2020 Volume 85 Issue 2 Pages 157-162
2020 Volume 85 Issue 2 Pages 157-162
Salvia officinalis L. (2n=2x=14) is a perennial plant from Lamiaceae family. It has a long history of medicinal and culinary use. This study was aimed to investigate the polyploidy induction in S. officinalis L. via seed treatment with colchicine concentrations of 0, 0.05, 0.1, 0.25 and 0.5% at 12, 24 and 48 h treatment time as a factorial experiment based on the completely randomized design with three replications. Polyploidy induction firstly detected by morphological, anatomical and physiological characteristics and confirmed by flow cytometry and chromosome observation from leaves. Results showed that the colchicine concentration and treatment duration have significant effects on the survival rate of plants and the percentage of tetraploidy induction. The percentage of survived plants was decreased by increasing the treatment time and colchicine concentration. Meanwhile, the highest percentage of induced tetraploid plants was obtained at 0.25 and 0.5% colchicine with a treatment time of 48 and 24 h, respectively. Tetraploid plants specified by increasing in leaf length, leaf width, plant height, number of leaves, number of nodes, internodes length, stomata size, catalase, peroxidase and polyphenol oxidase activities, total phenolic and flavonoid contents and antioxidant capacity and on the contrary, decreasing in stomata count as compared with diploid ones.
Polyploid plants are bigger than diploids in terms of morphological characteristics including leaf, stem and root size, which could be beneficial in crop yield and production (Dhooghe et al. 2011) especially in forage, vegetables, and medicinal plants. Polyploidy occurs in plants spontaneously but it could be induced in a short time using antimitotic substances such as colchicine which interferes with spindle formation (Eng and Ho 2019). Also, it was reported that the most successful way to induce polyploidy is through seed treatment; as seed treatment with colchicine before germination produces more tetraploid plants rather than germinated ones (Lehrer et al. 2008). Successful induction of polyploidy using colchicine was reported in some plants such as sage (S. polyploids L.) (Kobayashi et al. 2008), parsley (Petroselinum crispum L.) (Nasirvand et al. 2018), Sophora tonkinensis L. (Wei et al. 2018) and cabbage (Brassica oleracea L.) (Albertin et al. 2005).
Sage (S. officinalis) is a perennial plant from Lamiaceae family. In traditional medicine of Asia and Latin America, this plant is used for the healing of diverse types of disorders such as seizures, gout, rheumatism, inflammation, dizziness, paralysis, diarrhea and blood glucose (Garcia et al. 2016). Also, it has strong antioxidant properties due to the production of various secondary metabolites such as phenolic acids and flavonoids, which reduce the risk of cancer and cardiovascular diseases (Oboh et al. 2008, Alimpić et al. 2017).
Given that in most medicinal plants, vegetative organs are the main source for active compounds, the increased chromosomal level can be considered as a valuable and fast way to increase the production of pharmaceuticals (Dhawan and Lavania 1996). Induction of polyploidy by different concentrations of colchicine in other species of Salvia such as S. boweleyana (Duan et al. 2006) and S. hains (Hosseini Grouh et al. 2011) was performed successfully; which in both of them, chromosome duplicated plants had thicker and wider leaves, darker colors, stouter roots, and larger stomata compare to diploid plants. Han et al. (2018) obtained polyploid plants in Bletilla striata using 0.2% colchicine for 36 h. They found that tetraploid plants had thicker leaves, larger stomata, and more chloroplasts than diploid ones which these characteristics could be used as simple and efficient parameters for the screening of tetraploid plants.
Accordingly, this study was performed to induce polyploidy in S. officinalis using seed treatment and comparing morphological and physiological traits of tetraploid and diploid plants with the main aim of creating more genetic variability and selection of superior traits for use in breeding programs.
The experimental material consisted of S. officinalis L. seeds prepared from Pakanbazr Co., Iran. The seeds were firstly rinsed several times in sterile distilled water, then treated by immersion in different concentrations (0, 0.05, 0.1, 0.25 and 0.5% w/v) of colchicine at three treatment times (12, 24 and 48 h) as a factorial experiment based on the completely randomized design with three replications. The number of 450 treated seeds in each treatment condition was cultured in pots.
Morphological and physiological measurementsMorphological characters such as leaf length, leaf width, plant height, number of leaves, number of nodes and internode length were measured at two months cultivated plants. The number of stomata was measured in the epidermis of leaves by the nail varnish method (Smith et al. 1989) using a microscope for three leaves from diploid and tetraploid plants with a sample size of n=20. Several physiological traits, including catalase (CAT), peroxidase (POD), polyphenol oxidase (PPO) activities and total protein content were measured. The leaves from diploid and tetraploid plants were excised and rapidly frozen in liquid nitrogen and kept at −80°C for the extraction of protein and measurement of enzyme activities. So, 0.2 g of leaves were crushed into a fine powder using liquid nitrogen. Then, 1 mL of Tris-HCl buffer (0.05 M, pH=7.5) was added and after vortexing, the solution was centrifuged at 13000 rpm for 20 min at 4°C and the supernatant was transferred to the new tube and centrifuged at 4000 rpm for 15 min at 4°C. The supernatant was used for the measurement of enzyme activities. PPO and POD activities were measured according to Karo and Mishra (1976) and CAT activity according to Chance and Maehly (1955) by the absorbance reading at 420, 425 and 240 nm, respectively. Finally, enzyme activities were expressed in absorbance units (unit mg−1 protein min−1). Protein content was also determined according to the method of Bradford (1976). Bovine albumin was used to plot the standard protein curve, and the concentration of the protein was measured using a NanoDrop spectrophotometer at 595 nm and expressed as mg g−1 fresh weight.
The amount of 1 g of powdered dried leaves from studied plants was used for the measurement of phenolic and flavonoid contents and antioxidant capacity. Extraction was done by ultrasound for 30 min at 25°C in 80% methanol. The 30 µL concentered extract was mixed with 180 µL of distilled water. Later, 1200 µL of 10% folin was added to the solution and kept for 5–10 min. Then, 960 µL of 7.5% sodium carbonate was added to this solution and placed in darkness for 30 min. Finally, using gallic acid as a standard, the color change of the extracts was determined at 760 nm using a spectrophotometer. The data are expressed as mg gallic acid equivalent per mg plant powder (Shin et al. 2007). Total flavonoid content was also determined using a colorimetric assay (Shin et al. 2007). At first 50 µL of the extract was mixed with 150 µL 5% sodium nitrite and kept at room temperature for 5 min. Next, it was mixed with 300 µL of 10% aluminum chloride and incubated for 5–10 min. Then, 1 mL of 1 M sodium hydroxide was added to the solution and brought to 5 mL volume by adding distilled water. The absorption was read at 380 nm. The results are expressed as quercetin equivalents using a standard curve prepared from authentic quercetin. The total flavonoid content amount is reported as mg quercetin equivalent per mg plant powder. Antioxidant capacity was determined by 2,2-diphenyl-1-picrylhydrazyl (DPPH) free radical scavenging activity using the spectrophotometric method of Brand-Williams et al. (1995). 800 µL of DPPH solution was added to 200 µL of extract and stored for 30 min in dark conditions. The absorbance of the samples was then read at 517 nm. The antioxidant capacity of the extracts was calculated as the percentage of DPPH inhibition using the following equation: % DPPHSC=(Acont−Asamp)/Acont×100. Where Acont: Absorption rate of control (80% methanol only) and Asamp: Absorption rate of the sample.
Flow cytometric analysisFlow cytometry of samples prepared from young leaves of the diploid and tetraploid plants was used for the determination of the ploidy level. Nuclei were released from 0.5 cm2 of leaf tissue by chopping with a razor blade for 25 s in 500 µL of nuclei isolation buffer (Galbraith et al. 1983) containing 200 mM Tris (pH=7.5), 4 mM MgCl2·6H2O, and 0.5% Triton X-100. Then, 500 µL of staining solution 1 µg mL−1 of 4,6-diamino-2-phenylindole (DAPI) was added for DNA staining. After 3 min incubation, nuclei were passed through a 30-µm nylon filter to eliminate cell debris. The samples were analyzed using a PA-I flow cytometer (Partec, Munster, Germany) at the Iranian Institute of National Plant Gene-Bank lab at Seed and Plant Improvement Institute, Karaj, Iran. Also, chromosome observations from young leaves of both diploid and some tetraploid plants were performed according to the method described by Nasirvand et al. (2018).
Statistical analysisSurvival rate and tetraploidy induction, which emanated from the influence of colchicine were evaluated by analysis of variance. Morphological and physiological measurements are shown as mean±SE. A comparison of differences between the diploid and tetraploid plants was carried out using t-test. Significance level was defined as p≤0.01 and p≤0.05. Statistical analyses were carried out by the SPSS 16 software.
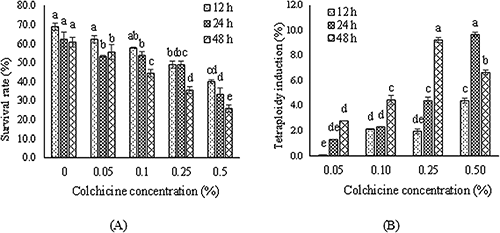
The survival rate and tetraploid induction were significantly affected by treatment time and colchicine concentration. The survived plants decreased by increasing colchicine concentration, and the percentage of induced tetraploid plants were increased in contrast. In a way, irrespective of the control treatment, the highest survival rate in treated plants, were achieved at 0.05 and 0.1% colchicine for 12 h. The lowest amount of survived plants was observed at 0.5% colchicine for 48 h (Fig. 1A). As shown in Fig. 1B, the efficiency of chromosome doubling was low and only some of the treated plants were induced as tetraploids. The highest induction efficiency of the tetraploids was 9.67% and 9.25% achieved at 0.25% colchicine for 48 h and 0.5% colchicine for 24 h, respectively. Considering the very low survival rate at 0.5% colchicine for 48 h, the efficiency of chromosome doubling was very low at this treatment (Fig. 1B).
In terms of polyploidy effects on morphological traits, it was observed that the leaf length and width, plant height, leaf number, node number, internode length, were higher in plants treated with colchicine than diploid ones (Table 1, Fig. 2A–D). In contrast, diploid plants had more stomata than tetraploid ones (Table 1). The size of stomata in tetraploids plants was larger than diploids (Fig. 2E1, E2). In respect of physiological traits as shown in Table 1, a considerable change in CAT, PPO and POD activities was obvious in induced tetraploid plants. However, total protein content was not affected by the ploidy level (Table 1). Also, induced tetraploid plants significantly showed increased total phenol and flavonoid contents and antioxidant capacity (Table 1).
| Trait | Tetraploid (4x) | Diploid (2x) | Significance |
|---|---|---|---|
| Leaf length (cm) | 4.60±0.06 | 4.32±0.05 | ** |
| Leaf width (cm) | 1.3±0.03 | 1.07±0.04 | ** |
| Plant height (cm) | 21.43±0.45 | 19.12±0.38 | * |
| Leaf number | 57.90±2.21 | 44.90±2.32 | ** |
| Node number | 11.65±0.35 | 9.75±0.36 | ** |
| Internode (cm) | 2.06±0.05 | 1.95±0.09 | * |
| Stomata number | 14.15±0.50 | 17.55±0.47 | ** |
| PPO (unit mg−1 protein min−1) | 10.90±0.85 | 7.27±0.99 | * |
| CAT (unit mg−1 protein min−1) | 8.17±0.61 | 4.87 ± .40 | ** |
| POD (unit mg−1 protein min−1) | 6.89±0.59 | 3.95±0.43 | ** |
| Total protein (mg g−1 FW (fresh weight)) | 1.06±0.25 | 1.04±0.47 | ns |
| Phenol TPC (mg galic acid g−1 DW (dry weight)) | 31.14±0.57 | 24.33±0.88 | ** |
| Flavonoid TFC (mg quercetin g−1 DW (dry weight)) | 10.57±0.22 | 7.99±0.57 | ** |
| Antioxidant activity (%) | 61.75±0.12 | 60.21±0.17 | * |
Data are shown as means±SE (n=20). *, **, Significantly differences according to two-sample Student’s t test at p<0.05 and p<0.01, respectively. ns refers to non-significant differences.
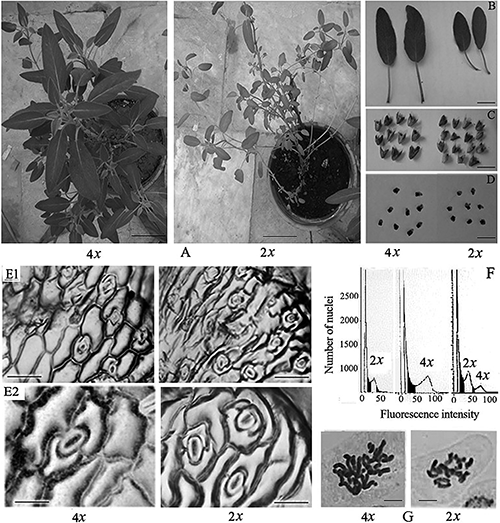
For the preliminary investigation of the ploidy level in established plants, morphological characteristics and stomata size and density were examined. Moreover, for confirmation of the ploidy level of putative tetraploids, the peaks of flow cytometry in the DAPI-stained nuclei histogram were investigated and revealed that the putative ploidy is correlated with the fluorescence intensity of DNA content of the stained nuclei. Peaks obtained by flow cytometry (Fig. 2F) of diploid and tetraploid plant specimens showed that DNA content was duplicated in tetraploid plants rather than diploids. The peak of a diploid control plant (2x) was set at channel 34, whereas, the peak of the tetraploid plants (4x) was appeared at channel 68. Rough chromosome observations confirmed that chromosome mass of induced tetrapliod plants was double of chromosomes of diploid plants (Fig. 2G).
Breeding of medicinal plants is crucial for access to high quality and yield. Artificial induction of polyploidy is one of the most important approaches to reach this purpose. Induced polyploid plants are not the only privilege in some physical and physiological properties, but also showed more resistance to diseases and high content of active compounds (Dhawan and Lavania 1996, Wei et al. 2018).
Colchicine concentration, treatment duration, and plant type are crucial parameters to be manipulated to reach the highest polyploidy rate (Eng and Ho 2019). Our results showed that the lower concentration of colchicine or short duration of treatment could increase the percentage of survived plants but the tetraploidy induction rate decreased significantly. This shows that to obtain effective tetraploid plants proper duration and concentration should be chosen according to plant species tolerance to colchicine. It seems that colchicine impaired enzymes involved in the germination process and reduce the germination rate (Roychowdhury and Tah 2011). Considering our results, at higher concentration of colchicine seedling survival rate was significantly reduced. Accordingly, the highest polyploidy induction efficiency was observed at 0.25 and 0.5% concentration of colchicine at 48 h and 24 h of treatment time, respectively. So, it seems that the optimum treatment to induce autotetraploidy in sage is 0.25% of colchicine with 48 h of treatment time.
The morphological evaluation of plants is a suitable approach for the initial selection of polyploids. Plant’s descriptive features constantly change with increasing chromosome numbers recognized as a gigas effect (Wei et al. 2018). If these changes happen in the commercial part of the plant as in medicinal and vegetable plants, it might become a valuable feature for plant improvement. In our results, tetraploid plants had a longer leaf length, leaf width, plant height, node number, and internode length. Considering that polyploid plants typically have larger and thicker leaves, stem, and roots, they play a key role in increasing medicinal plant yield.
In addition to morphological changes in this experiment, polyploidy significantly influences plant physiological processes like CAT, PPO, and POD antioxidant enzymes activities and phenolic, flavonoid and total antioxidant capacities. Increasing the ploidy level results in increased activity of enzymes and phenolic and flavonoid contents (Table 1). An increase in the ploidy level could influence physiological and biochemical processes and it changes photosynthesis (Warner and Edwards 1993), respiration (Byrne et al. 1981), photosynthetic electron transport (Timko et al. 1980), gene and enzyme activities and isoenzyme expression (Randall et al. 1977). By the way, it should be noted that these changes could be varied depending on the genotype and plant species. Zhang et al. (2010) reported in Dioscorea that antioxidant enzyme activity like superoxide dismutase, POD, CAT, ascorbate peroxidase and glutathione reductase in tetraploid plants is higher than diploids. Talebi et al. (2017) also reported that in colchicine-induced polyploid plants, stomata size and density, chloroplast number of guard cells, morphological traits (leaf length and width, internode length, leaf area, plant height, fresh and dry weight) and physiological and biochemical properties (net photosynthesis, protein content, CAT and POD activities) increased, significantly. However, in the present study total protein content in tetraploid plants did not differ from diploid ones.
In this study, the stomata density of tetraploid plants was significantly lower than diploids. This might due to increased cell size in the tetraploid plants as compared with diploids and consequently decreasing stomata density. Reducing stomata density in tetraploid plants reported on the parsley (Nasirvand et al. 2018), Sorghum bicolor (Sotoudeh Ardabili et al. 2015), and Jasione maritima (Castro et al. 2018). In contrast, tetraploid plants have larger stomata as compared with diploids. Ploidy level of the nucleus often causes structural changes such as stomata density, increasing the size of stomatal cells and number of chloroplasts in a guard cell. Increasing the size of stomatal cells due to polyploidy induction was also reported in some Salvia such as S. miltiorrhiza Bge (Gao et al. 1996), S. boweleyana (Duan et al. 2006), and S. hains (Hosseini Grouh et al. 2011).
The authors thank the University of Mohaghegh Ardabili for their financial supports and breeding and Seed and Plant Improvement Institute, Karaj, Iran for their help in the flow-cytometric analyses.