2018 Volume 58 Issue 6 Pages 1007-1012
2018 Volume 58 Issue 6 Pages 1007-1012
Only a few models for estimating electrical conductivity of molten slag have been developed, which are limited to systems with certain types of components. In this study, a new method for estimating the electrical conductivity of molten slag through neural network calculations is proposed and is compared with previous estimation approaches. The present estimation approach can reproduce the electrical conductivity of molten slag composed of SiO2, CaO, MgO, MnO, Al2O3, FeO, Fe2O3, and Na2O to an uncertainty of 30%. We found that the neural network calculation is applicable to various kinds of molten slag over wider mole fraction and temperature ranges than conventional models.
The physicochemical properties of molten slag are an essential factor in the design of iron and steelmaking process. Hence, considerable efforts have been made to comprehensively understand the physicochemical properties of molten slag. In particular, research on the relationships between the physicochemical properties and structure of molten slag is considered to be important because the physicochemical properties of molten slag are strongly related to its complicated three-dimensional structure. Measurements of the electrical conductivity of slag have played an important role in accumulating current knowledge of the structure of molten slag.1) Thus, many researchers have measured the electrical conductivities of molten slag for a variety of slag systems, compositions, and temperatures.2,3,4,5,6,7,8,9,10,11,12,13,14,15,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33) However, it is difficult to obtain all the necessary data for electrical conductivity by laboratory experiments, owing to the complex reactions of molten slag with surrounded materials at high temperatures.
To the best of our knowledge, four models for estimating the electrical conductivity of molten slag have been proposed by Jiao et al.,34) Lambert et al.,35) and Zhang et al.36,37) Jiao et al.34) empirically derived equation for the electrical conductivity of SiO2–CaO–MgO–MnO and Lambert et al.35) derived that for SiO2–CaO–MgO–FeO at a particular temperature and composition. Zhang et al.36,37) determined a relationship between the electrical conductivity and optical basicity and also found a relationship between the electrical conductivity and viscosity. They offered new interpretations of the electrical conductivity. The results derived from their equations are in good agreement with literature data for quaternary molten slag systems; SiO2–CaO–MgO–Al2O3, SiO2–CaO–MgO–MnO, and those partial sub-systems. These models can be applied only to systems with a limited number of components; however, slag arising from the steel industry contains several kinds of oxides with various mole fraction ratios. Therefore, the development of further models is essential for applications to processing industrial molten slag.
A neural network computation has recently been applied to estimate several kinds of physicochemical properties of molten slag.38,39,40,41) Results calculated with a neural network have reproduced experimental data for a wide variety of molten slags with high accuracy. The viscosity and surface tension of the molten slag have been successfully modeled through neural network computations by Hanao et al.38) and Nakamoto et al.,39) respectively. In addition, sulfide, phosphate, phosphide, nitride, and carbide capacity predictions based on neural network computations have been achieved by Derin et al.40,41) In the present work, we apply neural network computations to the estimation of electrical conductivity for molten slag in a multi component system composed of SiO2, CaO, MgO, MnO, Al2O3, FeO, Fe2O3, and Na2O to reproduce the experimental data with high precision and expand on the variety of molten slag systems.
A detailed explanation of the neural network computation is described elsewhere.38,39,40,41) A layer type neural network, which includes an input layer, a middle layer and an output layer, is used in the present work, as shown in Fig. 1. When the summation of inputs multiplied by the weights exceeds a critical value, the input signal is transmitted through a sigmoid function to the middle layer as a new signal. Then, when the summation of the middle layer values multiplied by the weights exceeds another critical value in the middle layer, it is transmitted as a final output value to the output layer. The back propagation approach is used to minimize the error between the calculated value as an output value and the literature data as teaching data. Through the back propagation approach, the connection weights, W and V, and the critical values, h, are adjusted. The coefficient η determines the shape of the sigmoid curve. This process is called “learning.”
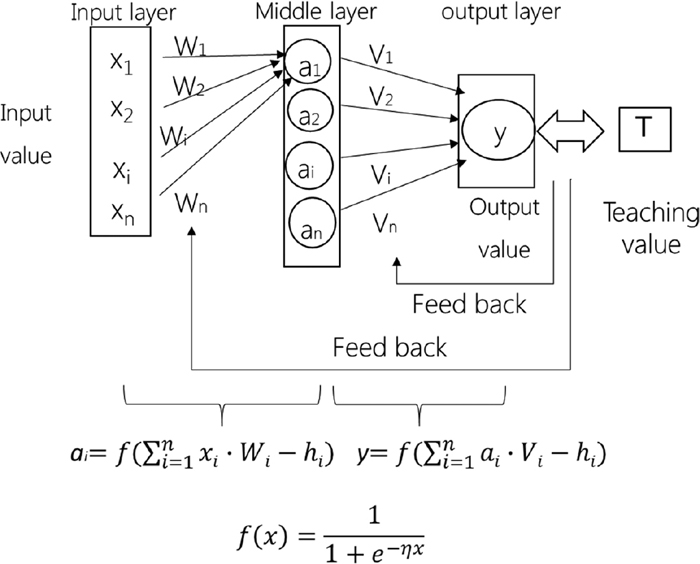
Schematic diagram of layered neural network used in neural network computation.
Original software for neural network computations developed by the Research Center of Computational Mechanics Inc., Osaka University, and Sumitomo Metal Industries Ltd.40,41) was used in this study. The conditions and procedures of the present calculation are explained below. The input values are temperature and the concentrations of the mole fraction of Al2O3, CaO, SiO2, FeO, Fe2O3, MgO, MnO, Na2O and the output value is the electrical conductivity. The total number of literature values used in this study was 835 and we used data that represented both FeO and Fe2O3 concentrations in the treated slag. The data are based on the Slag Atlas,2,3,4,5,6,7,8,9,10,11,12,13,14,15,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33) and are listed in Table 1. We selected 752 data items of electrical conductivity (90% of total number) at random as teaching data to conduct the learning process. Here, we constructed a neural network for estimation of the electrical conductivity through a learning process based on 90% of the total data. The number of units in the middle layer ranged from 1 to 12, the learning number was 5000000, and the initial values which are related to the promotion of learning efficiency given in the product were used. The electrical conductivities calculated by the constructed neural network were compared with the remaining data (10% of total number) as test data to identify the applicability of the constructed network through learning. Finally, we examined the accuracy of the estimation of electrical conductivity by the neural network.
| system | Temperature (K) | Ref. |
|---|---|---|
| SiO2–Al2O3, CaO, FeO, Fe2O3 MgO, MnO, Na2O | 1473–1973 | 2,3,4,5,6,7,8,9,10,11) |
| SiO2–Al2O3–CaO | 1573–1773 | 2), 6), 12), 13) |
| SiO2–Al2O3–FeO | 1553–1653 | 6), 14), 15) |
| SiO2–Al2O3–MgO | 1813–2073 | 8), 16,17,18) |
| SiO2–Al2O3–Na2O | 1273–1673 | 19), 20) |
| SiO2–CaO–FeO | 2023–2043 | 6), 14), 21,22,23) |
| SiO2–CaO–Fe2O3 | 1558–1663 | 23) |
| SiO2–CaO–MnO | 1673 | 5), 9), 10) |
| SiO2–CaO–Na2O | 1273–1673 | 24) |
| SiO2–FeO–Na2O | 1403–1543 | 25) |
| SiO2–MgO–MnO | 1773 | 11) |
| SiO2–MgO–Na2O | 1673–1873 | 26) |
| SiO2–Al2O3–CaO–MgO | 1673–1873 | 8), 27,28,29,30,31) |
| SiO2–Al2O3–CaO–MnO | 1673–1873 | 13), 27), 32) |
| SiO2–CaO–MgO–MnO | 1773 | 11) |
| SiO2–Al2O3–CaO–FeO–MgO | 1673–1873 | 33) |
Figure 2 shows the average error of the difference between the experimental and calculated values with respect to the number of units in the middle layer. The average errors were calculated by Eq. (1):
| (1) |
| (2) |
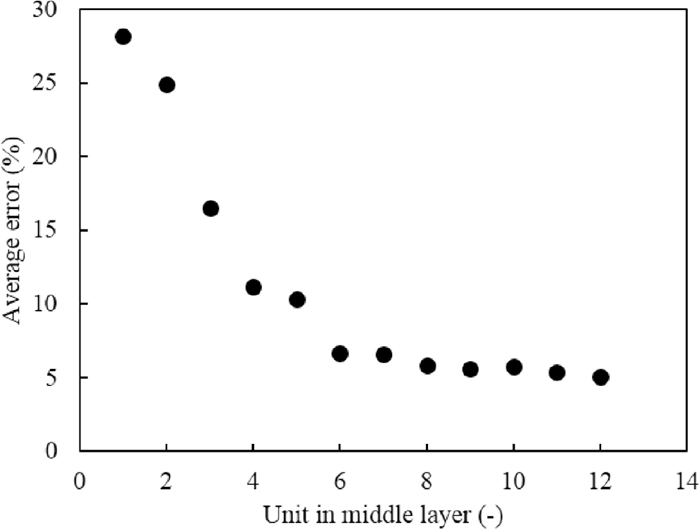
Change in average error with respect to the number of units in the middle layer.
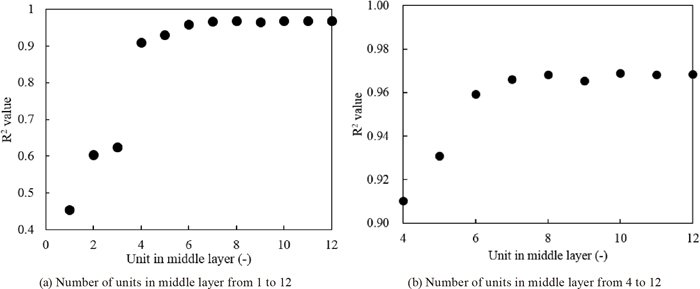
R2 as a function of the units in the middle layer. Number of units in the middle layer ranging.
Figure 4 shows a comparison between the experimental value and the calculated value obtained by the neural network computation with 7 units in the middle layer. Most of the data have less than a 30% error between the experimental values and those calculated by the neural network. As Mills1) reported the error of the experimental values among different research groups ranges up to approximately 30%, thus, the error in our calculation with the neural network is considered to be reasonable. On the basis of the above results, we found that the electrical conductivity of the molten slag could be reproduced with the neural network.
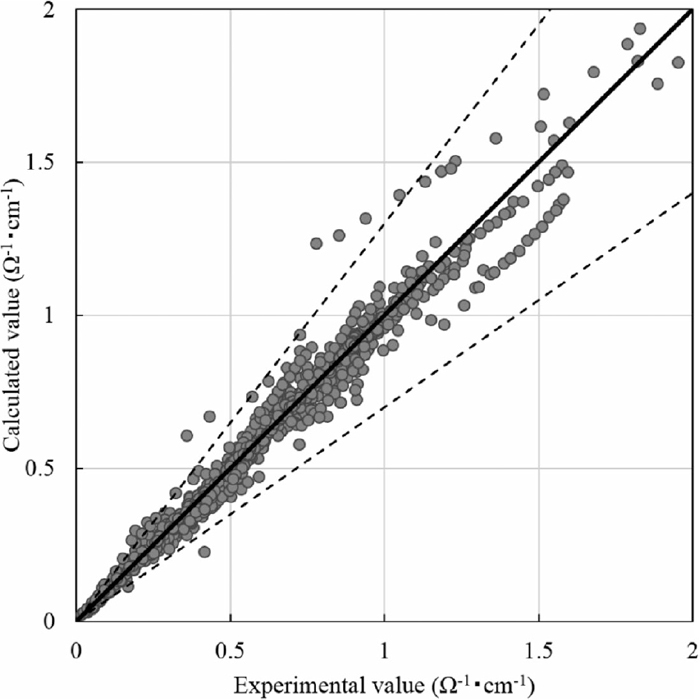
Comparison between experimental values of electrical conductivity and those calculated by neural network computation with 7 units in the middle layer.
Figure 5 shows a comparison between the experimental value and the calculated value obtained with the neural network computation with 7 units in the middle layer in terms of both teaching data and test data. Here, we identify the predictive performance of a neural network constructed under a learning process. In Fig. 5, solid circles express the electrical conductivity data used for learning, i.e., the teaching data, and solid triangles express electrical conductivity data, which were not used for learning, i.e. the test data. The error between the experimental value and the calculated value from the neural network was less than 30% even for the test data. These results mean that experimental data, which were not used for learning, were reproduced by the neural network computation with the unit number assessed by the R2-criterion.

Comparison between experimental values of electrical conductivity and those calculated by neural network computation, with 7 units in the middle layer, in terms of teaching data and test data.
To date four models have been proposed by Jiao et al.,34) Lambert et al.,35) and Zhang et al.36,37) to reproduce the experimental values of electrical conductivity.
Jiao et al.34) proposed an estimation of the electrical conductivity of SiO2–CaO–MgO–MnO slag from the mole fraction of the chemical components and temperature by the following equation.
| (3) |
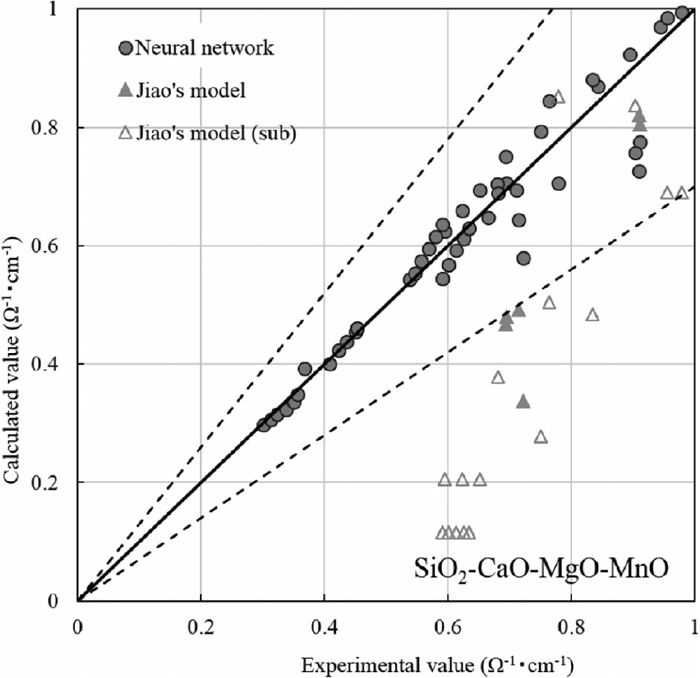
Comparison between experimental values of SiO2–CaO–MgO–MnO slag electrical conductivity and sub-systems (SiO2–CaO, SiO2–MgO, SiO2–MnO, SiO2–CaO–MnO, SiO2–MgO–MnO slag) electrical conductivity and those calculated by neural network and by Jiao’s model.
Lambert et al.35) proposed an estimation of the electrical conductivity of SiO2–CaO–MgO–Fe2O3 slag from the mole fraction of the chemical components at approximately 1673 K as follows.
| (4) |
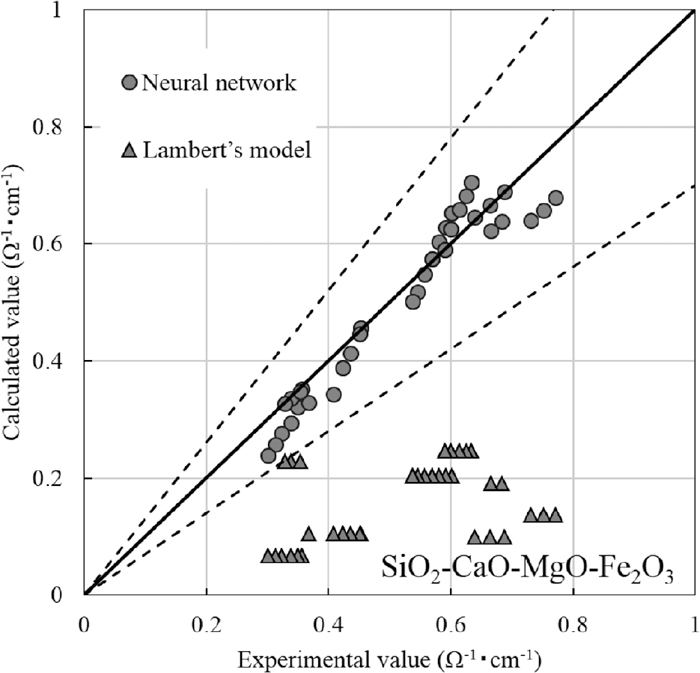
Comparison between experimental values of SiO2–CaO–MgO–Fe2O3 slag electrical conductivity and those calculated by neural network and by Lambert’s model.
Zhang et al.36) proposed an estimation of the electrical conductivity of Al2O3–CaO–SiO2–MgO and Al2O3–CaO–SiO2 slag using the optical basicity. Zhang’s model is more suitable for the electrical conductivity estimation of molten slag than the models of Jiao or Lambert because melt structures are taken into account in Zhang’s model. The equations derived by Zhang et al. are expressed as Eqs. (5) and (6).
| (5) |
| (6) |

Comparison between experimental values of Al2O3–CaO–SiO2–MgO and Al2O3–CaO–SiO2 slag electrical conductivity and those calculated by neural network and by Zhang’s model. (inside application range: the value of optical basicity is ranged from 0.58 to 0.67.)
On the basis of the above results, we concluded that the present calculation with the neural network is applicable to the estimation of 22 types of molten slag systems over a temperature range of 1273–1943 K, as shown in Table 1, with less than 30% error. In SiO2–CaO–MgO–MnO, SiO2–CaO–MgO–Fe2O3, Al2O3–CaO–SiO2–MgO and Al2O3–CaO–SiO2 slag systems, the present neural network calculation reproduced the literature data over a wider range of compositions and temperatures than conventional models.
Finally, the iso-electrical conductivity lines were predicted in the liquidus area of the Al2O3–CaO–SiO2 system and the result is shown in Fig. 9. In this figure, the calculated iso-electrical conductivity lines are drawn by solid lines and compositions where the experimental values are available are described by squares. The predicted iso-electrical conductivity lines well agreed with the reported experimental values, and indicated that the electrical conductivity decreases not only by SiO2 addition but the molar ratio of Al2O3/CaO increases, thus its composition dependence is somewhat complicated. The latter tendency may correspond with the nature pointed by several authors37,45,46) that the electrical conductivity of alkaline or alkaline-earth alminosilicate melts decreases by charge-compensated AlO4 structure units formation, which will decrease the transportation ability of the Ca ions acting as the charge-compensator in the Al2O3–CaO–SiO2 melt.

Iso-electrical conductivity lines in Al2O3–CaO–SiO2 melt at 1773 K predicted by neural network computation.
We applied a neural network computation to the prediction of the electrical conductivity of molten slag in multi component systems. On the basis of the present results, neural network calculations precisely reproduced the experimental data for the electrical conductivity of the molten slag. The neural network was capable of accurately estimating the electrical conductivity of various types of slag over a wider range of compositions and temperatures than conventional models.