios (A260/A280>1.8) were treated with RQ1 ribonuclease (RNase)-free deoxyribonuclease (DNase) I (Promega, WI, U.S.A.) and 1 µg of total RNA was reverse transcribed using SuperScript® III Reverse Transcriptase (Invitrogen) with oligo d(T) as the primer. Quantitative (q)RT-PCR was performed on the resulting cDNA using power Taqman PCR Master Mix kit (Applied Biosystems, CA, U.S.A.). For real-time PCR, cycling conditions were 50°C for 2 min, 95°C for 10 min, followed by 40 cycles at 95°C for 15 s and 60°C for 1 min. Quantitative analyses were conducted using StepOne plus Software V. 2.0 (Applied Biosystems) and results were normalized to a validated control gene, RpL32 (NM_001144655.3), using the ΔΔCt method.12) Target genes used in qRT-PCR were as follows: Dopamine 1-like receptor 1 (Dop1R1, NM_001170136.2), Resistant to dieldrin (Rdl, NM_001274686.1), Metabotropic GABA-B receptor subtype 1 (GABA-B-R1, NM_001259104.2), and Metabotropic GABA-B receptor subtype 2 (GABA-B-R2, NM_001300527.1).
Biochemical AssayPrior to performing the enzyme bioassay, collected adult male flies (1–2 d old) were placed in fly vials (25×100 mm) containing 2 mL of standard medium for 7 d. Flies were divided 10 groups (normal and each treatment, by dose). In each group, 150 newly enclosed male flies were kept in fly vials (10 flies per each vial). The medium was changed every 3 d for 1 week. The surviving flies were euthanized under CO2 anesthesia and immediately homogenized in 0.3 mL of ice-cold 1×N-(2-hydroxyethyl)piperazine-N′-2-ethanesulfonic acid (HEPES) buffer (2×HEPES buffer: pH=7.05 with sodium chloride 1.6 g, potassium chloride 0.074 g, disodium hydrogen phosphate 0.027 g, dextrose 0.2 g, HEPES free acid 1 g in 100 mL distilled water). The homogenates (50 flies per 1.5-mL tube) were centrifuged at 1500×g for 5 min at 4°C, after which 0.2 mL of each supernatant was transferred into a new 1.5-mL tube. Subsequently, each tube was centrifuged at 10000×g for 15 min at 4°C. The supernatant was transferred into a new 1.5-mL tube and the pellet was mixed with 0.1 mL of 1×HEPES buffer. The supernatant was considered to contain the cytosolic copper-zinc containing superoxide dismutase (CuZnSOD), catalase (CAT), and glutathione (GSH), and the pellet was considered to contain mitochondrial manganese-containing SOD (MnSOD). All preparation processes were performed with ice at 4°C. The protein content of each sample was determined using a BCA protein kit (Thermo Fisher Scientific, Rockford, IL, U.S.A.). In brief, SOD present in sample competes with the superoxide radicals of tetrazolium salt. As the SOD concentration increased, the rate of formazan dye formed from tetrazolium salt decreases. This assay was performed according to the method described by Newaz and Adeeb.13) SOD activity in the supernatant and pellet was calculated milligram of protein. Two types of sample (10 µL) were mixed with 0.2 mL of working mixture containing 5 mM potassium phosphate buffer (pH 8.0) 7.5 mL, 3 mM ethylenediaminetetraacetic acid (EDTA) 0.3 mL, 3 mM xanthine 0.3 mL, and 0.75 mM XTT 0.3 mL in 96-well plate. The reaction was initiated by 20 µL of xanthine oxidase. The absorbance was determined at 450 nm in spectrophotometer (Tecan Infinite 2000, Männedorf, Switzerland) using the kinetic mode for 20 min. The result was expressed as unit per milligram of protein. SOD (Sigma Co., U.S.A.) was used as a standard. The CAT activity assay is based on the measurement of hydrogen peroxide remaining in the sample following reaction with catalase. This assay was performed according to the method described by Aebi.14) CAT activity was measured from the supernatant and calculated per milligram of protein. Twenty-five-microliter samples were mixed with 50 µL of 5 mM potassium phosphate buffer (pH 7.0) in a 1.5-mL tube. Subsequently, 25 µL of 200 mM hydrogen peroxide was added to the 1.5-mL tube. After incubation for 1 min at room temperature, 0.9 mL of 15 mM sodium azide was added and the 1.5-mL tube was briefly vortexed. Ten of the reaction mixture was transferred into new 1.5 mL tube and mixed with 1 mL color reagent composed of chromogen reagent (0.25 mM 4-aminoantipyrine, 2 mM 3,5-dichloro-2-hydroxybenzenesulfonic acid) and fresh peroxidase solution. After incubation for 40 min at room temperature, the decreased level of hydrogen peroxide was measured at 520 nm using a spectrophotometer (Tecan Infinite 2000). The result was expressed as unit of hydrogen peroxide decomposed/min per milligram of protein. A hydrogen peroxide solution (Sigma Co.) were used as a standard. GSH is a major intracellular antioxidant, which plays a critical role in the cellular defense against oxidative stress in mammalian cells. This assay is based on the GSH recycling system by 5.5′-dithiobis-[2-nitrobenzoic acid] (DTNB) and GSH reductase. DTNB and GSH react to generate 2-nitro-5-thiobenzoic acid which has a yellow color. This assay was performed according to the method described by Smith et al.15) GSH activity was measured in the supernatant and calculated to milligram per protein. Twenty microliter samples were mixed with 0.15 mL of working mixture containing 0.1 M potassium phosphate buffer (pH 7.0, with 1 mM EDTA 0.8 mL, 6 unit/mL GSH reductase (Sigma Co.) 0.228 mL, and 3 mM DTNB 0.228 mL in 1.5-mL tube. After incubation for 10 min at 37°C, 0.05 mL of 2.5 mM reduced nicotinamide adenine dinucleotide phosphate (NADPH) was added. The absorbance was determined at 412 nm using a spectrophotometer (Tecan Infinite 2000) in kinetic mode for 10 min. The result was expressed as nmol of GSH per milligram of protein. GSH (Sigma Co.) was used as a standard. Prior to this assay, 5% 5-sulfosalicylic acid (SSA) was added to the standard to remove proteins from samples and for to prevent of GSH oxidation.
Statistical AnalysesAll statistical analyses were performed using the Statistical Package for Social Sciences version 12.0 (SPSS Inc., Chicago, IL, U.S.A.). Differences between groups were evaluated by one-way ANOVA and Duncan’s multiple tests. Log-rank tests of lifespan and survival curves were performed by using prism (GraphPad, San Diego, CA, U.S.A.). All data are reported as means±standard error (S.E.).
The effects of functional beverages were investigated by measuring the locomotor activity of fruit flies exposed to sucrose-agar media containing commercial drink ingredients. This assay was performed by visualizing the circadian rhythm using behavioral patterns and measuring the total activity divided with subjective daytime and nighttime (Fig. 1A). Ascorbic acid supplementation had little effect on the circadian rhythm, as compared to that on the normal group during both the subjective nighttime and daytime (Figs. 1B, C). However, the circadian rhythm was greatly diminished by taurine supplementation in a time-dependent manner. Furthermore, caffeine disrupted the circadian rhythm of flies and resulted in circadian rhythms that were irregular in frequency (vertical axis) and amplitude (horizontal axis). As shown in Fig. 1, total movement, as calculated by the circadian rhythm, showed that activity in flies exposed to ascorbic acid was similar to that in the normal group, whereas, activity in the taurine group was significantly decreased. Caffeine significantly increased the activity of flies compared with those in the normal group, during subjective nighttime. Interestingly, during the subjective daytime, taurine and caffeine supplementation resulted in diminished activity compared with that observed under normal and ascorbic acid supplementation (Fig. 1).

(A) Typical actograms of individual control flies (n=10) and flies exposed various drink ingredients (ascorbic acid: n=8, taurine: n=8, caffeine: n=8). The graphic representation of activities generated by Actogram J program and average activity in a 30-min interval was calculated over 5 d. (B, C) Activity during subjective nighttime and daytime. Values are the means±standard error of the mean (S.E.M.) for each group. Mean values with different letters over the bars are significantly different (p<0.05) according to Duncan’s multiple test and bars marked with the same letter were not significantly different at the p≤0.05.
The average movement shown through the circadian rhythm was calculated as the total movement activity (Fig. 2). The circadian rhythm and total movement activity were analyzed by measuring the movement and dividing by the subjective daytime and nighttime. The circadian rhythm of groups supplemented with ascorbic acid at all doses did not differ from that of normal group during subjective daytime and nighttime and behavior patterns following an almost similar tendency. However, the circadian rhythm of flies was sharply decreased following taurine consumption in a dose- and time-dependent manner. Interestingly, caffeine consumption led to disruption of the circadian rhythm of flies and time- and dose-independent increases in both the frequency (vertical axis) and the amplitude (horizontal axis) of the circadian rhythm. Flies supplemented with 0.05% caffeine exhibited the highest total activity, whereas those supplemented with 1.5% taurine showed the lowest activity in the subjective nighttime. During subjective daytime, the ascorbic acid- and caffeine-treated groups showed no significant difference in total movement compared to the normal group. However, those supplemented with 0.8 and 1.5% taurine exhibited decreased total movement compared to normal and other groups.

(A) Typical actograms of individual control flies and flies exposed various drink ingredients. The graphic representation of activities generated by Actogram J program and average activity in a 30-min interval was calculated over 3 d. (B, C) Activity during subjective nighttime and daytime. The total movement activity was calculated as number of counts shown in circadian rhythm. Values are the means±S.E.M. for each group. Mean values with different letters over the bars are significantly different (p<0.05) according to Duncan’s multiple test and bars marked with the same letter were not significantly different at the p≤0.05.
The dose-dependent effects of different treatments on the climbing ability of flies exposed to sucrose-agar media with ascorbic acid, taurine, and caffeine for 1 week is shown in Fig. 3. Ascorbic acid and taurine affected the climbing ability of adult flies in a dose-dependent manner, both in the subjective nighttime and daytime following exposure to ascorbic acid, taurine, and caffeine. Taurine supplementation resulted a significant decrease in the climbing ability at both times. In particular, the inhibitory effects on climbing ability were about 96.43 and 85.46% in response to 1.5% taurine-supplementation at night and day, respectively, compared with the normal group. The climbing abilities also showed slightly decreased patterns in response to supplementation with ascorbic acid, in a dose-dependent manner. In contrast, caffeine-supplementation had no significant effects compared to the untreated group. However, there was a tendency of individuals to continue moving during subjective nighttime and daytime following caffeine supplementation.

The number of fruit flies in zone of glass vial which was over 7 cm from the bottom of vial was counted. Climbing ability of flies showed at subjective nighttime (A), and subjective daytime (B). Values are the means±S.E.M. for each group. Mean values with different letters over the bars are significantly different (p<0.05) according to Duncan’s multiple test and bars marked with the same letter were not significantly different at the p≤0.05.
The effects of ascorbic acid, taurine and caffeine on the lifespan of D. melanogaster were investigated by counting the number of surviving flies each day. The results were shown by survival curve and MLS. As shown in Fig. 4, maximum lifespan of the normal group (untreated group) was 69 and 35 d in MLS. Compared with the normal group, ascorbic acid supplementation did not result in any significant difference in lifespan (Fig. 4A). However, 1.5% taurine supplementation led to a significant decrease in maximum lifespan (53 d) and MLS (30 d) compared with the normal group (Fig. 4B). Decreased survival was more significant following caffeine supplementation. That is, lifespan was significantly diminished in flies exposed to 0.025 and 0.05% caffeine supplementation (Fig. 4C). At both concentrations, the maximum lifespan was 61 d, and MLS was 27 and 29 d.
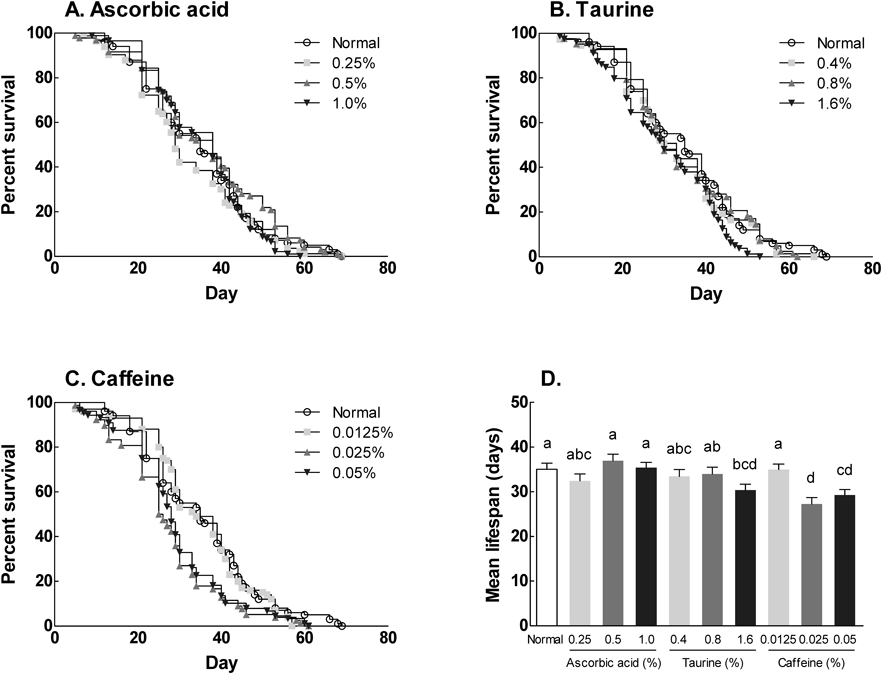
The lifespan of each ingredient-exposed fruit flies was compared with that of normal diet-exposed group. Significant differences of percent survival (%) among groups were analyzed by log-lank statistical methods (n=100). Values are the means±S.E.M. for each group. Mean values with different letters over the bars are significantly different (p<0.05) according to Duncan’s multiple test and bars marked with the same letter were not significantly different at the p≤0.05.
To investigative susceptibility to acute oxidative stress, flies were exposed to 10% hydrogen peroxide (H2O2) following the application of different doses of ascorbic acid, taurine, and caffeine. Resistance to oxidative stress was observed every 2 h for 48 h. For this experiment, the maximum survival of D. melanogaster was adjusted to 46–50 h in response to 10% H2O2. The survival curve and MLS were shown in Fig. 5, and the MLS was calculated from the survival curve. Normal flies treated with 10% H2O2 showed a 46 h maximum lifespan, and 33 h MLS (Fig. 5). Ascorbic acid (Fig. 5A) and caffeine supplementation (Fig. 5C) did result in any decrease on lifespan, including survival rate, and MLS compared to the normal group. However, supplementation with 1.5% taurine resulted in a significant decline in maximum lifespan, and MLS (38, and 26 h, respectively) (Fig. 5B). Taken together, these ingredients were not able to extend the lifespan of flies in response oxidative stress. However, high concentrations of taurine decreased the lifespan of D. melanogaster following oxidative stress.
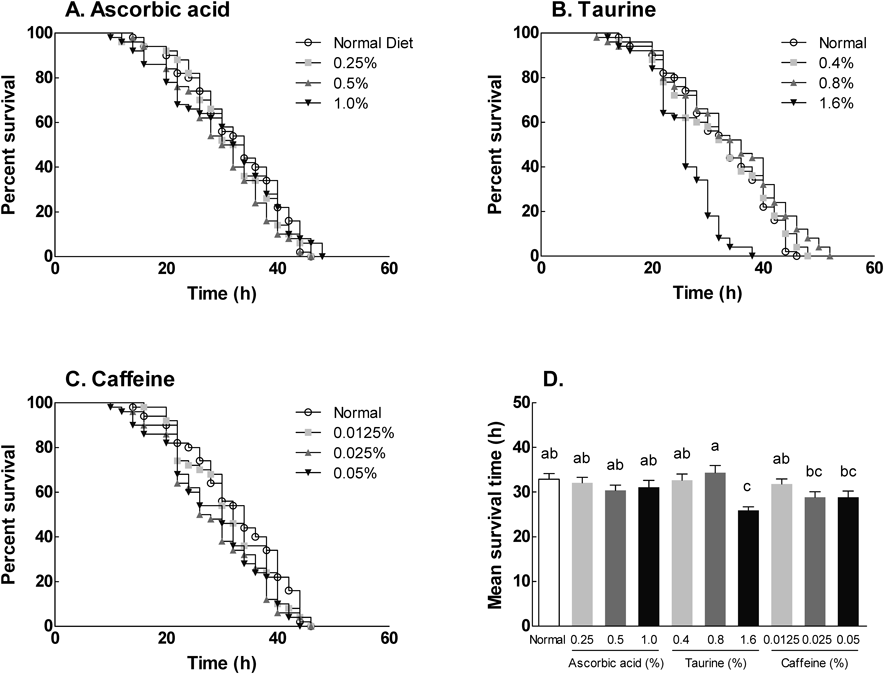
Each ingredient-exposed fruit flies were induced oxidative stress with H2O2 and the lifespan was compared with normal group. Significant differences of percent survival (%) among groups were analyzed by log-lank statistical methods (n=100). Values are the means±S.E.M. for each group. Mean values with different letters over the bars are significantly different (p<0.05) according to Duncan’s multiple test and bars marked with the same letter were not significantly different at the p≤0.05.
To investigate whether the neuronal mechanism of ascorbic acid, taurine, and caffeine involved transcriptional processes, mRNA levels for Dop1-R1, Rdl, GABAB-R1, and GABAB-R2 were determined (Fig. 6). As shown in Fig. 6A, ascorbic acid and caffeine significantly increased mRNA levels of Dop1-R1 compared with those in the normal group. Caffeine increased mRNA levels of Dop1-R1 (1.56, 1.72 and 1.76-fold increase compared to normal group) and ascorbic acid also significantly induced the increase of Dop-R1 levels (1.52-fold increase in 1% ascorbic acid compared with normal group). Meanwhile, taurine increased the levles of GABA receptors, such as Rdl, GABAB-R1, and GABAB-R2, in a dose-dependent manner (Figs. 6B, C, D). Transcript levels of Rdl, GABAB-R1 and GABAB-R2 were 2.26, 1.94, and 1.77-fold in 1.5% taurine compared with normal group, respectively. In contrast, caffeine significantly and dose-dependently decreased mRNA levels of GABA receptors and the most decreased levels of Rdl, GABAB-R1 and GABAB-R2 genes were showed in the group supplemented with 0.05% caffeine (0.20, 0.43 and 0.66-fold decrease in 0.05% caffeine compared with normal group).

The experiment was performed in a 12 : 12 h light : dark cycle for 1 week. Values are the means±S.E.M. for each group. Mean values with different letters over the bars are significantly different (p<0.05) according to Duncan’s multiple test and bars marked with the same letter were not significantly different at the p≤0.05.
Ascorbic acid supplementation significantly increased CuZnSOD activity (cytoplasm) and MnSOD activity in the pellet (mitochondria) in dose-dependent manner (Figs. 7A, B). For CuZnSOD activity, the greatest effect was observed with 0.5 and 1% ascorbic acid supplementation (10.28 and 19.88 U/mg protein, respectively) compared with the normal group (5.32 U/mg protein), whereas only 1% supplementation revealed significantly different results compared with the normal group. However, there was no significant increase in CuZnSOD activity following taurine and caffeine supplementation (Fig. 7A). MnSOD activities also showed a similar tendency. That is, ascorbic acid supplementation group (0.25%, 0.67 U/mg protein; 0.5%, 0.74 U/mg protein; 1%, 0.80 U/mg protein) led to a significant increase compared with the normal group (0.43 U/mg protein), whereas no increase was observed in the other groups (taurine and caffeine). However, groups supplemented with ascorbic acid were not significantly different. In contrast, CAT activity was different following the addition of SOD. For CAT activity, caffeine-supplemented groups (96.88 mM/min/mg protein in 0.0125% caffeine; 113.17 mM/min/mg protein in 0.025% caffeine; 143.91 mM/min/mg protein in 0.05% caffeine) exhibited significantly higher activity compared with the normal group (58.2 mM/min). In particular, 0.05% caffeine-supplementation resulted in the highest CAT activity. Among the ascorbic acid-supplemented groups, only 1% that supplemented with 1% ascorbic acid showed significantly increased activity (98.53 mM/min/mg protein) (Fig. 7C). GSH content was measured in the supernatant of homogenates (Fig. 7D) within contrast to other antioxidant activities, ascorbic acid, taurine and caffeine resulted in significant dose-dependent increases. The 0.5% ascorbic acid, 0.8% taurine, and 0.025% caffeine groups (0.25, 0.24, and 0.24 nM/mg protein, respectively) showed significantly higher GSH content compared with the normal group (0.2 nM/mg protein). The groups supplemented with 1% ascorbic acid, 1.5% taurine and 0.05% caffeine showed the highest significant increase (0.35, 0.31, and 0.3 nM/mg protein, respectively) within each group. Taken together, these results indicate that ascorbic acid supplementation increased the antioxidant activities of D. melanogaster. Furthermore, taurine and caffeine also resulted in increases in some antioxidant activities, especially CAT and GSH content. Oxidative stress might increase antioxidant activity, but changes in the lifespan did not coincide with increases in the antioxidant activities.

The experiment was performed in a 12 : 12 h light : dark cycle for 1 week. Values are the means±S.E.M. for each group. Mean values with different letters over the bars are significantly different (p<0.05) according to Duncan’s multiple test and bars marked with the same letter were not significantly different at the p≤0.05.
The consumption of functional drinks, containing vitamins, taurine, caffeine, and herbal extracts is increasing, as demonstrated by their large market growth.1) Although the ingredients of functional drinks are being investigated in various studies on behavior or lifespan, few studies have investigated the relationship between behavior, lifespan, and the antioxidant activity of vitamin and energy drinks. Therefore, we aimed to investigate the effects of functional drinks, as well as their major ingredients, on behavior patterns, survival, and antioxidant activity using the well-characterized study model species D. melanogaster.
In the present study, we utilized commercial drinks to investigate significant difference in locomotor activity during subjective nighttime and daytime (Fig. 1). Taurine, which is present in energy drinks, significantly decreased the subjective nighttime and daytime activity compared with the normal and ascorbic acid. In contrast, caffeine significantly increased subjective nighttime activity as compared to that in the normal, ascorbic acid, and taurine groups. The active ingredients in vitamin drinks, such as ascorbic acid are essential for collagen and carnitine and acts as co-factor for the enzyme dopamine-β-hydroxylase.16) Furthermore, ascorbic acid inhibits T-cells apoptosis and maintains T-cell proliferation to enhance immune responses.17) Also, Lin et al. reported the interaction of caffeine and/or taurine on locomotion and sleep of Drosophila, and addressed the potential impact of energy drink consumption.10) Taurine is the second most abundant amino acid in the mammalian brain and is also present in mushroom bodies (MBs) in Drosophila.18) It GABAA receptor agonists (GABAA-Rs or Rdl) and associated activation of glycine receptors (Gly-Rs), result in cellular membrane stability and modulation of intracellular calcium levels.19) Caffeine in energy drinks inhibits adenosine receptors and also increases levels of epinephrine, which can positively affect physical and mental performance.20)
We also demonstrated the dose-dependent effects of drink ingredients on locomotor activity in fruit flies, and showing that high levels of taurine and caffeine altered locomotor activity during both the subjective nighttime and daytime of the circadian rhythm (Fig. 2). In addition, we assessed the dose-dependent effects of ascorbic acid, taurine, and caffeine on climbing ability (Fig. 3). The climbing ability of fruit flies induced by taurine and caffeine supplementation might be associated with the mitigation and arousal effect. These results are consistent with those of a previous report showing that taurine reduces locomotor activity in a dose-dependent manner while caffeine increases locomotor activity.10) It is well known that circadian rhythms are fundamental biological phenomena resulting from molecular genetic mechanisms known as circadian clocks, our results indicated that taurine and caffeine might exert biological effects on the circadian rhythm.
We studied the dose-dependent effects of ascorbic acid, taurine, and caffeine on the lifespan of fruit flies (Fig. 4). We also found that the administration of 1.6% taurine, and 0.025 and 0.05% caffeine was associated with decreased survival flies (Fig. 5). Although mean lifespan of flies was found to be similar to controls for concentrations of 0.1 and 10 mM ascorbic acid, higher concentrations of ascorbic acid (10 and 50 mM) enhanced the toxic effects of iron, with the iron pool significantly increased in adult whole-body extracts.21) Also, a concentration of 10 and 50 mM ascorbic acid diminished lifespan of male flies by 6.9 and 12.8%, compared to the normal group and 1 mM ascorbic acid treatment.22) Taken together, these results show that administration of ascorbic acid did not affect the lifespan of fruit flies. However, taurine and caffeine at relatively high doses and concentrations compared with those present in commercial drinks reduced the lifespan of flies. Nikitin et al. reported a negative dosage-dependent effect of caffeine on longevity and other life history parameters.23)
To better understand the effect of drink ingredients on dose-dependent changes in locomotor activity, lifespan and survival, we utilized qRT-PCR and analyzed enzyme activities to compare the effects of antioxidant activity on these parameters (Figs. 6, 7). In Drosophila, caffeine modulates the cAMP- protein kinase A (PKA) pathway, resulting in increased the cAMP-levels and fragmented sleep.24) Also, Dop1R1 levels have been shown to mediate caffeine-induced arousal and higher-order brain functions such as motor control, learning and memory. Furthermore, GABA exerts its effects through ionotropic GABAA, to produce fast synaptic inhibition, and metabotropic GABAB receptors, to produce slow, prolonged inhibitory neurotransmission. GABAB receptor agonists are involved in inhibiting dopamine release and regulating glutamatergic systems of dopamine.25) The sleep–wake cycle disturbance associated with behavioral and biological processes can be partially explained through alteration in neuronal signaling. There are indications that neuronal signals generated by taurine or caffeine administration at synapses could interact with various receptors, cause receptor desensitization and alteration of neuromodulator homeostasis, and lead to neurological disorders. Administration of ascorbic acid is significantly related to the adverse effect of reactive oxygen and nitrogen species, which can cause oxidative damage to DNA, proteins, and lipids and reduces superoxide anions via the modulation of antioxidant enzymes activity (SOD, glutathione peroxidase (GPx)).26,27) Nonaka et al. reported that taurine reduces endoplasmic reticulum (ER) stress induced by homocysteine and affects the expression and secretion of extracellular SOD (EC-SOD).28) In addition, chronic caffeine ingestion modulated lipid peroxidation and the concentration of reduced-GSH, which similarly increased the activities of SOD and GSH reductase (GR).29) Our results indicate a relationship among lifespan, antioxidants and behavior induced by ascorbic acid, taurine and caffeine, which are major active ingredients in vitamin and energy drinks. Ascorbic acid has an enhancing effect that maintains the stability of the body rhythm and increases anti-oxidative activity. In contrast, taurine and caffeine are neuromodulators that induce behavioral inhibition and arousal. These behavioral changes following taurine and caffeine supplementation are thought to affect the antioxidant activity and lifespan of flies. Growing evidence suggests that several functional beverages are associated with antioxidant activity, lifespan and locomotor activity. The results of the present study might represent fundamental data on the industrial application of the functional drink market.
This research was supported by the Kwang Dong Pharmaceutical Co., Ltd.
The authors declare no conflict of interest.