2019 Volume 35 Issue 1 Pages 56-60
2019 Volume 35 Issue 1 Pages 56-60
This case report presents a case of acute encephalopathy found in a one-year-old boy, which was caused by secondary carnitine deficiency due to cefteram pivoxil. Prolonged comatose state developed after convulsions associated with fever. He had lost his appetite and taken oral cefteram pivoxil for six days. On admission, he was poorly nourished and showed sluggish light reflexes as well as positive Babinski reflexes. Laboratory studies showed hypoglycemia and hyperammonemia as well as metabolic acidosis. Serum free carnitine was decreased, whereas pivaloyl carnitine level was elevated. Diffusion-weighted MRI on the 8th day of admission exhibited high signal intensity with decreased apparent diffusion coefficient in the cerebral cortex of the bilateral frontal, parietal and occipital lobes, whereas T2-weighted MRI showed no signal intensity. Follow up MRI on the 30th day exhibited cerebral atrophic changes. 99mTc-ethylcysteinate dimer SPECT exhibited decreased cerebral blood flow in the bilateral frontal lobes as well as left parietal lobe on the 10th day. He suffered severe neurological sequelae with spastic quadriplegia. We conclude that careful use of pivalic acid-containing antibiotics is necessary for the prevention of secondary carnitine deficiency especially in poorly nourished pediatric patients. Prophylactic administration of L-carnitine may be taken into consideration to prevent the provocation in such conditions.
Carnitine has a critical role in transferring long-chain fatty acids across the barrier of the inner mitochondrial membrane to gain access to the enzymes of β-oxidation. The deficit of carnitine impairs β-oxidation, leading into hypoglycemia. It also results in the cytosolic accumulation of unoxidized fatty acyl-CoA molecules, which are believed to inhibit the urea cycle, causing hyperammonemia. Several reports have shown the secondary carnitine deficit under the treatment with pivalic acid-containing antibiotics (PCA) in pediatric patients.1,2) Pivalic acid is cleaved from PCA, such as cefteram pivoxil, combined with serum free carnitine, and formed as pivaloyl carnitine. Because pivaloyl carnitine is massively excreted from the kidneys, secondary carnitine deficit develops in patients treated with PCA.3,4) Here we report a pediatric case of acute encephalopathy associated with hyperammonemia and hypoglycemia due to free carnitine deficit under the treatment of oral cefteram pivoxil administrated for six days.
This one-year-old boy was referred to our hospital for prolonged comatose state after convulsions associated with fever. He was a product of 30 weeks’ gestation delivered by caesarian section, weighing 593 g at birth. Apgar scores were 7 at 1 min and 9 at 5 min. He had no family history of inherited metabolic or neurological disorders. His early developmental millstones were normal. At one year and 5 months of age, he developed prolonged comatose state, following three times of generalized convulsions associated with fever and appetite loss after six-day oral administration of cefteram pivoxil under the diagnosis of upper respiratory infection. On admission, he was poorly nourished boy with height of 73.8 cm (−2.9 SD) and weight of 7.2 kg (−3.4 SD). Conscious level was impaired as E4V2M4 on Glasgow coma scale. Neurological examinations showed sluggish light reflexes as well as mild spasticity in the left upper limb and bilaterally positive Babinski reflexes. Laboratory studies showed hypoglycemia (glucose 26 mg/dl) and hyperammonemia (NH3 440 μg/dl) as well as metabolic acidosis (pH 7.201, HCO3 20.0 mmol/L, BE −8.2 mmol/L, anion gap 24 mmol/L). Tandem mass spectrometry tests showed decrease in serum free carnitine of 8.5 nmol/ml (normal value 31.3 + 8.4 nmol/ml) and increase in pivaloyl carnitine detected as C5-acylcarnitine of 3.30 nmol/ml (normal value 0.10 + 0.05 nmol/ml). Organic acid analysis of urine using gas chromatography–mass spectrometry showed no elevation of N-isovaleroylglycine. He was treated with mild therapeutic hypothermia therapy with rectal temperature at 35.5 degree Celsius, combined with intravenous administration of methylprednisolone 30 mg/kg/day for 3 days. Administration of L-carnitine 80 mg/kg/day started and continued from the onset. Diffusion-weighted MRI on the 1st day of admission showed subtle high signal intensity in subcortical white matter of bilateral frontal lobes, although T2-weighted spin echo images as well as fluid attenuated inversion recovery (FLAIR) images were normal (Fig. 1). On the 8th day, diffusion-weighted MRI exhibited high signal intensity with decreased apparent diffusion coefficient in the cerebral cortex of the bilateral frontal, parietal and occipital lobes (Fig. 1). However, T2-weighted spin echo as well as FLAIR images were normal. 99mTc-ethylcysteinate dimer (99mTc-ECD) SPECT exhibited decreased cerebral blood flow in the left frontal, parietal and occipital lobes on the 10th day (Fig. 1). On the 30th day after the onset, MRI showed mild cerebral atrophic changes. He is now 3 years of age and has become confined to the bed for severe neurological sequelae with spastic quadriplegia.
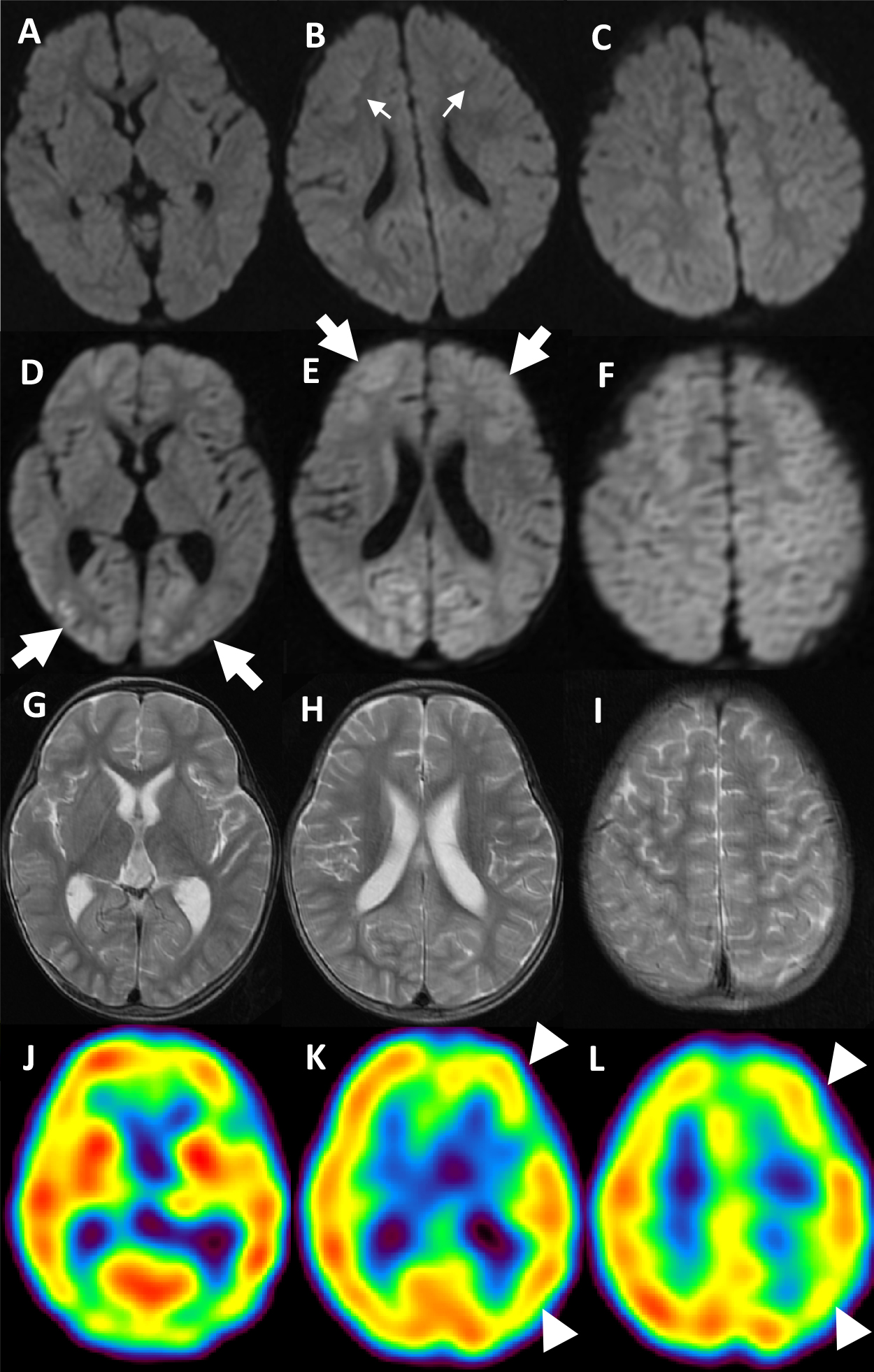
Diffusion-weighted MRI on the 1st day (A, B, C), diffusion-weighted MRI (D, E, F) and T2-weighted spin eho images (G, H, I) on the 8th day, the and 99mTc-ECD SPECT on the 10th day of admission (J, K, L)
On the 1st day, diffusion-weighted images show subtle high signal intensity in subcortical white matter of bilateral frontal lobes (thin arrows in B). On the 8th day, diffusion-weighted images show decreased diffusivity in the cerebral cortex of the bilateral frontal, parietal and occipital lobes (thick arrows in D and E), whereas T2-weighted spin echo images present no signal change in these regions. 99mTc-ECD SPECT exhibits decreased cerebral blood flow in the left frontal, parietal and occipital lobes (arrowheads in K and L).
Acute encephalopathy associated with hyperammonemia and hypoglycemia due to serum free carnitine deficit developed under the treatment with cefteram pivoxil in this patient. Secondary massive loses of serum free carnitine from the kidney as a form of pivaloyl carnitine is suggested as a major mechanism for free carnitine deficit. In addition, it may be caused by low carnitine reserve in his decreased skeletal muscle mass (−3.4 SD as body weight) as well as poor dietary intake before the provocation of acute encephalopathy, since carnitine is stored mainly in the skeletal muscles, and 75% is of dietary origin.5) It can be suggested that cytotoxic edematous changes in the cerebral cortex on diffusion-weighted MRI is regarded as a failure of energy production causing membrane ionic pump insufficiency due to glucose deprivation as well as toxic effects of hyperammonemia such as failure of potassium buffering in astrocytes caused cytotoxic edema.6) Acute encephalopathy due to secondary carnitine deficit in children has been reported to exhibit various distribution of the lesions on MRI. Three literatures have shown diffusion-weighted MRI features of acute encephalopathy due to carnitine deficiency secondary caused by pivalate-conjugated antibiotics. The common distribution of the lesion with reduced diffusivity in these literatures includes cortex of the frontal and parietal lobes. Okumura et al.7) reported high signal intensity in the cortex of unilateral frontal and parietal regions on diffusion-weighted MRI in a three-year-old boy with acute encephalopathy due to carnitine deficiency secondary caused by cefteram pivoxil administrated for 12 months. Subsequent study on fluid-attenuated inversion-recovery MRI on the 20th day after the onset illustrated high signal intensity in the same lesion detected on diffusion-weighted MRI. Kajimoto et al.8) showed high signal intensity in the cortex and subcortical white matter of the bilateral frontal and parietal lobes on diffusion-weighted MRI at the 4th day after the onset found in one-year old girl with acute encephalopathy due to secondary carnitine deficiency caused by the administration of cefditoren pivoxil for 25 days. Nishiyama and his colleagues9) exhibited high signal intensity in the cortex in the right frontal lobe and bilateral parietal lobes as well as in the deep white matter of the right parietal lobe on diffusion-weighted MRI at the 9th day after the onset of acute encephalopathy due to carnitine deficiency after the administration of tebipenem pivoxil for 3 days. However, the signal intensity was not changed on the simultaneous FLAIR study. The distribution of the brain lesion with decreased diffusivity due to hypoglycemia caused by secondary carnitine deficiency may be different from these patterns. In the pediatric series, Ogawa et al.10) presented high signal intensity on diffusion-weighted MRI in the cortex and subcortical white matter in the occipital lobe over both cerebral hemisphere in a 7-month-old girl with severe hypoglycemia due to secondary carnitine and biotin deficit caused by rigorous use of hypoallergenic formulas. The distribution of the decreased diffusivity on MRI in our patient was more widely distributed, i.e. located in the bilateral frontal, parietal and occipital lobes. The reason why our patient showed widely distributed lesions is unclear. However, dramatic high ammonia level (440 μg/dl), although it was seen transiently, may have contribution of the severely and widely distributed lesions. More information is necessary to characterize the radiological features of acute encephalopathy due to secondary carnitine deficit.
In conclusion, careful use of pivalic acid-containing antibiotics is necessary for prevention of secondary carnitine deficit especially in poorly nourished pediatric patients. Prophylactic administration of L-carnitine may be taken into consideration to prevent the provocation in such conditions.
日本小児放射線学会の定める利益相反に関する開示事項はありません.