巻号一覧

後続誌
36 巻, 1 号
選択された号の論文の13件中1~13を表示しています
- |<
- <
- 1
- >
- >|
新年のご挨拶
-
尾嶋 正治2015 年 36 巻 1 号 p. 1-2
発行日: 2015/01/10
公開日: 2015/01/22
ジャーナル フリーPDF形式でダウンロード (201K)
巻頭言
-
中山 武典2015 年 36 巻 1 号 p. 3
発行日: 2015/01/10
公開日: 2015/01/22
ジャーナル フリーPDF形式でダウンロード (200K)
特集: 腐食,防食の表面科学 —インフラの長寿命化に貢献する表面科学—
-
篠原 正原稿種別: 総合報告
2015 年 36 巻 1 号 p. 4-11
発行日: 2015/01/10
公開日: 2015/01/22
ジャーナル フリーMany structural metallic materials are exposed to atmospheric environments. And corrosion in these environments, so called “Atmospheric Corrosion”, commonly initiates and develops under thin water films formed by dew or rain drops. A composition of water film depends on the air pollutant deposition rate, and changes with the humidity and temperature conditions of the atmosphere. Many sensors and the measurement techniques have been developed to evaluated corrosivities in atmospheric environments and corrosion behaviors in those environments. In this paper, the present state of atmospheric corrosion researches are discussed based on the roles of water and deposits and environmental factors which are evaluated by those sensors and measurement techniques.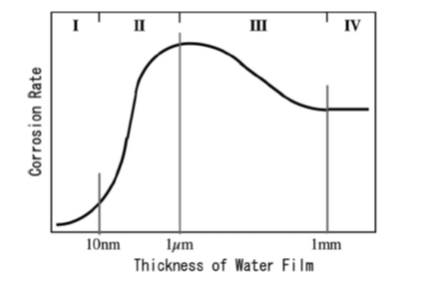 抄録全体を表示PDF形式でダウンロード (1464K)
抄録全体を表示PDF形式でダウンロード (1464K) -
大塚 俊明原稿種別: 研究紹介
2015 年 36 巻 1 号 p. 12-17
発行日: 2015/01/10
公開日: 2015/01/22
ジャーナル フリーThe corrosion of metallic materials is caused by action of aqueous or gaseous environment and the products of the corrosion greatly depend on the environmental condition. It is desired that the identification of the products is done under the in-situ environmental condition. Raman spectroscopy is one the methods to identify them under the condition. The application of Raman spectroscopy to detection of the corrosion products of steel, zinc, and titanium is introduced. 抄録全体を表示PDF形式でダウンロード (1124K)
抄録全体を表示PDF形式でダウンロード (1124K) -
千葉 亜耶, 武藤 泉, 菅原 優, 原 信義原稿種別: 研究紹介
2015 年 36 巻 1 号 p. 18-23
発行日: 2015/01/10
公開日: 2015/01/22
ジャーナル フリーStainless steels suffer from pitting corrosion in chloride-containing environments. Sulfide inclusions, such as MnS, are known to act as the initiation sites of pitting. The pit initiation mechanism at MnS inclusions in chloride-containing environments is as follows: 1) dissolution of MnS inclusions leads to the deposition of elemental sulfur on and around the inclusions; 2) the coexistence of elemental sulfur and chloride ions results in the dissolution of the steel matrix, forming the trenches at the boundaries between the inclusions and the steel matrix, 3) rapid active dissolution occurs locally at the bottom of the trenches, because of the decrease of pH due to the hydrolysis reaction of Cr3+ and the electrode potential decrease at the bottom of the trench due to the solution resistance. Dissolution behavior of sulfide inclusions is of key importance in pit initiation process. Insoluble sulfide inclusions, such as CrS, TiS, and Ti4C2S2, are unlikely to act as the pit initiation sites. Cr-or Ti-oxide films on the inclusions act as a barrier against inclusion dissolution. An applied stress causes micro-cracks on MnS inclusions, which promotes pit initiation of stainless steels. The micro-crack initiation is closely related to oxide film formation on MnS inclusions.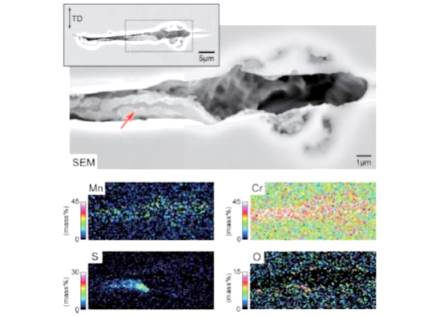 抄録全体を表示PDF形式でダウンロード (1516K)
抄録全体を表示PDF形式でダウンロード (1516K) -
片山 英樹原稿種別: 研究紹介
2015 年 36 巻 1 号 p. 24-28
発行日: 2015/01/10
公開日: 2015/01/22
ジャーナル フリーA study on atmospheric corrosion monitoring by electrochemical impedance method has been reviewed. A new type of corrosion sensor, which consists of pin and ring electrodes embedded concentrically in epoxy resin, was successfully applied for the corrosion monitoring. The sensor response showed that the corrosion rate of steel in the sunny day was higher at night than in daytime. The corrosion loss calculated from the monitoring data agreed well with that estimated from the surface roughness by a laser microscope. In addition, the monitoring system has been applied to estimate corrosion resistance at each part of steel model structure exposed to outdoor environment. Low solution resistance and high corrosion rate were measured at all parts in rainfall time. From the corrosion monitoring for about a year, the corrosion loss was larger at horizontal part than at perpendicular part, and the under side of flange gave the largest corrosion loss. 抄録全体を表示PDF形式でダウンロード (832K)
抄録全体を表示PDF形式でダウンロード (832K) -
貝沼 重信原稿種別: 研究紹介
2015 年 36 巻 1 号 p. 29-34
発行日: 2015/01/10
公開日: 2015/01/22
ジャーナル フリーMany severe corrosion damages have been observed in steel structures constructed in a period of high economic growth. To ensure the safety of such structures, it is important to devise a method how corrosion develops in the service. In our research, semi-variogram analyses were carried out on time-dependent corrosion surfaces of unpainted steel plates exposed to atmospheric environment in order to quantitatively clarify the spatial autocorrelation structures and their time-dependence. In addition, by using the spatial statistics and an ordinary kriging technique, numerical simulation for the time-dependent corrosion surfaces in atmospheric environments was proposed and the validity was confirmed by comparing with corrosion surfaces obtained in various exposure fields. 抄録全体を表示PDF形式でダウンロード (1780K)
抄録全体を表示PDF形式でダウンロード (1780K) -
押川 渡, 中野 敦原稿種別: 研究紹介
2015 年 36 巻 1 号 p. 35-40
発行日: 2015/01/10
公開日: 2015/01/22
ジャーナル フリーAtmospheric corrosion progresses with wetting and drying steps under a very thin film of water. The relative humidity and amount of attached sea salt influence the formation of a thin water film in particular. Thermodynamic calculations of the water film's composition and thickness showed that the thickness of the water film increases rapidly at a relative humidity of 76% RH. The amount of attached sea salt is 1 g/m2 and the water film thickness is approximately 30 μm. In Okinawa, the corrosion environment deteriorates more severely due the increase in sea salt caused by typhoons as well as the high temperatures and humidity. In areas where the amount of rainfall is miniscule, the amount of sea salt tends to be cumulatively enhanced; however, in environments that receive a large amount of rain, this tendency is lessened due to cleaning. An ACM sensor comprised of a Fe-Ag pair is effective for the evaluation of corrosion environments.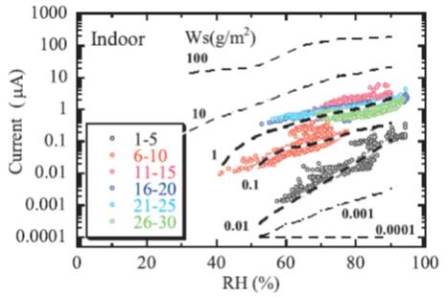 抄録全体を表示PDF形式でダウンロード (1408K)
抄録全体を表示PDF形式でダウンロード (1408K) -
坂入 正敏, 蔭山 明宏, 高木 翔悟原稿種別: 研究紹介
2015 年 36 巻 1 号 p. 41-46
発行日: 2015/01/10
公開日: 2015/01/22
ジャーナル フリーA focusing pulse YAG laser beam irradiation (laser machining) technique and micro-electrochemical cell technique were applied to investigate the corrosion behavior of coated steels. These techniques make it possible to investigated the corrosion behavior with control of areas and shapes. In this paper, the application of the techniques for atmospheric corrosion behavior of size controlled model scratches formed on coated steels, detection of atmospheric corrosion generated hydrogen and hydrogen permeation efficiency at scratched areas are described. 抄録全体を表示PDF形式でダウンロード (917K)
抄録全体を表示PDF形式でダウンロード (917K)
連載企画
環境問題と表面科学
-
吉川 和身2015 年 36 巻 1 号 p. 47-48
発行日: 2015/01/10
公開日: 2015/01/22
ジャーナル フリー気候変動,生物多様性,資源循環等の分野で地球環境は持続可能性の危機に直面しており,我が国が先進的環境技術を生かし環境課題への対処に主導的な役割を担うことが期待されている。ナノテクノロジー・材料技術は,汚染物質の測定分析や分離・分解,有用資源の分離回収,省エネルギー,再生可能エネルギー等の分野で環境分野に大きな貢献を果たしており,環境省の環境研究総合推進費においてもナノテクノロジー・材料技術に係る研究課題が採択・実施されている。中央環境審議会は2014年7月に環境政策の中長期的な基本戦略を提言しており,環境技術に関しては,① 持続可能なエネルギー・資源の生産・消費を支える技術の開発・普及,② 地域振興や豊かで健康的な社会システム・ライフスタイルを支える技術の開発・普及,③ 我が国が培ってきた先進的環境技術の国際展開が謳われている。こうした提言を踏まえた政策展開に当たり環境研究・技術開発の進展に期待するところは大きく,環境技術としてのナノテクノロジー・材料技術の発展に向け産学官の英知が集まることを期待したい。 抄録全体を表示PDF形式でダウンロード (417K)
抄録全体を表示PDF形式でダウンロード (417K)
談話室
海外研究体験記
-
南部 英2015 年 36 巻 1 号 p. 49-50
発行日: 2015/01/10
公開日: 2015/01/22
ジャーナル フリーPDF形式でダウンロード (269K)
表面科学技術者資格認定試験例題
-
2015 年 36 巻 1 号 p. 51
発行日: 2015/01/10
公開日: 2015/01/22
ジャーナル フリーPDF形式でダウンロード (1354K)
先端追跡
-
2014 年 36 巻 1 号 p. 52
発行日: 2014/12/10
公開日: 2015/01/22
ジャーナル フリーPDF形式でダウンロード (141K)
- |<
- <
- 1
- >
- >|