2021 年 4 巻 1 号 p. 1-5
2021 年 4 巻 1 号 p. 1-5
Photoreceptor cell death leads to blindness in dry age-related macular degeneration (AMD) patients. However, no current therapy exists to treat drug for dry AMD patients. Here, we investigated the neuroprotective effects of ropinirole, a dopamine agonist used to treat Parkinson’s disease, in vitro and in vivo murine dry AMD models. Ropinirole prevented photoreceptor cell death induced by excessive light exposure in vitro. Next, we exposed mice to intensive white fluorescent light, performed morphological analysis of the outer nuclear layer (ONL), and examined electroretinogram (ERG) recordings. Although light exposure reduced ONL thickness and ERG amplitudes, the oral administration of ropinirole improved ONL thickness and a- and b-wave amplitudes, similar to pramipexole treatment. Furthermore, the continuous administration of ropinirole using an osmotic pump attenuated the decrease in retinal thickness induced by chronic light exposure. These findings indicated that ropinirole represents a novel candidate drug for dry AMD treatment.
Photoreceptor cells play a crucial role in the conversion of light into neural signals in retina and are responsible for visual acuity. In age-related macular degeneration (AMD) patients, central vision is impaired due to gradual photoreceptor degeneration.1) There are mainly two types of AMD: dry (non-exudative) and wet (exudative) AMD. Anti-vascular endothelial growth factor (VEGF) drugs have been approved for the treatment of wet AMD, while no drugs have been developed for dry AMD. Because the retina is susceptible to oxidative stress, excessive light exposure can result in photoreceptor cell death or dysfunction.2,3) Indeed, we have previously reported that excessive light exposure causes retinal degeneration, accompanied by the thinning of the outer nuclear layer (ONL) and electroretinogram (ERG) amplitude reductions, and treatment with anti-oxidative agents and other various types of agents have been shown to attenuate these detrimental effects.4-6)
In this study, we focused on ropinirole and pramipexole (Fig. 1). Ropinirole and pramipexole are selective non-ergoline dopamine D2/D3 agonist,7) that were initially developed for the treatment of Parkinson’s disease. In the retina, both dopamine D2 and D3 receptors have been detected,8) and it is indicated that both cones and rods feature dopamine D2 receptor.9,10) Several studies have reported the neuroprotective properties of ropinirole.11,12) Ropinirole was found to increase precursor cell proliferation in the subependymal zone,13) and its antioxidant activity was demonstrated using the 1,1-diphenyl-2-picrylhydrazyl (DPPH) assay.14) Recently, induced pluripotent stem cell (iPSC)-based screening demonstrated that ropinirole was the most effective candidate drug for the treatment of amyotrophic lateral sclerosis (ALS) pathogenesis.15) Taken together, these findings suggested that ropinirole might be able to prevent photoreceptor degeneration in AMD pathology.
The Chemical Structures of Ropinirole and Pramipexole
The chemical structures of ropinirole (4-[2-(dipropylamino)ethyl]-1,3-dihydro-2H-indol-2-one) (A) and pramipexole ([S]-2-amino-4,5,6,7-tetrahydro-6-[propylamino]benzothiazole) (B).
In the present study, we evaluated the potential effects of ropinirole, both in vitro and in vivo, in murine models of dry AMD induced by light irradiation. Our findings suggested that ropinirole may be beneficial for the treatment of dry AMD pathogenesis.
The murine photoreceptor cell line, 661W, was kindly gifted by Dr. Muayyad R. Al-Ubaidi (University of Houston, Houston, TX, USA). The cells were cultured in Dulbecco’s modified Eagle medium (DMEM) supplemented with 10% fetal bovine serum (FBS; Biosera, Kansas City, MO, USA), 100 Unit/mL penicillin (Meiji Seika Pharma Co., Ltd., Tokyo, Japan), and 100 µg/mL streptomycin (Meiji Seika Pharma Co., Ltd.) in a humidified atmosphere with 5% CO2 at 37°C.
Light-Induced Cell Death AssayThe light-induced cell death assay was performed as previously described.16) Briefly, 661W cells were seeded in 96-well plates at a density of 3.0 × 103 cells/well, followed by 24 h of incubation at 37°C. The medium was replaced with DMEM containing 1.0% FBS. Ropinirole hydrochloride (Wako, Osaka, Japan) or N-acetyl-L-cysteine (NAC; Wako) were added to the medium, and the cells were exposed to 2,500 lux white fluorescent light for 24 h in a 5% CO2 atmosphere at 37°C. Hoechst 33342 (Invitrogen, Carlsbad, CA, USA) and propidium iodide (PI; Invitrogen) were added to the culture medium for 15 min at the final concentrations of 8.1 μM and 1.5 μM, respectively. Images were captured using an Olympus IX70 inverted epifluorescence microscope (Olympus, Tokyo, Japan).
AnimalsAdult male ddY mice were purchased from Japan SLC (Hamamatsu, Japan) and maintained under controlled lighting conditions (12-h light/12-h dark). Eight-week-old ddY mice were used in all experiments. All experiments were performed in accordance with the Association for Research in Vision and Ophthalmology Statement for the Use of Animals in Ophthalmic and Vision Research and were approved and monitored by the institutional animal care and use committee of Gifu Pharmaceutical University.
Acute Light-Induced Retinal Damage Mouse Model GenerationAcute light-induced retinal damage model mice were generated according to the previously described method.16) Mice were maintained in the dark for 24 h before experiments. Thirty minutes before light exposure, the pupils of the mice were dilated with 1% cyclopentolate hydrochloride (Santen, Osaka, Japan). The drugs were orally administrated to the mice just before light damage. Ropinirole hydrochloride (Wako) and pramipexole dihydrochloride (Tokyo Chemical Industry, Tokyo, Japan) were orally administrated at doses of 80 mg/kg and 1.0 mg/kg, respectively. Non-anesthetized mice were placed in a cage with a reflective interior, and light damage was induced by exposing to 8,000 lux of white fluorescent light for 3 h. All mice were placed in darkness for 24 h, and then returned to a normal light/dark cycle.
Histological AnalysisAt each sampling time point, the mice were euthanized by cervical dislocation, and the eyes were enucleated. The enucleated eyes were fixed with 4% paraformaldehyde (PFA) in 0.1 M phosphate buffer (pH 7.4) for at least 24 h at 4°C. After paraffin embedding, retinal sections (5 μm thickness) containing the optic nerve disc were prepared, followed by hematoxylin and eosin (H&E) staining. All images were captured using a fluorescence microscope (All-in-One BZ-X710; Keyence, Osaka, Japan). The thickness of the outer nuclear layer (ONL) was measured at 240 μm intervals from the optic disc in a blinded manner. Data from three sections were averaged for each eye.
ERG AnalysisAt 5 d after light damage, ERG was recorded to test the electrical activity of the retina. Mice were maintained in complete darkness for 24 h before ERG measurements, and then they were anesthetized using ketamine (120 mg/kg, intraperitoneally, i.p.; Daiichi-Sankyo, Tokyo, Japan) and xylazine (6 mg/kg, i.p.; Bayer Health Care, Tokyo, Japan). The pupils of mice were dilated with 1% tropicamide and 2.5% phenylephrine (Santen), and flash ERG was recorded in the eyes of dark-adapted mice. Flash ERG was recorded for the eyes of dark-adapted mice using white fluorescent stimuli, at intensity ranges from -2.92 to 0.98 log cds/m2.
Chronic Light-Induced Retinal Damage Model and Osmotic Pump ImplantationStarting one week before light irradiation, 2 mg/kg/d ropinirole was continuously administrated using an ALZET osmotic pump (model 2006; Durect Corporation, Cupertino, CA, USA) filled with ropinirole. The device was implanted subcutaneously and continuously delivered ropinirole at 0.15 μL/h. One week after implantation, the mice were irradiated with a blue light-emitting diode (LED) at 100 lux, from 08:00 AM to 20:00 PM, for 4 weeks. Finally, the eyes of the mice were enucleated, and the tissues were evaluated by histological analysis.
Measurement of Blood Ropinirole ConcentrationsMice were separated into two groups: a blank group and a ropinirole infusion group. Ropinirole was administrated with an ALZET osmotic pump (model 2006; Durect Corporation) at a rate of 0.15 μL/h. At 1 and 7 d after the implantation, the mice were anesthetized with isoflurane and blood samples were collected from the tail vein (n = 5). At 14 d, blood samples were collected from the inferior vena cava (n = 5). The blank value was measured using a blood sample collected from untreated mice (blank group; n = 9).
Statistical AnalysisThe data are expressed as the mean ± standard error of the mean (SEM). Statistical comparisons were conducted using Student’s t-test and Tukey’s test. Statistical analysis was performed using the SPSS Statistics software (IBM Corporation, Armonk, NY, USA). A p value < 0.05 was considered significant.
To evaluate the effects of ropinirole on retinal photoreceptor cells, 661W cells were exposed to intense light irradiation (Fig. 2A). After exposure to 2,500 lux fluorescent light, the proportion of PI positive cells was measured. Exposure to excessive fluorescent light caused photoreceptor cell death, which was attenuated by treatment with the antioxidant NAC (1 mM). Ropinirole, at the concentrations of 0.01-10 μM, prevented cell death (Fig. 2B).
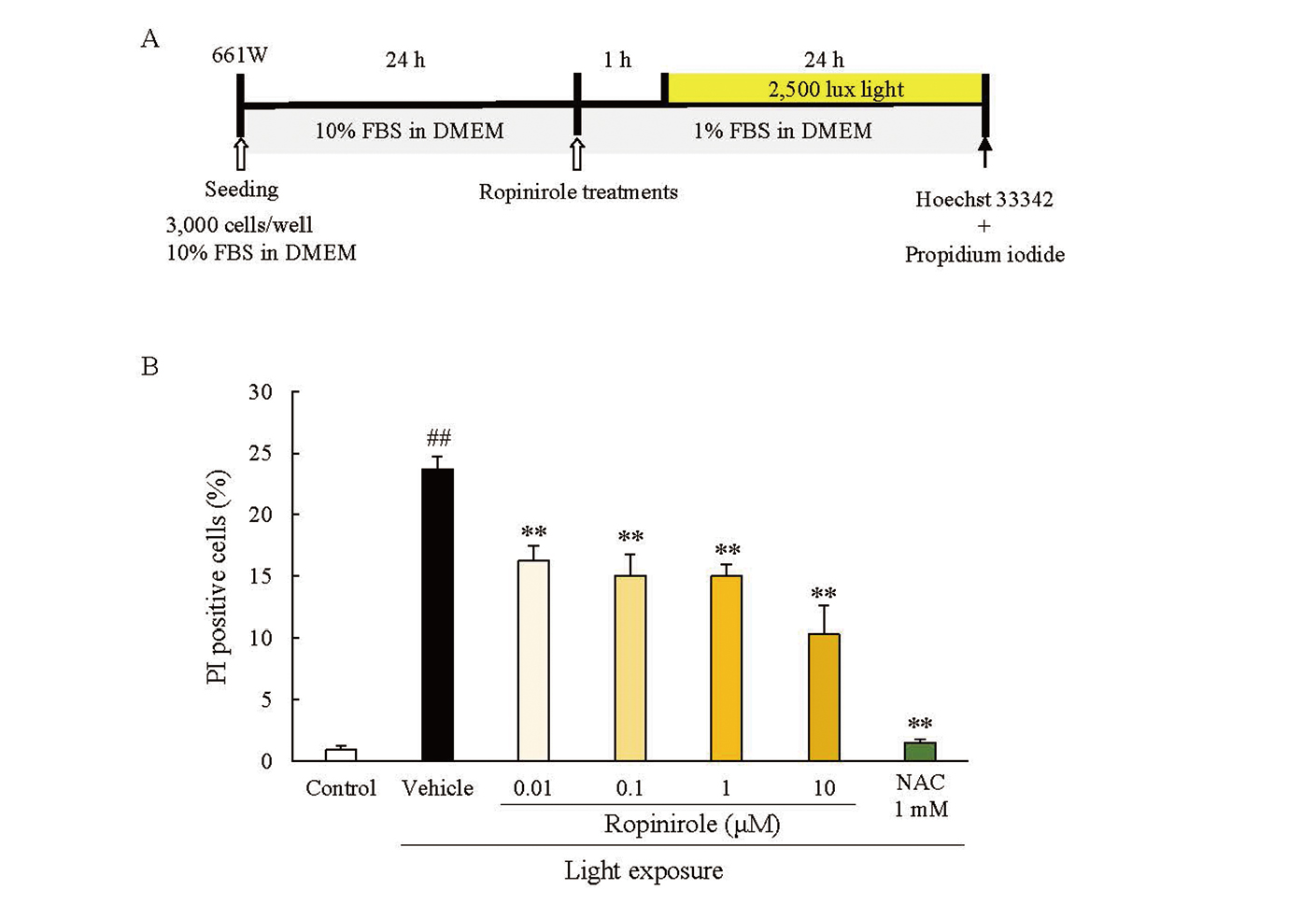
Retinal Photoreceptor Cell Death was Suppressed by Ropinirole
(A) The experimental design for the 661W cell death assay. At 24 h after seeding, the culture medium was changed to DMEM with 1% FBS, and then either ropinirole or NAC (1 mM) was added to the medium. After an hour incubation, 661W cells were exposed to 2,500 lux white fluorescent light for 24 h. The cells were stained with Hoechst 33342 and propidium iodide (PI) to perform the cell death assay. (B) Quantitation of (A). Data are presented as the mean ± SEM (n = 6). ##, p < 0.01 vs. Control; **, p < 0.01 vs. Vehicle (Tukey’s test). NAC, N-acetyl-L-cysteine.
We evaluated the potential effects of ropinirole against light-induced retinal damage (Fig. 3A). Because pramipexole was previously reported to suppress light-induced retinal damage,17) we orally administered ropinirole and pramipexole, both of which are non-ergoline dopamine receptor agonists, prior to light exposure. Ropinirole and pramipexole suppressed the thinning of the ONL induced by acute light damage (Fig. 3B-C). Furthermore, ERG analysis showed that ropinirole improved retinal functions, as 17 well as pramipexole (Fig. 3D-E).
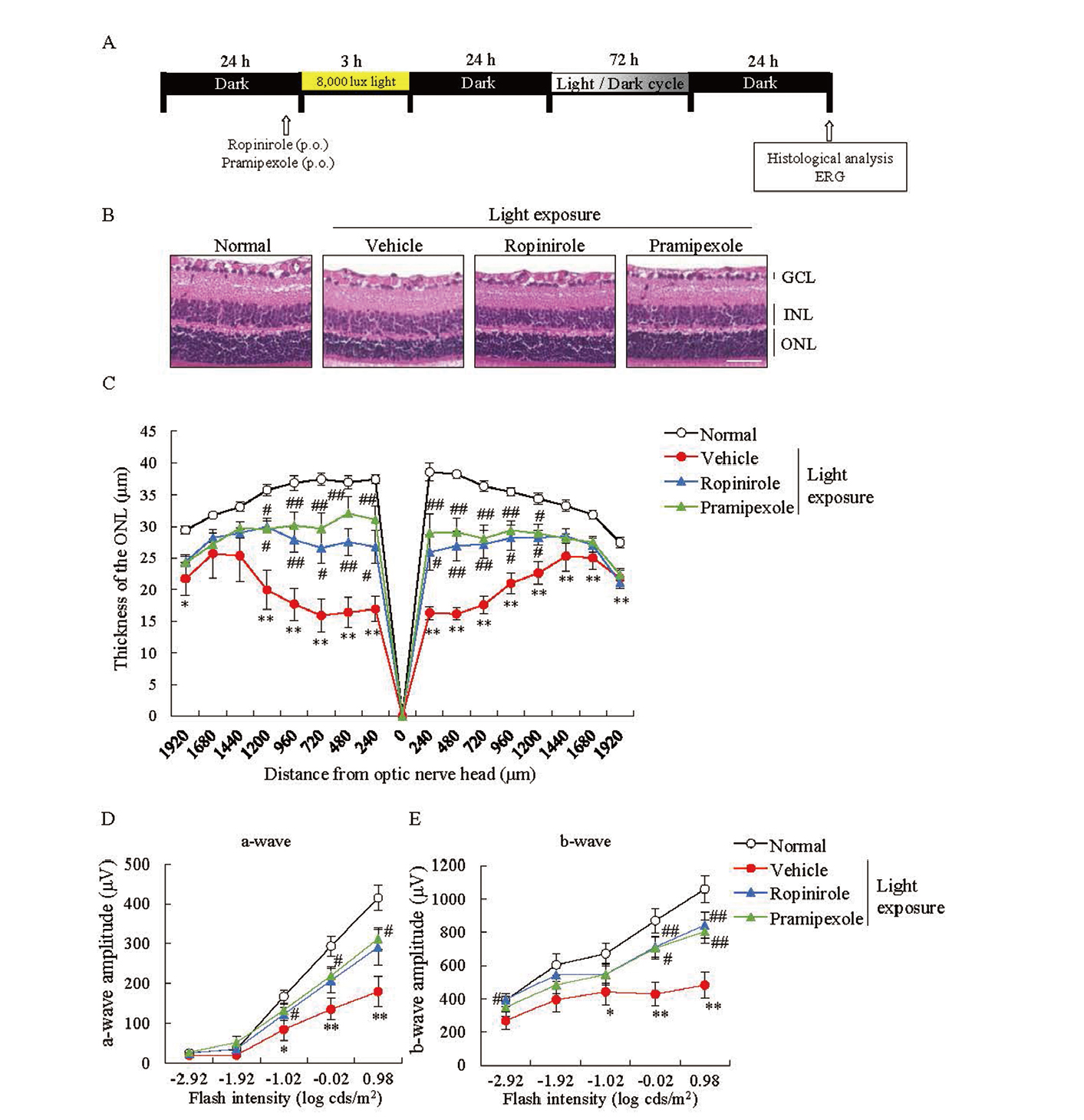
Acute Light-Induced Retinal Damage was Rescued by the Oral Administration of Ropinirole or Pramipexole
(A) The experimental design for the murine retinal degeneration model. Thirty minutes before light exposure, the eyes of the mice were dilated. Mice were then treated with either ropinirole (80 mg/kg) or pramipexole (1.0 mg/kg) by oral administration, just before light exposure. Mice were exposed to 8,000 lux white fluorescent light for 3 h. Five days after light exposure, ERG measurements and H&E staining were performed. (B-C) Histological analysis of the outer nuclear layer. (B) Representative images of retina stained with H&E staining. Scale bar = 50 μm. (C) The quantitation of ONL thickness after light exposure. Ropinirole and pramipexole treatments suppressed the thinning of the ONL. Data are shown as the mean ± SEM (n = 7-10). *, p < 0.05 vs. Normal; **, p < 0.01 vs. Normal; #, p < 0.05 vs. Vehicle; ##, p < 0.01 vs. Vehicle (Student’s t-test). (D-E) Average dark-adapted ERG a-wave (D) and b-wave (E) amplitudes. Data are shown as the mean ± SEM (n = 7-10). *, p < 0.05 vs. Normal; **, p < 0.01 vs. Normal; #, p < 0.05 vs. Vehicle; ##, p < 0.01 vs. Vehicle (Student’s t-test). ERG, electroretinogram; H&E staining, hematoxylin and eosin staining; GCL, ganglion cell layer; INL, inner nuclear layer; ONL, outer nuclear layer.
To evaluate the protective effects of ropinirole against chronic light-induced retinal damage, mice were subjected to blue LED (100 lux) exposure for 12 h each day for 4 weeks (Fig. 4A). After the implantation of an osmotic pump filled with ropinirole, the blood concentrations of ropinirole were measured to clarify that sufficient concentrations of ropinirole were present in the blood. The blood concentrations of ropinirole reached to 2.93-8.96 ng/mL (Table 1) with daily administration of ropinirole 2 mg/kg/d. The continuous administration of ropinirole suppressed the histological changes induced by chronic light exposure (Fig. 4B-C).
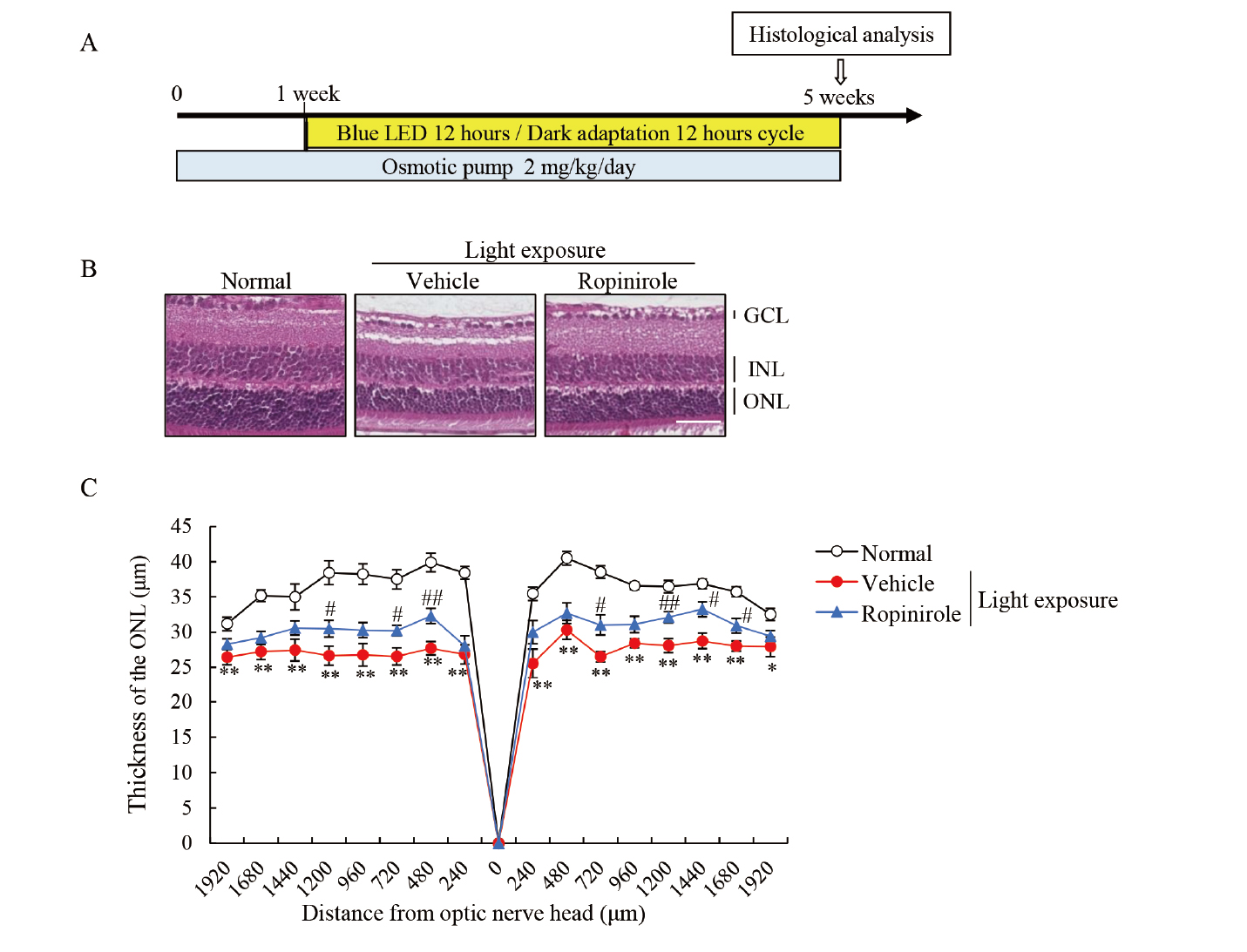
Chronic Light-Induced Retinal Damage was Rescued by Continuous Ropinirole Administration
(A) The experimental design for the chronic retinal degeneration model. Ropinirole (2 mg/kg/d) was continuously administrated by the implantation of an osmotic pump. One week after the implantation, mice were exposed to a 12-h blue LED (100 lux)/12-h dark cycle for 4 weeks. Finally, a histological analysis was performed using H&E staining. (B) Representative images of retina stained with H&E staining. Scale bar = 50 μm. (C) Quantitative analysis of the ONL in mice implanted with an osmotic pump filled with ropinirole. Data are shown as the mean ± SEM (n = 8-10). **, p < 0.01 vs. Normal; #, p < 0.05 vs. Vehicle; ##, p < 0.01 vs. Vehicle (Student’s t-test). H&E staining, hematoxylin and eosin staining; GCL, ganglion cell layer; INL, inner nuclear layer; ONL, outer nuclear layer.
| Time | 1 d | 7 d | 14 d | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Concentration (ng/mL) | 2.93 | ± | 0.44 | 6.53 | ± | 0.78 | 8.96 | ± | 3.11 |
Data are shown as ± SEM (n = 5)
Ropinirole has been reported to exert neuroprotective effects in some studies.11,12,15) However, the effects of ropinirole against photoreceptor cell degeneration have not been determined, especially under the circumstances of AMD pathology. In the present study, we provided evidence that the oral or continuous administration of ropinirole could prevent the detrimental effects associated with light exposure, and these favorable effects were observed in both acute and chronic retinal damage models (Figs. 3 and 4).
In the present study, the protective effects of ropinirole were confirmed using an in vitro cell death assay (Fig. 2). Ropinirole, at a concentration of 0.01 μM (2.97 ng/mL) prevented photoreceptor cell death (Fig. 2). Previous reports showed that dairy administration of ropinirole has neuroprotective effects at 1.0 or 2.0 mg/kg/d12,18) in mice. When ropinirole (2.0 mg/kg/d) was continuously administered by an osmotic pump, the blood concentration was measured at 2.93-8.96 ng/mL (Table 1). These data indicate that a continuous administration of 2.0 mg/kg/d ropinirole using osmotic pump resulted in a sufficient amount of ropinirole in the blood. Because ropinirole is known to be able to cross the blood-brain barrier,19) the blood level of ropinirole in mice with osmotic pump implantation might be sufficient to exert direct protective effects on photoreceptor cells. Moreover, we used the osmotic pump implantation to develop ropinirole as a patch formation for patients with dry AMD.
Oxidative stress is involved in the disease progression of AMD, and we have previously confirmed that NAC, an exogenous antioxidant, could dramatically attenuate light-induced retinal dysfunction,4) which suggested a pivotal role for oxidative stress in AMD pathology. Non-ergot derivatives, including ropinirole and pramipexole, have been reported to present antioxidant activity.14) The present results revealed that ropinirole suppressed cell death in a photoreceptor cell line (Fig. 2), and prevented the loss of retinal functions and histological changes in mice (Figs. 3-4). These data indicated that ropinirole might exert neuroprotective effects via antioxidative properties.
Dopamine acts as a key neurotransmitter in the retina.20) Dopamine in the retina is released by one subtype of retinal neuron, the dopaminergic amacrine cell, and it activates dopamine receptors. In patients with Parkinson’s disease, who are characterized by the loss of dopaminergic neurons in the brain, the increased prevalence of visual impairments has been noted.21) In Parkinson’s disease patients, dopamine levels decrease in the retina, as well as within the striatum.22) These findings indicated that dopamine in the retina plays an important role in maintaining the visual system. Although ropinirole and pramipexole have different binding affinities for the D3/D2 dopamine receptor,7) the present study showed that retinal damage was improved by both of these drugs. Taken together, retinal dopamine-modulation therapy, using either ropinirole or pramipexole, may represent a novel therapeutic approach for the treatment of retinal degeneration.
In conclusion, we clarified that the potential efficacies of ropinirole in both in vitro and in vivo dry AMD models. Therefore, these findings indicate that ropinirole may be a new candidate drug for dry AMD treatment.
We thank Anri Nishinaka, Kei Takahashi and Mayu Moriguchi for providing excellent technical assistance.
Conflict of interestH.H. received a research grant from Hisamitsu Pharmaceutical Co., Inc. N.T., K.T. and Y.M. are employees of Hisamitsu Pharmaceutical Co., Inc.