2013 Volume 53 Issue 6 Pages 1057-1061
2013 Volume 53 Issue 6 Pages 1057-1061
In order to produce 316L stainless steel separators with low contact resistivity and low cost, an attempt was made to reduce the contact resistivity of 316L steel by acid treatment. In the present work, the contact resistivity and corrosion resistance of acid-treated 316L stainless steels were investigated. Elemental composition, thickness and structure of the passive film on the steel surface were analyzed with transmission electron microscopy (TEM) and X-ray photoelectron spectroscopy (XPS). It is found that the contact resistivity of the stainless steel is closely related to the ratio of Fe in oxide-form in the passive film, irrespectively of the thickness of passive film. When the proportion of Fe in oxide-form to the sum of Fe and Cr in oxide-form, i.e. Feox/(Feox+Crox) is lower than ca. 40 at%, the contact resistivity is reduced to lower than 10 mΩ cm2. Besides, the corrosion resistance of the acid-treated stainless steel having low contact resistivity is very high. Power generation test shows that I – V characteristic and durability of the cell assembled using the acid-treated 316L steel separators of low contact resistivity are equivalent to those of the cell assembled using carbon-coated separators.
Separators used in polymer electrolyte fuel cells (PEFCs) are required to have good electrical conductivity, gas impermeability, mechanical precision for gas-tightness and sufficient corrosion resistance. Carbon composite has good chemical stability in a fuel cell environment,1) but the carbon separators occupy considerably volume and cost in PEFC stacks. Thinner carbon separator has a problem in their strength. Since a metallic separator is superior in strength as compared to a carbon separator, a thinner separator made of metal is possible. Researchers expect that PEFC cell stacks will be made more compact by the use of thinner metallic separators.
Stainless steels are corrosion resistant alloys because of the formation of a passive film on their surface, but this passive film has also high electric resistivity that causes an increase of contact resistance between the stainless steel plate and the membrane electrolyte assembly (MEA).
A way to reduce the contact resistivity is gold coating onto stainless steels.2,3,4,5) To give a good adhesion to the gold coating on the surface of stainless steels, the passive film on the surface must be removed, and the passive film is usually removed before or during the gold coating process.6) However, noble metals such as gold are so expensive that gold coating is hard to be practically adopted in terms of cost. Besides, there is a problem of pin-hole corrosion which causes dissolutions of metal ions which contaminate electrolyte membrane or catalysts, since it is difficult to obtain a perfect gold coating without any defect such as pin-hole. Once there is a pin-hole or small scratch reaching the substrate of stainless steels, galvanic corrosion may occur between gold coating and stainless steels. To avoid the galvanic corrosion, carbon coating is proposed instead of gold coating;7,8,9,10,11,12) however, the cost of carbon-coated separators is still considerably high.
To produce the stainless steel separators of low cost, reducing the electric resistivity of passive film by modifying the structure and/or composition of surface passive film is an effective way. Recently, a technique has been proposed,13) by which electric resistivity of passive film can be reduced via the doping of fluoride into the passive film, but the mechanism is unclear. Chromium can be enriched in the passive films of stainless steels as a result of selective dissolution during polarization,14,15) but the effect of alloying elements on the electric resistivity of passive film is not understood, although a research work suggests that the elements such as Cr may have an influence on the contact resistivity of stainless steels.16)
In our previous works, we found that acid treatment of stainless steels may reduce the contact resistivity to such an extent that it can be employed to produce stainless steel separators for PEFC, and this acid treatment is very low-cost. In the present study, the contact resistivity and corrosion resistance of acid-treated 316L stainless steels are investigated, and the elemental composition and thickness as well as structure of passive film on 316L stainless steel surface are analyzed with X-ray photoelectron spectroscopy (XPS) and transmission electron microscopy (TEM). The factors affecting the contact resistivity is discussed. Besides, power generation test using a PEFC short stack assembled with the acid-treated 316L stainless steel separators of low contact resistivity is carried out to confirm the power generation performance and durability of the cell prepared with the acid-treated 316L stainless steel separators.
Materials used in the present study were two commercially-available 316L stainless steel sheets, which were manufactured by different steel makers, marked as steel A and steel B hereinafter in this paper. The thickness of the two stainless steel sheets was 0.2 mm, and their surface was finished by bright annealing. The two stainless steel sheets had almost the same chemical compositions as shown in Table 1.
| C | Si | Mn | P | S | Cr | Ni | Mo | |
|---|---|---|---|---|---|---|---|---|
| Steel A | 0.010 | 0.66 | 1.17 | 0.034 | 0.001 | 17.40 | 12.14 | 2.07 |
| Steel B | 0.013 | 0.53 | 0.98 | 0.027 | 0.002 | 17.46 | 12.54 | 2.09 |
Before characteristic investigation, the two stainless steel sheets were subjected to the same acid treatment. Firstly, the stainless steel sheets were degreased and immersed into the mixture of 1% of fluoric acid and 10% of nitric acid at 55°C for 5 min, and followed by rinsing in water, in order to remove the old passive film formed during steel manufacturing process. Subsequently, these sheets were immersed into 20% of nitric acid at 55°C for 5 min, then rinsed in water and dried in the air to produce new passive film on the surface of stainless steel sheets.
2.3. Contact Resistivity MeasurementContact resistivity between the carbon paper (0.20 mm thickness) and the stainless steel was measured with the 4-wire method. The pressure of about 1.0 MPa was applied during measurement. The details of this measurement were described elsewhere.12)
In order to investigate the change of contact resistivity with elapsed time after acid treatment, the contact resistivity of these specimens was measured before and after they were exposed to air for a certain period. The contact resistivity measurement was performed one day before XPS analysis. The relationship between contact resistivity and composition of passive film was investigated on the basis of contact resistivity measurement and XPS analysis.
2.4. Analyses of Passive FilmThe XPS analyses were performed by using Quantum-2000 system (ULVAC-PHI, INC.) with a Al-Kα line and an electron beam for the X-ray source and neutralizing, respectively. Ar sputter etching was not performed before the XPS analysis.
The passive film on stainless steel surface was also observed using a transmission electron microscope (JEOL: JEM-4000EX). The acceleration voltage was 400 kV. Thin foils for TEM were prepared by using a focused ion beam instrument.
2.5. Anodic PolarizationAnodic polarization test was performed using a Solartron Model 1280C potentiostat to investigate the corrosion resistance of the stainless steels. The specimens were immersed into 500 cm3 of 0.5 M H2SO4 aqueous solution, and the exposure area was 1 cm2. The polarization sweep was started at the rest potential with a scan rate of 20 mV min–1. The counter and reference electrodes were Pt plate and saturated Ag/AgCl, respectively. All measurements were performed at room temperature.
2.6. Power Generation Test Using PEFC Short StackA cell was assembled with commercially-available MEA and a pair of separators (i.e. anode separator and cathode separator) that made from acid-treated 316L steel (steel A). For comparison, another cell was also assembled with MEA and a pair of carbon-coated 316L steel separators. The details about the carbon-coated separators were reported previously.12) For each cell, the amount of catalyst was 0.4 mg cm–2 and the thickness of electrolyte membrane was 20 μm, while the electro-active area was 50 cm2. The two cells were connected in serial to form a PEFC short stack. During power generation test, hydrogen gas without humidity was fed to anode at a flow rate of 2.08×10–6 m3 s–1 (0.125 L min–1), and ambient air was fed to cathode at a flow rate of 1.88×10–4 m3 s–1 (11.25 L min–1) for each cell. These gases were fed at atmospheric pressure, and the test was conducted at room temperature (ca. 23°C). The electrical current and voltage (i.e. the I – V characteristic) of each cell were measured during power generation test, and the durability of the cells was tested for about 1000 hours by monitoring the change of cell voltage while keeping current density constant at 0.1 A cm–2.
The contact resistivity of acid-treated steel A and B after exposed to air for various times is summarized in Fig. 1. Before the acid treatment, the contact resistivity of both steel A and B is in the range of 100–800 mΩ cm2. After the acid treatment, the contact resistivity of steel A decreases to lower than 10 mΩ cm2, and keeps unchanged over 240 days in the air. The contact resistivity of steel B is also reduced after acid treatment, but the value is higher than 20 mΩ cm2 and increases rapidly with elapsed time. Within one month in the air, the contact resistivity of steel B becomes higher than 100 mΩ cm2. Although the acid treatment process for both steel A and B is the same, the contact resistivity of steel A and B acid-treated is completely different. This is probably due to the difference in the ratios of Fe, Cr, Ni and Mo elements on the steel surface between the two steels as described later in section 3.3. The surface roughness Ra of both steel A and B was ca. 0.06 μm, and did not change before and after acid treatments. Therefore, it could be thought that the surface roughness was not the principal factor determining the contact resistivity of the steels in the present work.
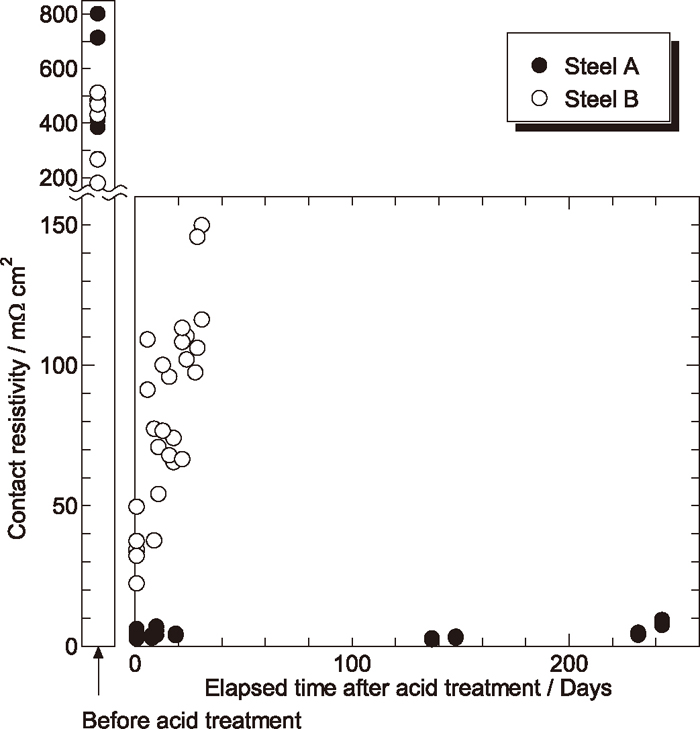
Change of contact resistivity of acid-treated steel A and B with elapsed time.
Transmission electron microscopic images of passive film formed on the surface of steel A and B are presented in Fig. 2. Before acid treatment, steel A and B have passive film with thickness of ca. 3 nm on their surface (Figs. 2(a) and 2(b)). The structure of the passive film is amorphous. After the acid treatment, the thickness of passive film on the surface of steel A and B is ca. 2–5 nm (Figs. 2(c) and 2(d)). The thickness of passive film is not greatly changed even after the acid-treated specimens are exposed to air for over 100 days (Figs. 2(e) and 2(f)). These results suggest that the thickness of passive film is not the determination factor for the contact resistivity of stainless steel in the present work. Lattice structure which is observed in the passive film on the surface of acid-treated steel A and B is probably due to the existence of metallic Cr and Fe in the passive film.

TEM images of passive film formed on the surface of stainless steels: (a) steel A before acid treatment; (b) steel B before acid treatment; (c) steel A within a few days after acid treatment; (d) steel B within a few days after acid treatment; (e) acid-treated steel A after exposed to air for 227 days; (f) acid-treated steel B after exposed to air for 142 days.
To investigate the change of elemental composition and chemical state of passive film formed on the surface of steel A and B before and after the acid treatment, XPS analyses were conducted. Although the spectra of C, O, Cr, Fe, Ni and Mo regions were acquired, only metal elements (i.e. Cr, Fe, Ni and Mo) ratio was calculated. In the region of Cr 2p3/2, a peak was separated to three components, i.e. metallic Cr, Cr2O3 and Cr(OH)3, which corresponds to the peaks at 574.4, 576.9 and 577.3 eV, respectively.17) In the region of Fe 2p3/2, a peak was separated to the components of metallic Fe, FeO and Fe2O3, corresponding to the peaks at 706.8, 709.3 and 711.2 eV, respectively.17) In this paper, Cr2O3 and Cr(OH)3 are expressed as Cr oxide, and FeO and Fe2O3 as Fe oxide. The elemental compositions on the surface of passive film are shown in Fig. 3, where the XPS analysis results of the steel A and B before acid treatment, the acid-treated steel A and B after exposed to air for one day, and those after exposed to air for over 100 days are presented.
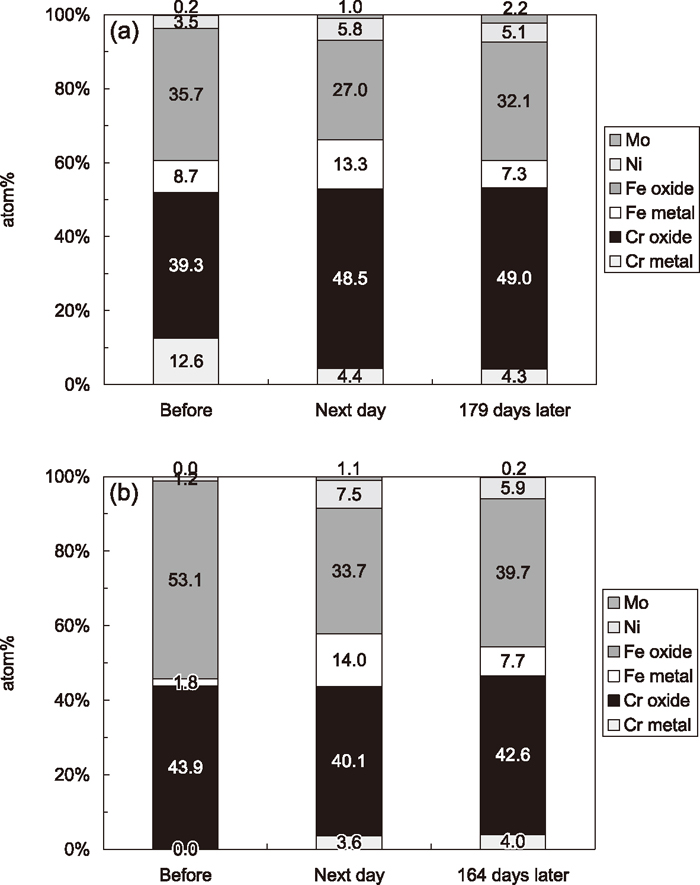
Elemental compositions on the surface of passive film decided by XPS: (a) steel A; (b) steel B (Ni includes the metallic Ni and Ni in oxide-form, and Mo includes the metallic Mo and Mo in oxide-form).
Before acid treatment, Cr enrichment in passive films formed on stainless steels is observed in both steel A and steel B, but the ratio of total Cr (in both metallic and oxide form) is higher in passive film of steel A than steel B. The compositions of passive films formed on steel A and B are slightly different although their compositions of bulk are almost the same as shows in Table 1. This is probably due to slightly difference in finishing of bright annealing in their steel maker’s factories.
By the acid treatment, the ratio of Fe in oxide-form is considerably decreased from 35.7 at% to 27.0 at% for steel A (Fig. 3(a)), and from 53.1 at% to 33.7 at% for steel B (Fig. 3(b)). The ratio of total Fe (in both metallic and oxide form) is also decreased by the acid treatment. It should be noted that acid treatment does not change the ratio of total Cr (in both metallic and oxide form) in passive films; however, the ratio of Cr in oxide-form in passive film is increased by the acid treatment for steel A, while this phenomenon is not observed for steel B.
When several months elapsed after the acid treatment, the ratio of Fe in oxide-form became higher. For steel A (Fig. 3(a)), the ratio of Fe in oxide-form was increased from 27.0 at% to 32.1 at% after exposed for 179 days. For steel B (Fig. 3(b)), it became from 33.7 at% to 39.7 at% after exposed for 164 days. The increase in the ratio of Fe in oxide-form after long period exposure is attributed to the oxidation of metallic Fe.
3.4. Factors Affecting Contact ResistivityCr and Fe oxides, which account for a large percentage of passive film and have comparatively higher electrical resistance than metallic Cr, Fe, Ni and Mo, may be the main factors affecting the contact resistivity. Statistical analysis of experimental data of contact resistivity and surface composition shows that there exists a relationship between contact resistivity and Feox/(Feox+Crox). Where, Feox and Crox mean the ratio of Fe and Cr in oxide-form, respectivety. The Feox/(Feox+Crox) may more accurately represent the ratio of Fe in oxide-form in passive film.18) Figure 4 displays the relation between contact resistivity and Feox/(Feox+Crox) in the passive film formed on various specimens obtained by XPS. The details of specimens are listed in Table 2. Before acid treatment, the contact resistivity of specimens (i.e. as received specimens), which are marked as A1, A2, A3, B1 and B2 in Fig. 4, is in a wide range of 100–800 mΩ cm2. After acid treatment, the contact resistivity is reduced, accompanied with reduction of the Feox/(Feox+Crox). It can be seen from Fig. 4 that the contact resistivity is mainly dependent on the Feox/(Feox+Crox). When the Feox/(Feox+Crox) in the passive film is reduced to less than ca. 40 at%, the contact resistivity is reduced below 10 mΩ cm2. The reasons for the dependency of contact resistivity on the Feox/(Feox+Crox) are not completely understood at present, but the high electric resistivity of trivalent Fe is probably one of the reasons.
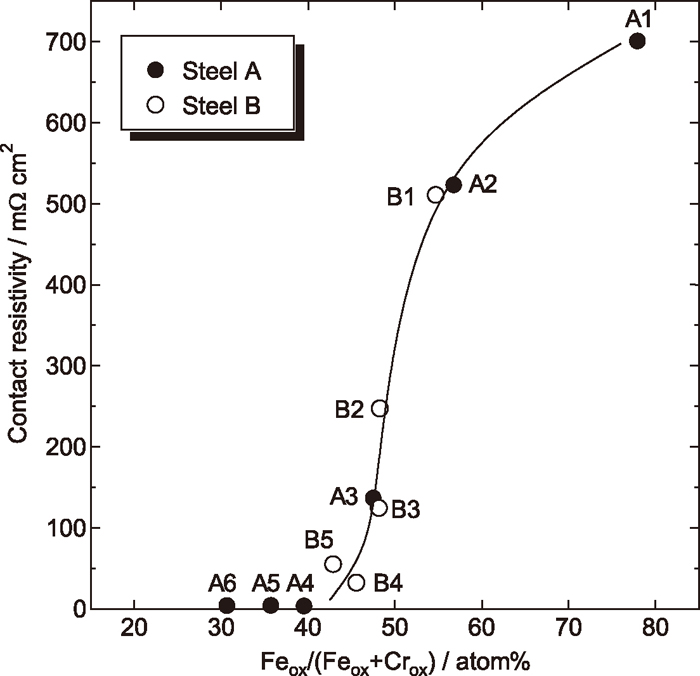
Relation between contact resistivity of 316L steel and ratio of Fe in oxide-form in passive film.
| Marks | Steel type | 1st acid treatment | 2nd acid treatment | Elapsed time |
|---|---|---|---|---|
| A1 | A | (as received) | – | – |
| A2 | A | (as received) | – | – |
| A3 | A | (as received) | – | – |
| A4 | A | 10% HNO3 + 1%HF, 55°C, 5 min | 20% HNO3, 55°C, 5 min | 179 days |
| A5 | A | 10% HNO3 + 1%HF, 55°C, 5 min | 20% HNO3, 55°C, 5 min | 1 day |
| A6 | A | 10% HNO3 + 1%HF, 55°C, 5 min | 20% HNO3, 55°C, 5 min | 133 days |
| B1 | B | (as received) | – | – |
| B2 | B | (as received) | – | – |
| B3 | B | 10% HNO3 + 1%HF, 55°C, 5 min | 20% HNO3, 55°C, 5 min | 105 days |
| B4 | B | 10% HNO3 + 1%HF, 55°C, 5 min | 20% HNO3, 55°C, 5 min | 1 day |
| B5 | B | 10% HNO3 + 1%HF, 55°C, 5 min | 20% HNO3, 55°C, 5 min | 48 days |
As described previously in Fig. 1, the contact resistivity of acid-treated steel B increases with elapsed time, while that of acid-treated steel A keeps almost unchanged. This is because that the Cr enrichment is insufficient and the Feox/(Feox+Crox) is over 40 at% on the surface of steel B, but sufficient Cr enrichment is present on the surface of steel A, making the Feox/(Feox+Crox) below 40 at% over 100 days.
3.5. Anodic Polarization CurveThe polarization curves of steel A and B before and after acid-treatment are shown in Fig. 5. The plateau region from 0.5 to 0.8 V is passivity zone. The current density in the passivity zone is greatly reduced for steel A after acid treatment, suggesting that the corrosion resistance of steel A is improved by the acid treatment. The current density of steel B in the passivity zone is not greatly changed by the acid treatment although the slope of polarization curves is altered, implying that the acid treatment has almost no influence on the corrosion resistance of steel B. It can also be seen that the current density in the passivity zone is lower for the acid-treated steel A than acid-treated steel B. That is to say, the corrosion resistance of the acid-treated steel A is superior to that of the acid-treated steel B. By comparing anodic polarization curves (Fig. 5) with surface compositions (Fig. 3), it can be found that the corrosion resistance is closely related to the ratio of Cr in oxide-form on the surface of stainless steels, and the changes in the ratio of Cr in oxide-form after acid treatment can explain the corrosion resistance variations of steel A and B. The improved corrosion resistance of acid-treated steel A results from the high ratio of Cr in oxide-form in the passive film.
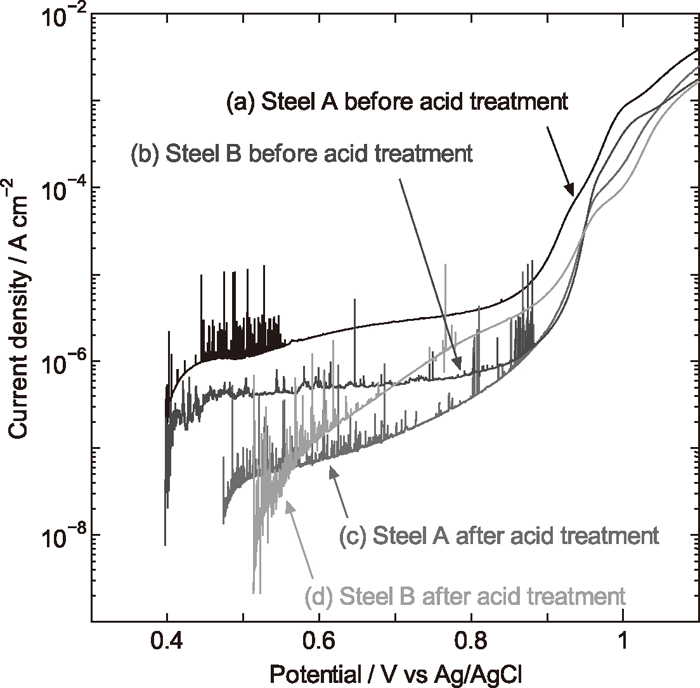
Polarization curves of stainless steels in 0.5 M H2SO4: (a) steel A before acid treatment; (b) steel B before acid treatment; (c) steel A after acid treatment; (d) steel B after acid treatment.
I-V characteristic of the cell prepared with acid-treated stainless steel separators is shown in Fig. 6, in which I-V characteristic of the cell prepared with carbon-coated separators is also presented for reference. It is obvious that the cell voltage of the acid-treated SUS316L separators is equivalent to that of the carbon-coated separators.
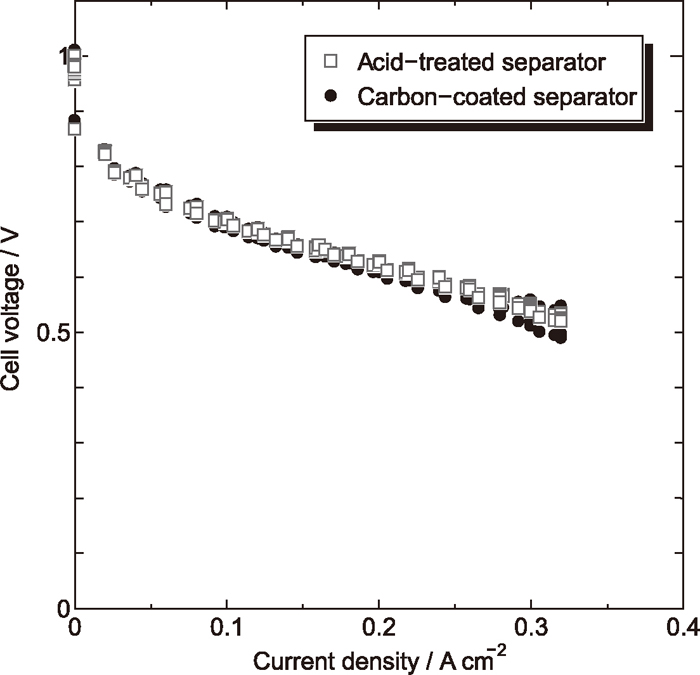
I-V characteristic of cells assembled with acid-treated 316L separators and carbon-coated 316L separators.
Figure 7 compares the durability of the acid-treated SUS316L separators with that of carbon-coated SUS316L separators. The voltage spikes, which appear about every 168 h, are due to measurement of current-voltage property. The cell of the acid-treated SUS316L separators shows a comparable durability with the cell of carbon-coated SUS316L separators over 1000 hours. After the durability test, the surface of these metallic separators had no obvious change in appearance.
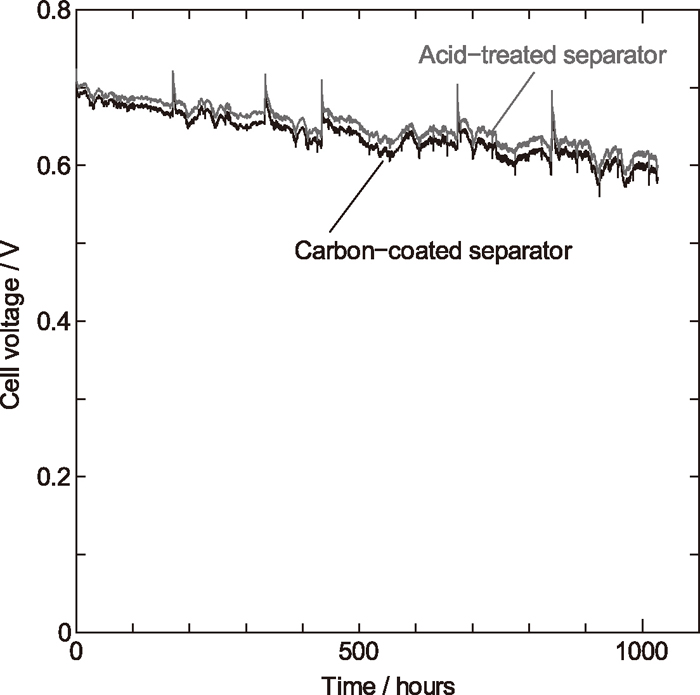
Durability test of cells assembled with acid-treated 316L separators and carbon-coated 316L separators. The constant current density is 0.1 A cm–2.
The contact resistivity and corrosion resistance of two 316L stainless steels were investigated, and the elemental composition and thickness of passive film formed on the stainless steel surface were analyzed with XPS and TEM. The contact resistivity of steel A is greatly reduced and its corrosion resistance is improved after acid treatment, and the reduced contact resistivity is almost not changed after exposed to air for over 240 days. In contrast, the contact resistivity of steel B is decreased to some extent immediately after acid treatment but rebounds back to the level before acid treatment within one month, and the corrosion resistance is not altered by the acid treatment. The contact resistivity of 316L stainless steels is closely related to the ratio of Fe in oxide-form in the passive film, irrespectively of the thickness of passive film. When the proportion of Fe in oxide-form to the sum of Fe and Cr in oxide-form, Feox/(Feox+Crox), is lower than ca. 40 at%, the contact resistivity is lower than 10 mΩ cm2. Power generation test using a PEFC short stack shows that I-V characteristic and durability of the cell assembled with acid-treated stainless steel separators are equivalent to those of the cell of carbon-coated separators. The results obtained in the present work suggest that the acid-treated 316L stainless steel separators can be used for PEFC.