2014 Volume 122 Issue 3 Pages 131-136
2014 Volume 122 Issue 3 Pages 131-136
Japanese people are widely believed to be the descendants of the Jomon and Yayoi people. The dual-structure model, which attempts to explain the formation of the Japanese population, hypothesizes that the indigenous Jomon people migrated from the northern island of Hokkaido and the Ryukyu Islands to the other islands of Japan, where they resided before the Yayoi started to arrive in Kyushu (the westernmost main island of the Japanese archipelago) from the Korean peninsula. Regarding Y chromosome DNA polymorphisms, it is assumed that Jomon males frequently belong to haplogroups C or D, while Yayoi males frequently belong to haplogroup O. These findings suggest that the frequencies of haplogroup C, D, and O might differ between Hokkaido and northern Kyushu males and exhibit geographical gradients in Japan. However, the data of Y chromosome haplogroup frequencies in modern Japanese males is still limited. Here, we investigated whether the frequency of Y chromosome haplogroups differs between males from different regions of Japan. We recruited 2390 males from nine populations in seven cities in mainland Japan and typed their Y chromosome haplogroups. We did not detect any marked variability in the frequencies of these haplogroups among Japanese males, except for a difference between Nagasaki and Kawasaki students. In conclusion, modern Japanese males appear to be genetically homogenized in mainland Japan because of genetic drift and recent frequent gene flow.
According to historical and archeological evidence, the Japanese population is believed to be composed of descendants of the Jomon and Yayoi people. The indigenous people, the Jomonese, resided on the islands of Japan at least 12000 years ago, and the Yayoi started to arrive in southwest Japan from the Korean peninsula approximately 2300 years ago (Chard, 1974). The Jomon people made pottery, lived a hunter-gatherer lifestyle, and were isolated for several thousand years before the Yayoi migration began. The Yayoi people practiced agriculture, weaving, metalworking, and rice cultivation, and quickly spread northeastward in Japan (Hanihara, 1991).
As the Y chromosome does not recombine, the Y chromosome is passed unchanged from father to son. Therefore, the Y chromosome is a useful tool for genealogical research and investigating evolutionary history. According to the haplogrouping and nomenclature system of the Y Chromosome Consortium (YCC) (Karafet et al., 2008), Japanese males mainly belong to haplogroups C, D, and O, which can be further subclassified. Hammer and Horai (1995) have suggested that the Y Alu polymorphism (YAP), which is a marker of haplogroup D, migrated to Japan with the Jomon people, because YAP+ is absent in present continental Asian populations. This hypothesis was supported by Tajima et al. (2004) and Hammer et al. (2006), since YAP+ is the dominant type in Ainu and Ryukyuan males, which are presumed to be remnant populations descended from the Jomon people.
Furthermore, it was reported that haplogroups D and C began their expansion throughout Japan ~20000 and 12000 years ago, respectively, while haplogroup O2b1 began its expansion ~4000 years ago (Hammer et al., 2006). Therefore, it is thought that haplogroups D and C belong to the Jomon male lineage, and haplogroup O belongs to the Yayoi male lineage. Several studies using Y chromosome polymorphic markers have been reported; however, most of them were studies of Ainu or Ryukyuan populations to elucidate Japanese migration, and the sample sizes were small. Based on our knowledge on genetic variation in the Y chromosome, having a larger sample size is important in order to understand the migration/movement of Japanese males in historical and modern contexts.
In the present study, we recruited 2390 male volunteers from nine populations in seven Japanese cities and typed their Y chromosome haplogroups in order to evaluate the differences in haplogroup frequency between different regions of modern Japan.
A total of 2390 males from nine populations, who inhabited seven Japanese cities, were enrolled in our study. Some of the subjects in this study had been described in previous reports (Iwamoto et al., 2013a, b). These populations included college students (S) from Nagasaki (n = 300), Tokushima (n = 388), Kanazawa (n = 298), Kawasaki (n = 321), and Sapporo (n = 302), and adult males (A) from Fukuoka (n = 102), Osaka (n = 241), Kanazawa (n = 232), and Sapporo (n = 206). The geographic locations of the above cities are depicted in Figure 1.
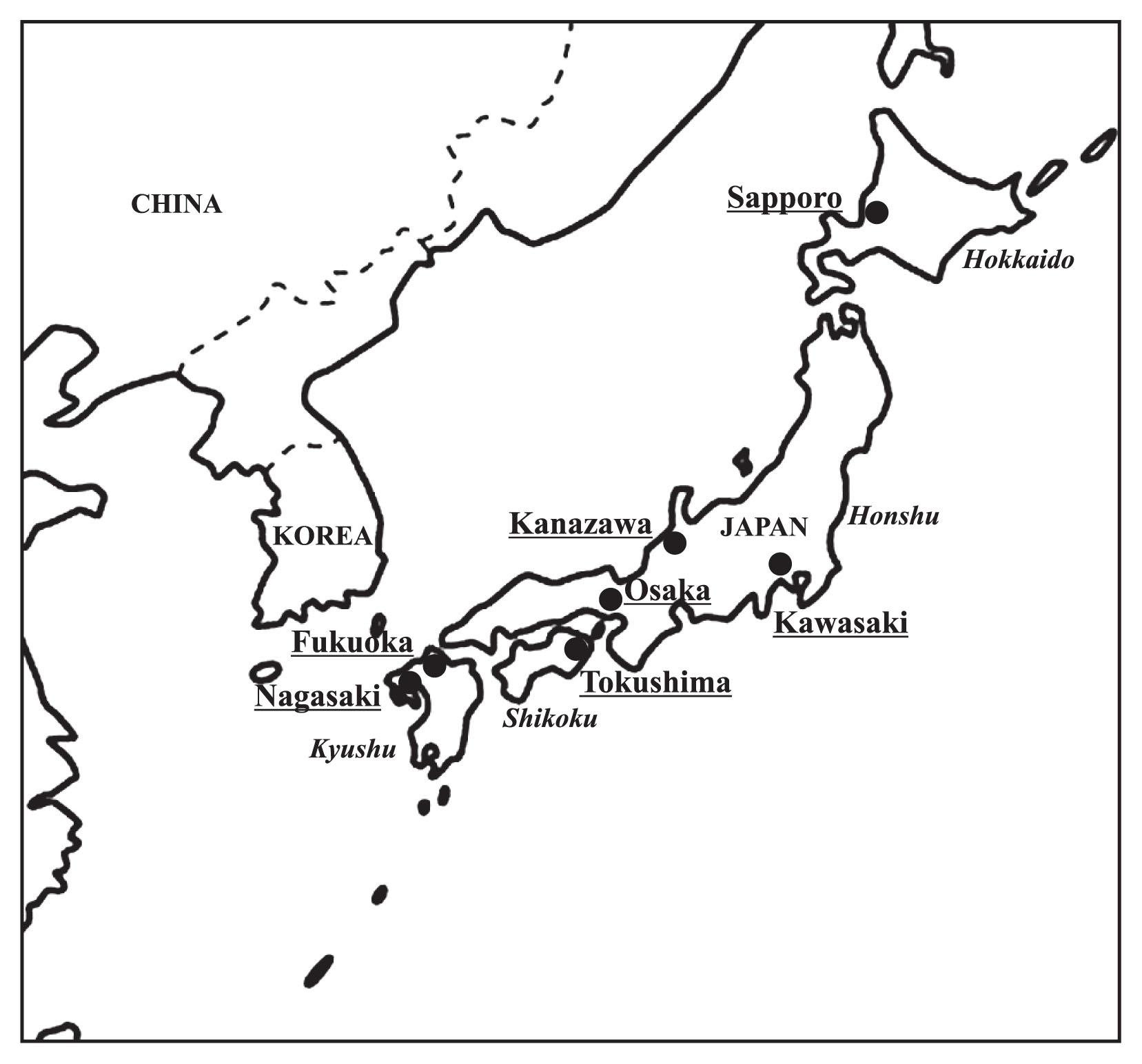
Geographic locations of Japanese males examined in this study.
This study was approved by the ethics committees of the University of Tokushima and St. Marianna Medical University. All of the participants provided written informed consent.
Y chromosome haplogroupingGenomic DNA was extracted from peripheral blood samples with the QIAamp DNA Blood kit (Qiagen; Hilden, Germany). Y chromosome haplogrouping was carried out as previously described (Sato et al., 2013). Briefly, 24 bi-allelic Y-chromosome markers were typed according to polymerase chain reaction (PCR) product size (YAP and MSY2.2) (Hammer and Horai, 1995; Bao et al., 2000); the presence or absence of particular PCR products (12f2b) (Rosser et al., 2000) or PCR-restriction fragment length polymorphisms (M174, LLY22g, M119, SRY465, 47z, M324, and JST002611) (Shinka et al., 1999; Underhill et al., 2001); or using denaturing high-performance liquid chromatography (RPS4Y, M213, M9, M95, M122, and M134) (Underhill et al., 2001), direct sequencing methods (M15, M175, and P31) (Underhill et al., 2001; Su et al., 2000), or allele-specific PCR primers (M55, M8, M217, M214, and JST021354) (Kumagai et al., 2010). The primer sets, PCR amplification conditions, and restriction enzymes used are shown in Supplementary Table S1. Y haplogroups were defined according to the nomenclature of the YCC (Karafet et al., 2008) (Figure 2). In a previous report, it was found that the 12f2.2 and M125 markers belong to the same lineage (de Carvalho et al., 2006). Our data analysis produced the same results (data not shown). Therefore, we considered the 12f2.2 allele to represent the D2a1 lineage.
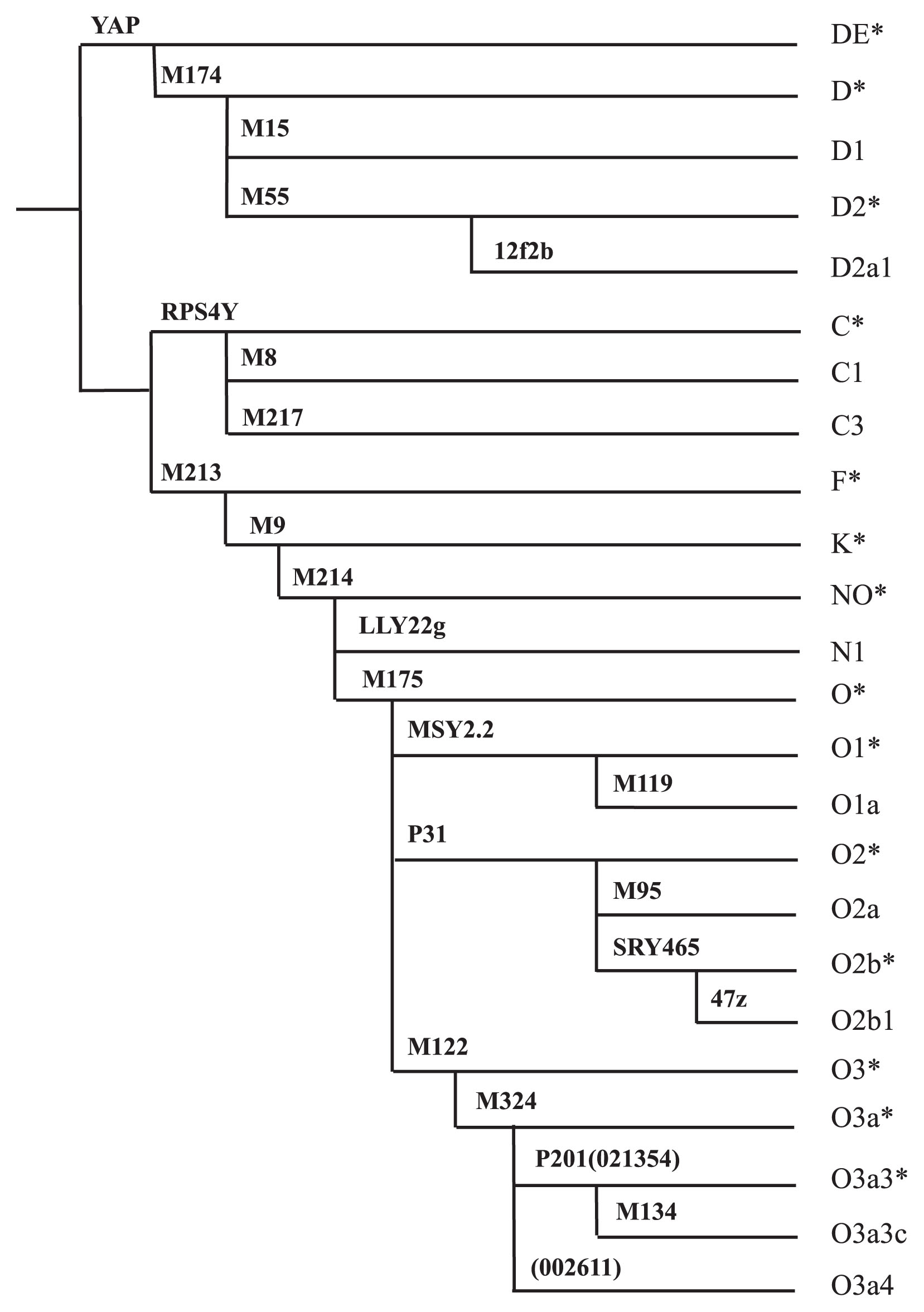
Phylogenetic tree constructed with the 25 Y chromosome binary markers.
To estimate haplogroup diversity among cities, pairwise FST values were calculated using ARLEQUIN version 3.5 (Excoffier and Lischer, 2010).
Y chromosome haplogrouping revealed that Japanese males belong to 16 haplogroups (Table 1). O2b1 was the most dominant haplogroup among Japanese males (22.0%), and D2a1 (17.4%) and D2* (14.7%) also displayed high frequencies. Frequency distributions of the haplogroups among the nine populations are presented in Table 1, but we did not detect any marked variability among the populations. Pairwise FST values were calculated for comparison between each of the nine Japanese populations, but no significant differences were detected, except for in the comparison between the Nagasaki students and the Kawasaki students (Table 2).
| Population (n) | C1 | C3 | D1 | D2* | D2a1 | F* | K* | N1 | O1a | O2a | O2b* | O2b1 | O3* | O3a3* | O3a3c | O3a4 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Nagasaki S (300) | 10 (3.3) | 16 (5.3) | 0 (0.0) | 34 (11.3) | 56 (18.7) | 1 (0.3) | 3 (1.0) | 4 (1.3) | 0 (0.0) | 3 (1.0) | 32 (10.7) | 70 (23.3) | 4 (1.3) | 8 (2.7) | 39 (13.0) | 20 (6.7) |
| Fukuoka A (102) | 6 (5.9) | 8 (7.8) | 0 (0.0) | 18 (17.6) | 16 (15.7) | 1 (1.0) | 3 (2.9) | 1 (1.0) | 2 (2.0) | 0 (0.0) | 9 (8.8) | 27 (26.5) | 2 (2.0) | 2 (2.0) | 6 (5.9) | 1 (1.0) |
| Tokushima S (388) | 22 (5.7) | 33 (5.9) | 0 (0.0) | 49 (12.6) | 70 (18.0) | 0 (0.0) | 6 (1.5) | 4 (1.0) | 7 (1.8) | 8 (2.1) | 40 (10.3) | 90 (23.2) | 3 (0.8) | 14 (3.6) | 36 (9.3) | 16 (4.1) |
| Osaka A (241) | 15 (6.2) | 18 (7.5) | 1 (0.4) | 30 (12.4) | 43 (17.8) | 1 (0.4) | 2 (0.8) | 4 (1.7) | 3 (1.2) | 2 (0.8) | 25 (10.4) | 43 (17.8) | 5 (2.1) | 12 (5.0) | 24 (10.0) | 13 (5.4) |
| Kanazawa S (298) | 10 (3.4) | 19 (6.4) | 0 (0.0) | 44 (14.8) | 53 (17.8) | 0 (0.0) | 2 (0.7) | 7 (2.3) | 0 (0.0) | 11 (3.7) | 34 (11.4) | 63 (21.1) | 3 (1.0) | 13 (4.4) | 23 (7.7) | 16 (5.4) |
| Kanazawa A (232) | 11 (4.7) | 13 (5.6) | 0 (0.0) | 36 (15.5) | 40 (17.2) | 1 (0.4) | 6 (2.6) | 2 (0.9) | 7 (3.0) | 0 (0.0) | 22 (9.5) | 43 (18.5) | 3 (1.3) | 8 (3.4) | 23 (9.9) | 17 (7.3) |
| Kawasaki S (321) | 18 (5.6) | 19 (5.9) | 1 (0.3) | 61 (19.0) | 45 (14.0) | 0 (0.0) | 1 (0.3) | 5 (1.6) | 3 (0.9) | 1 (0.3) | 32 (10.0) | 78 (24.3) | 9 (2.8) | 8 (2.5) | 26 (8.1) | 14 (4.4) |
| Sapporo S (302) | 14 (4.6) | 15 (5.0) | 1 (0.3) | 44 (14.6) | 56 (18.5) | 0 (0.0) | 8 (2.6) | 2 (0.7) | 4 (1.3) | 1 (0.3) | 26 (8.6) | 70 (23.2) | 5 (1.7) | 13 (4.3) | 28 (9.3) | 15 (5.0) |
| Sapporo A (206) | 7 (3.4) | 15 (7.3) | 0 (0.0) | 35 (17.0) | 37 (18.0) | 0 (0.0) | 6 (2.9) | 2 (1.0) | 2 (1.0) | 4 (1.9) | 16 (7.8) | 41 (19.9) | 2 (1.0) | 12 (5.8) | 20 (9.7) | 7 (3.4) |
| All (2390) | 113 (4.7) | 146 (6.1) | 3 (0.1) | 351 (14.7) | 416 (17.4) | 4 (0.2) | 37 (1.5) | 31 (1.3) | 28 (1.2) | 30 (1.3) | 236 (9.9) | 525 (22.0) | 36 (1.5) | 90 (3.8) | 225 (9.4) | 119 (5.0) |
S, college students; A, adult men.
| Nagasaki S | Fukuoka A | Tokushima S | Osaka A | Kanazawa S | Kanazawa A | Kawasaki S | Sapporo S | Sapporo A | |
|---|---|---|---|---|---|---|---|---|---|
| Nagasaki S | — | 0.18919 | 0.72973 | 0.38739 | 0.37838 | 0.39640 | 0.01802* | 0.60360 | 0.25225 |
| Fukuoka A | 0.00316 | — | 0.63964 | 0.21622 | 0.48649 | 0.36937 | 0.92793 | 0.72072 | 0.69369 |
| Tokushima S | −0.00097 | −0.00158 | — | 0.74775 | 0.85586 | 0.64865 | 0.18018 | 0.94595 | 0.52252 |
| Osaka A | −0.00010 | 0.00215 | −0.00107 | — | 0.72072 | 0.93694 | 0.05405 | 0.60360 | 0.66667 |
| Kanazawa S | 0.00011 | −0.00020 | −0.00133 | −0.00126 | — | 0.65766 | 0.27928 | 0.74775 | 0.82883 |
| Kanazawa A | 0.00016 | 0.00079 | −0.00057 | −0.00248 | −0.00086 | — | 0.23423 | 0.82883 | 0.87387 |
| Kawasaki S | 0.00378* | −0.00408 | 0.00123 | 0.00291 | 0.00083 | 0.00103 | — | 0.41441 | 0.39640 |
| Sapporo S | −0.00091 | −0.00234 | −0.00205 | −0.00080 | −0.00109 | −0.00178 | 0.00013 | — | 0.87387 |
| Sapporo A | 0.00131 | −0.00167 | −0.00076 | −0.00155 | −0.00173 | −0.00222 | 0.00054 | −0.00220 | — |
S, college students; A, adult males.
Haplogroup C is subclassified into C1, C2, C3, C4, C5, and C6. Haplogroups C1 and C3 displayed frequencies of 6.1% and 4.9%, respectively, among the Japanese males (Table 1). Next, we produced a line chart showing the frequencies of these haplogroups among the nine populations from west to east (Figure 3). In Kyushu, the C1 and C3 frequencies in the Fukuoka adults showed a higher tendency than that in the Nagasaki students, and in Honshu, the C1 frequency in the Kanazawa students showed a lower tendency than that in other Honshu populations.
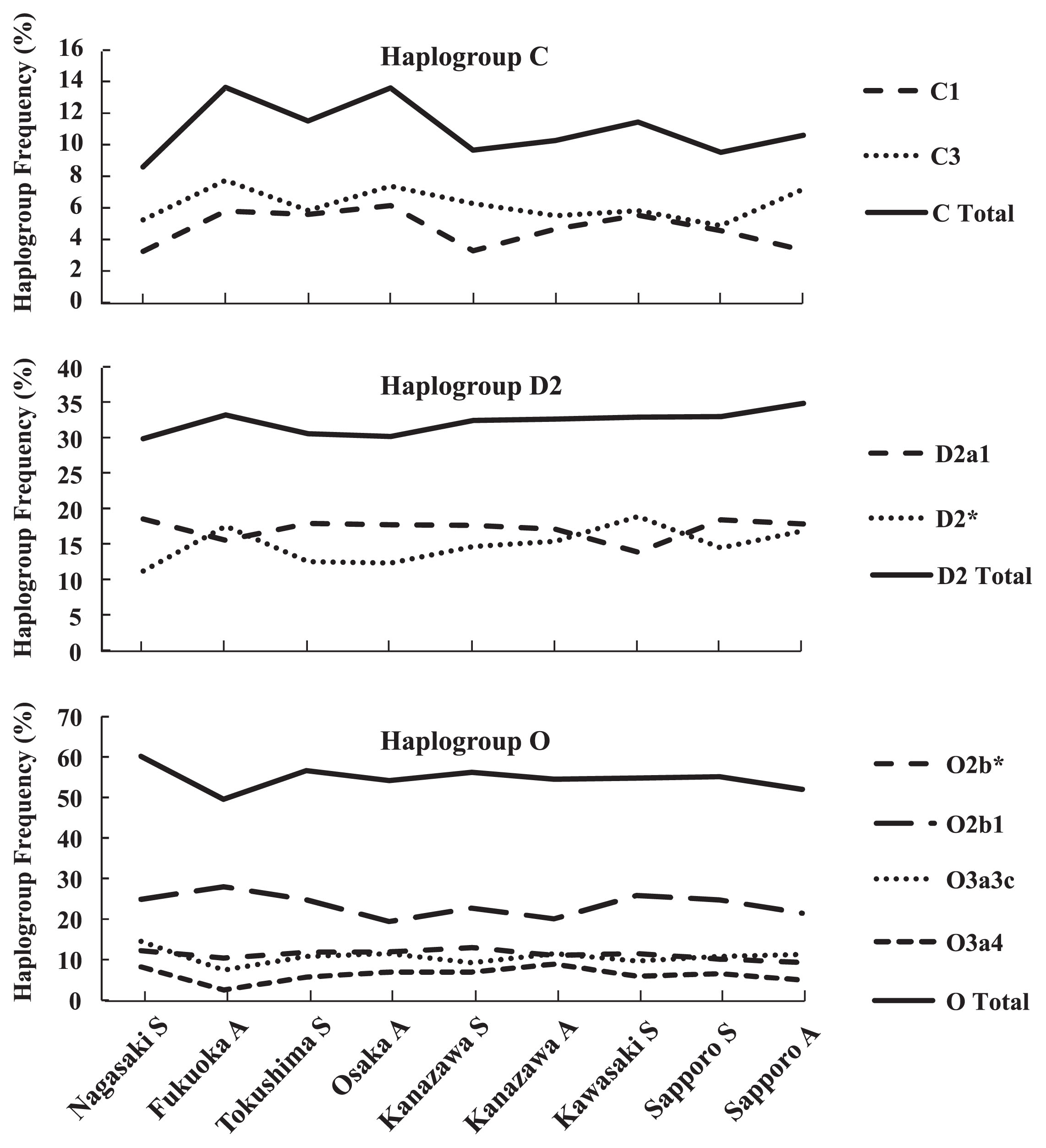
Line chart of haplogroup C, D2, and O frequencies of the nine populations of Japanese males. C Total: sum of the frequencies of C1 and C3. D2 Total: sum of the frequencies of D2a1 and D2*. O Total: sum of the frequencies of O2b1, O2b*, O3a3c, O3a4, O3a3*, O3*, O2a, and O1a. S and A indicate college students and adult males, respectively.
Tajima et al. (2004) and Hammer et al. (2006) reported that haplogroup C3, but not C1, is found in the Ainu population, and haplogroup C1, but not C3, is found in Okinawa. In addition, it has been suggested that human migration might occur predominantly from north Asia to northern Japan, including Hokkaido, because haplogroup C3 displays a high frequency in Nivkhi in northern Sakhalin and the Kamchatka peninsula. However, no marked geographical gradient was detected in the frequencies of haplogroups C3 or C1 in this study.
Haplogroup DAlthough haplogroup D is subclassified into D1, D2, and D3, only D1 and D2 are observed in Japanese males (at frequencies of 0.1% and 32.1%, respectively) (Table 1). Haplogroup D2 includes the D2a1 lineage, which is defined by the presence of the 12f2.2 marker (Figure 2). Previous studies have reported that haplogroup D2 was detected at the highest frequencies among the Ainu population (87.6%) and in Okinawa (55.6%), and the D-M55* lineage (D2*) displayed higher frequencies than the D-M125 lineage (D2a1) in the Ainu population and in Honshu, Kyushu, and Okinawa (Tajima et al., 2004). In the present study, the frequency of haplogroup D2* peaked in the Fukuoka and Kawasaki students. On the other hand, the frequency of haplogroup D2a1 was the least in the Fukuoka and Kawasaki students (Figure 3). The frequency of haplogroup D2 (sum of D2* and D2a1) tended to increase from west to east, except in the Fukuoka students, where it peaked; however, Sapporo did not display a significantly higher D2 frequency than Honshu. These results suggest that haplogroup D2 males are equally spread throughout Japan.
Haplogroup OAccording to the YCC, haplogroup O is subclassified into O1a*, O1a1, O1a2, O2a*, O2a1, O2b*, O2b1, O3a*, O3a1, O3a2, O3a3, O3a4, O3a5, and O3a6. Japanese males belong to O2b1 (22.0%), O2b* (9.9%), O3a3c (9.4%), O3a4 (5.0%), O3a3* (3.8%), O3* (1.5%), O2a (1.3%), and O1a (1.2%) (Table 1). Figure 3 shows a line chart of the frequencies of O2b1, O2b*, O3a3c, O3a4, and haplogroup O (sum of O2b1, O2b*, O3a3c, O3a4, O3a3*, O3*, O2a, and O1a) among the nine populations. The O2b1 frequency in the Fukuoka adults tended to be higher than those in the other populations, whereas their O3a3c and O3a4 frequencies tended to be lower than those of the other populations. Furthermore, their O3a3c and O3a4 frequencies displayed similar distributions throughout Japan.
These results suggest that O2b1 and O3a3c/O3a4 migrated into different areas of northern Kyushu and spread throughout Japan. Among the adult populations, Fukuoka displayed the lowest haplogroup O frequency. Hammer et al. (2006) reported that the highest haplogroup O frequencies were observed in Kyushu; however, our results suggest that this might not be true. Rather, our findings suggest that the distributions of haplogroup O vary among different parts of Kyushu and between O sub-clades. In addition, our results showed that the frequencies of haplogroup O lineages in Sapporo were almost the same as those observed in other Japanese areas. This indicates that haplogroup O males are spread equally throughout Japan (except in Okinawa).
In the present study, we did not detect any marked variability in the frequency distribution of Y chromosome haplogroups in mainland Japan, except for the difference of pairwise FST values between the Nagasaki and Kawasaki students. In addition to the Y chromosome polymorphism, mitochondrial DNA (mtDNA) polymorphisms are widely used as a maternally inherited marker to elucidate human migration patterns. In Japan, the frequency of the mitochondrial haplogroup M7 showed a tendency to decrease, whereas the frequency of the haplogroup N9b showed a tendency to increase from south to north (Shinoda, 2007; Umetsu et al., 2001). These findings suggest that the mobility pattern of males may be different from that of females, because the distribution of Y haplotypes did not show a geographical gradient in contrast to mtDNA. In conclusion, our data for Y haplogroups suggest that modern Japanese males have frequently migrated, which along with genetic drift has resulted in the genetic homogenization of the mainland Japanese.
We thank all of the volunteers who participated in this study. We are grateful to Drs. Miki Yoshiike and Shiari Nozawa (St. Marianna University) for sample collection, and Miss Yukiko Unemi and Keiko Tsuji (Tokushima University) for their excellent technical assistance. This study was supported by the Ministry of Health and Welfare, Japan (grant nos. H10-Seikatsu-017 and H13-Seikatsu-14 to T.I.) and the Japan Society for the Promotion of Science (nos. 1113001 and 1214001 to T.I.).