2019 Volume 84 Issue 4 Pages 335-341
2019 Volume 84 Issue 4 Pages 335-341
In Xenopus laevis embryos, gastrulation involves external and internal dynamic cell movements and rearrangements. However, the exact position of the internal archenteron when the external yolk plug closes remains unknown. In this study, we observed the position of the yolk plug and archenteron during gastrulation in X. laevis. The yolk plug closed on average at a position 22° ventral to the vertical or gravity axis of the late gastrula. We also demonstrated that the proximal end of the archenteron cavity, the gastrocoel, was always located on the gravity axis, although the shape and length of the archenteron varied among embryos. This is the first report showing that the position of the archenteron can be determined by the position of the proximal end of the gastrocoel when the yolk plug closes. In addition, time-lapse analysis of live embryos revealed that embryo rotation changed the position of the future neural tube between the late gastrula and neurula stages. Our results present an intermediate model between the classical model and a recently proposed model for amphibian gastrulation.
Gastrulation in amphibians progresses by extensive morphogenetic cell movements and rearrangements. In the frog, at the beginning of gastrulation, bottle cells change shape and invaginate to form the blastopore just below the equator on the future dorsal side of the embryo. The dorsal lip of the blastopore is the site of the Spemann organizer in amphibians and plays a central role in the establishment of the body axis in vertebrates (Carron and Shi 2016). The blastopore forms a ring around the yolk plug. Cells in the animal region spread downward to the vegetal region and cover the whole embryo by means of epiboly. During this, the yolk plug becomes smaller. While the diameter of the yolk plug diminishes to below one-quarter of the embryo diameter, the anteroposterior axis of the embryo extends so that the yolk plug is located at the posterior end. The line that becomes the neural groove then appears on the dorsal side of the yolk plug along the midline of the embryo surface. The yolk plug is eventually internalized as the embryo grows longer and develops into the neurula, in which the neural tube forms (Keller 1975, 1986, Keller et al. 1992).
If we concentrate on dynamic internal changes in the embryo, cells in the marginal zone move inside the gastrula through the blastopore and involute under the roof of the blastocoel, traveling toward the future anterior region of the embryo to form the archenteron. The blastocoel is reduced in size with the movement of these cells. During this process of gastrulation, there are dramatic cell movements and rearrangements inside and outside the embryo. To date, there are no reports showing the exact position of the archenteron as the yolk plug closes in the late gastrula stage.
Here we examined the position of the yolk plug in X. laevis gastrulae. Embryos were bisected to determine the position of the archenteron geometrically at the time when the yolk plug closes. In addition, embryos were observed continuously using live time-lapse imaging from the early gastrula to the neurula stages. Our observations demonstrate for the first time that the proximal end of the cavity of the archenteron—the gastrocoel—is on the gravity axis of the late gastrula when the yolk plug closes ventrally, just before rotation of the Xenopus embryo.
Adult X. laevis were purchased from Watanabe Zoushoku (Hyogo, Japan). Embryos were obtained, fertilized and dejellied by standard methods (Sive et al. 2000).
Time-lapse photographyEmbryos from the late gastrula to early neurula stages were incubated at 23°C in a glass petri dish containing 0.1×Marc’s Modified Ringers solution (MMR) with their positions fixed. Photomicrographs were taken from the top of embryos every 10 min using a digital optical microscope (Nikon ECLIPSE Ni-U, Tokyo, Japan), or from the bottom of embryos using a digital optical microscope (Nikon SMZ745T).
Bisection of embryosEmbryos were kept at 23°C in 0.1×MMR solution, selected once every hour from the early gastrula stage to early neurula stage, fixed for 24 h at 23°C with 5% formalin in 0.1×MMR solution and then stored at 4°C. Fixed embryos at the desired stage were embedded in 4% agar (Wako Pure Chemical Industries, Ltd., Osaka, Japan) containing 0.1×MMR. Then, embryos were bisected sagittally through the center of the yolk plug with a razor under a digital optical microscope (Nikon SMZ745T). The diameter of the yolk plug of each gastrula was measured using NIS-elements software (Nikon) and NIH ImageJ (https://imagej.nih.gov/ij/). The angle from the center of the yolk plug to the gravity axis of the bisected embryo was calculated by elementary geometry, using the following formula:
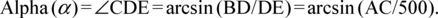 |
The position of the center of the yolk plug was defined as E. The angle from the center of the yolk plug to the gravity axis (CD) of the bisected embryo was defined as α. The radius of the embryo was assumed to be 500 µm (Fig. 1).
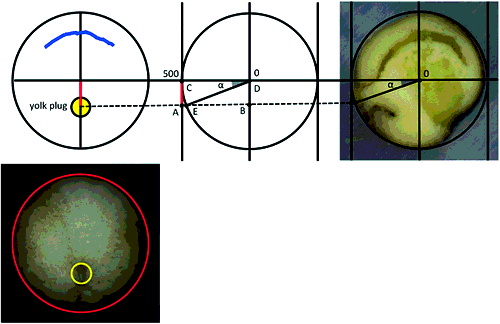
To investigate where the yolk plug closes during gastrulation, we first observed the closing direction of the yolk plug in embryos from the early gastrula to neurula stages by live time-lapse imaging. In embryos observed from the vegetal pole, the circular yolk plug formed (Fig. 2A) and occupied the vegetal hemisphere at the early gastrula stage (Fig. 2B). As the yolk plug decreased in size, its dorsal lip moved towards the opposite side (Fig. 2A–F, yellow arrow; Movie S1). In many embryos, the ventral lip of the yolk plug remained in the same position relative to the blastopore, while the circular yolk plug became smaller (Fig. 2A–F, pink arrowhead).
We then observed the position of the yolk plug from the vegetal pole just before it completely closed in live embryos. Viewing the vegetal hemisphere as a circle, we placed the center of the blastopore at stage (st.) 10.5 and that of the embryo on the diameter to investigate the angle of the yolk plug relative to the blastopore at the late gastrula stage (Fig. 2G). We used a vertical diameter (the line connecting 12 and 6 o’clock) and divided this circle into eighths (A–H clockwise), as shown in Fig. 2G. Assigning 12 o’clock as 0°, in many embryos, the center of the yolk plug closed at 180–225° from the center of the blastopore at st. 10.5; that is, the center of the yolk plug closed in area E (55%, Fig. 2G, H). In some embryos, the center of the yolk plug closed at 135–180° from the center of the blastopore; in area D (35%, Fig. 2G, H). In one embryo the yolk plug closed in area A (5%, Fig. 2G, H) and in another embryo it closed in the area H (5%, Fig. 2G, H). On average, the center of the yolk plug closed at 183.7° from the center of the blastopore at st. 10.5 (3.7° clockwise from the line through the center of the blastopore and the circle) at the late gastrula stage. Therefore, the center of the closing yolk plug was located within areas D to E (Fig. 2G, orange area, 2I, n=20). The yolk plug closed on the ventral side of the embryo.
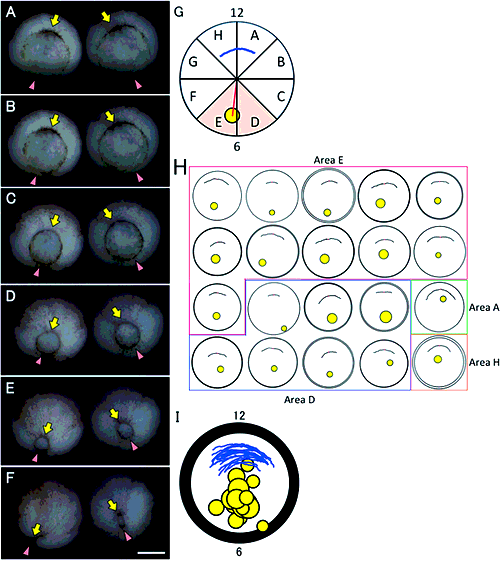
Morphological and positional changes in the archenteron during gastrulationTo examine morphological changes in the archenteron within the embryo when the yolk plug becomes smaller, embryos were bisected and the archenteron together with the blastocoel was observed. For example, when an 800-µm diameter yolk plug was situated at the vegetal pole, the archenteron with a closed or open gastrocoel involuted along the surface of the embryo (Fig. 3A, white arrowhead). The blastocoel in this embryo was large (Fig. 3A black arrow). When a 500-µm diameter yolk plug was located at the vegetal pole, the gastrocoel was enlarged, although in some embryos it remained closed (Fig. 3B, white arrowhead). The blastocoel became smaller in these embryos (Fig. 3B, black arrow).
Just before the yolk plug closed completely, at 200 µm in diameter, the archenteron extended to various degrees. In some embryos, the archenteron tip extended 45° beyond the equatorial plane and the gastrocoel opened like a deformed crescent (Fig. 3C; white arrowhead). In some embryos, the tip of the archenteron extended to the animal pole and the gastrocoel had spread into the dorsal half of the embryo (Fig. 3D; white arrowhead). The blastocoel became much smaller at this stage (Fig. 3C, D, black arrow). Therefore, for a given yolk plug size, the morphology of the archenteron varied among embryos during gastrulation.
To investigate elongation of the archenteron when the yolk plug became smaller, we measured the diameter of the yolk plug and then bisected embryos to measure the length of the archenteron. We measured the distance between the blastopore and the tip of the archenteron along with the curve of a closed gastrocoel or the external curve of an open gastrocoel to determine the length of the archenteron (Fig. 3E; between the white arrow and the black arrow). For simplicity, the diameter of the yolk plug was classified into five size groups: (1) >1000 µm, (2) 999–700 µm, (3) 699–400 µm, (4) 399–100 µm, and (5) <99 µm.
Group (1)>1000 µm diameter: archenteron length was 38–566 µm (n=59, mean±standard deviation, 179±127.6 µm). Group (2) 999–700 µm diameter: archenteron length was 57–1283 µm (n=80, 561±315.5 µm). Group (3) 699–400 µm diameter: archenteron length was 623–1705 µm (n=72, 1189±216.3 µm). Group (4) 399–100 µm diameter: archenteron length was 981–2264 µm (n=56, 1520±225 µm). Group (5) < 99 µm: archenteron length was 1302–2642 µm (n=93, 1890±273.3 µm) (Fig. 3F). Thus, for a given yolk plug size, the archenteron length varied among the embryos, as reported previously (Ewald et al. 2004). We also found that the yolk plug diameter and archenteron length varied at any given time point, even when the eggs had been fertilized at the same time.
The relationship between the diameter of the yolk plug and archenteron length was investigated by correlation analysis (n=360). Although archenteron length varied among the embryos for a given yolk plug size, the significant correlation coefficient of 0.94 (p<0.05) indicated that the archenteron became longer as the yolk plug diameter became smaller (Fig. 3F).
To examine the position of the archenteron just before the yolk plug had closed completely with a diameter of 200 µm, we measured the distance between the centers of the embryo and the yolk plug for the geometric calculation (Figs. 1, 2G, red line). When the radius of the embryo was assumed to be 500 µm, the distance between the center of the embryo and the center of the yolk plug was 184 µm on average (n=18, except for those in areas A and H). When the distance between the center of the embryo and center of the yolk plug was 184 µm, the angle of the yolk plug to the gravity axis (angle α in Fig. 3G) was geometrically 21.59°; that is, the center of the yolk plug was situated in a position 21.59° (on average) ventral to the gravity axis of the bisected embryo (n=18, Figs. 1, 3G). Therefore, the yolk plug closed at a position 22° ventral to the gravity axis of the late gastrula.
To investigate the position of the archenteron, we bisected late gastrulae, set the center of the yolk plug at a position 22° ventral to the gravity axis (Fig. 3G, black arrowhead, 3H, white arrowheads), and observed the inside of the embryo. During this period, the proximal end of the gastrocoel was almost always located on the gravity axis (Fig. 3H, black arrowheads). Although the morphology and length of the archenteron varied among gastrulae, the proximal end of the gastrocoel was invariably situated on the gravity axis at the late gastrula stage.
The rotation of embryos between late gastrulae and early neurulaeTo observe the embryonic development from early gastrula to neurula under normal gravity conditions, live time-lapse imaging was performed from the upper water surface side (animal pole) or from the underside looking upward (the vegetal pole) in a glass petri dish. The embryos rotated while the yolk plug was closing (Movie S1). When embryos were observed from the underside looking upward, a linear structure formed at the surface of the embryo as the yolk plug closed (Movie S1). Many embryos rotated from the anterior side of this linear structure. When rotation was complete, at least half of the linear structure was observed from the upper water surface side (Movie S2). While embryos with a closed yolk plug rotated, the speed of rotation accelerated during the last third of this process. These results indicated that embryos had formed the future neural groove on the underside looking upward and then rotated so that most of the neural tube eventually faced the water surface.
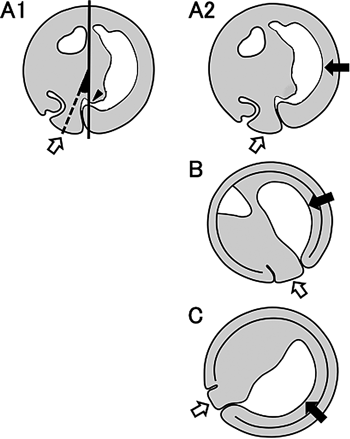
In this study, we examined the morphology and positions of the yolk plug and archenteron during gastrulation in X. laevis embryos. We found that the yolk plug closed on the ventral side at 22° to the gravity axis (Fig. 4A1, white arrow). At this time, the proximal end of the gastrocoel was always located on the gravity axis (Fig. 4A1, black arrowhead). This is the first report showing that in X. laevis embryos, the proximal end of the gastrocoel is invariably located on the gravity axis of the late gastrula with the closing yolk plug, although the morphology and length of the archenteron varied among the embryos. This finding suggests that the position of the archenteron can be determined by the position of the proximal end of the gastrocoel in the late gastrula. Keller (1975) reported that the blastopore closed at a point 18–22° ventral to the vegetal pole of st. 10, the blastula. In this study, the yolk plug closed on average at 22° ventral to the gravity axis. Keller also arbitrarily defined the animal and vegetal poles and found that a line through these poles was approximately normal to the gravitational equatorial plane of the embryo. Vogt (1925) reported that normal early development proceeded under the gravity axis at least until the blastula stage. These findings imply that the animal–vegetal axis might be the same as the gravity axis. Keller examined the position of the yolk plug while embryos were held with the desired hemisphere uppermost by flat glass bridges pushed against the vitelline membrane. We simply observed the yolk plug position and also normal development under natural gravity conditions from the vegetal pole viewpoint during gastrulation. Whether the arbitrarily defined animal–vegetal axis is the same as the gravity axis should be investigated rigorously in future studies.
We also observed embryos from the gastrula to neurula stages under natural conditions by live time-lapse imaging. Embryos formed the future neural groove on their underside looking upward and then rotated at the early neurula stage, and eventually, the neural tube formed on the upper water surface side. It should be noted that embryos form the future neural groove on their underside looking upward, not in the entire animal pole region. The rotation causes the neural tube to become situated on the upper water surface side or the side perpendicular to the water surface. From our observation of bisected Xenopus embryos with the yolk plug set at the right position, before rotation, the archenteron was involuted, then elongated and formed its cavity on the dorsal side. The blastocoel moved away from the internal surface around the pole. The elongating archenteron formed the large cavity and, around the same time as the blastocoel became smaller. The archenteron region may get lighter with the heavier region becoming situated on the opposite side. Consequently, the altered weight distribution may cause rotation physically, by changing the gravity balance inside of the embryo. It is still unclear whether this rotation is indispensable for early development. Black et al. (1996) reported that it might not be required for early development of Xenopus embryos because neural plate development was unaffected if rotation was prevented. It is conceivable that the embryo rotation could occur physically shortly after the proximal end of the gastrocoel moves to the gravity axis.
Concentrating on embryos with closing yolk plugs, we note that there are major differences between the current and classical models of amphibian gastrulation. In the classical model, the yolk plug closes on the dorsal side, as the ventral edge of the yolk plug moves to the dorsal side (Fig. 4B, white arrow) and the archenteron extends from the blastopore to the animal pole (Fig. 4B, black arrow). The tip of the archenteron extends to the ventral side beyond the animal pole and lies exclusively within the entire animal region, or else it reaches towards the animal pole and is situated on the dorsal side along the animal–vegetal axis (reviewed by Beetschen, 2001). In contrast, here we show that the yolk plug closes on the ventral side near the vegetal pole, as the dorsal edge of the yolk plug near the blastopore moves to the opposite side (Fig. 4A2, white arrow), and the archenteron forms across both the animal and vegetal regions on the dorsal side (Fig. 4A2, black arrow). Thus, the ventral position of the closing yolk plug observed in the current model differs from the dorsal side in the classical model.
Gastrulation in X. laevis embryos as proposed by Koide et al. (2002) and Yanagi et al. (2015) differs from that in the classical model. In Koide’s model, the blastopore moves toward the vegetal pole, consistent with our observation. As gastrulation progresses, the yolk plug closes near the equatorial plane on the ventral side (Fig. 4C, white arrow) and the archenteron forms in the vegetal region (Fig. 4C, black arrow). Here we present an intermediate model between the classical model and Koide’s model. In our model, as the yolk plug closes vegetally near the gravity axis, not near the equatorial plane, the archenteron is situated across the animal and vegetal regions on the dorsal side (Fig. 4A2). When the proximal end of the gastrocoel is on the gravity axis, the blastopore might also be located on the gravity axis, which could possibly be the animal–vegetal axis at this time (Fig. 3H). This is consistent with Vogt’s report (1925) that the closed blastopore is situated on the opposite side of the animal pole. It is also consistent with the formation of the neural tube on the dorsal side. By rotation, the embryo forms the neural tube on the upper water surface side. If there is no embryo rotation in our model, the neural tube should form on the underside looking upward. In the classical model, with the yolk plug on the dorsal side, the neural tube should form on the underside looking upward by rotating. However, our observation demonstrated that the embryo rotated and eventually formed the neural tube on the upper water surface side.
In the current model, the archenteron is situated around the animal and vegetal poles. It is conceivable that the embryo of the classical model rotates 45–90° together with the animal–vegetal axis. However, it is unlikely that the embryo actually rotates 45–90°. When the anterior of the archenteron involutes over the equatorial plate, the blastocoel occupies almost the entire animal region of the embryo on the ventral side. Just before the yolk plug closes, the blastocoel moves inward and becomes smaller, but its position does not change. If the animal–vegetal axis rotates to the dorsal side as gastrulation proceeds, the position of the blastocoel should move to the dorsal side. Therefore, gastrulation proceeds on the dorsal side. To rigorously investigate in what direction the archenteron involutes to the animal–vegetal pole, we need to label the two poles (hemispheres) and observe the inside of the labeled gastrula continuously. Such analyses will elucidate the relationship between the direction of the body axis and the formation of the archenteron during early development.
After fertilization, the Xenopus embryo develops with a less yolky animal region on the upper water surface side compared with the yolky vegetal region on the underside looking upward. In this study, we found for the first time that the proximal end of the Xenopus gastrocoel is invariably on the gravity axis of the late gastrula with a closing yolk plug. Based on our observations of the embryo, we propose the following process of Xenopus gastrulation. When gastrulation begins, the blastopore involutes below the equator. The dorsal lip of the blastopore moves to the ventral side and the yolk plug gradually becomes smaller. The archenteron elongates and extends on the dorsal side. The archenteron forms a cavity (the gastrocoel), and around the same time as the blastocoel in the animal region begins to diminish. The gastrocoel expands, and around the same time as the blastocoel in the animal region becomes much smaller. The proximal end of the gastrocoel is situated on the gravity axis as the yolk plug closes ventrally. Changes in spatial distribution inside the embryo occur in the direction of gravity. The future neural groove begins to form in the dorsal region, on the underside looking upward. Then, the embryo physically rotates to achieve gravity balance. As a result, the neural tube mainly faces the water surface at the neurula stage.
With Supplementary files of Movies S1 and S2.
We thank Mr. K. Hashigoe, Mr. S. Hashioka, Dr. H. Haramoto, Dr. G. Nakamoto (Ehime University, Japan), Dr. K. Matsumoto (Ehime University Senior High School, Japan), Dr. F. Prodon (University of Geneva, Switzerland), Mr. T. Higuchi (INOHARA & Co., Ltd.), Mr. N. Nishida (Ehime Institute of Industrial Technology Paper Technology Center, Japan), Dr. C. Kikuchi (MKC Consulting), and Dr. M. Nakamura for their help and advice. This work was supported by a grant from the dean office of our graduate school. We thank Dr. G. Quinn, Charles Allan, Ph.D., and James M. Cummins Ph.D. from Edanz Group (www.edanzediting.com/ac) for editing drafts of this manuscript.