2015 Volume 43 Issue 4 Pages 253-264
2015 Volume 43 Issue 4 Pages 253-264
Dengue fever (DF), one of the most important emerging arboviral diseases, is transmitted through the bite of container breeding mosquitoes Aedes aegypti and Aedes albopictus. A household entomological survey was conducted in Dhaka from August through October 2000 to inspect water-holding containers in indoor, outdoor, and rooftop locations for Aedes larvae. The objective of this study was to determine mosquito productivity of each container type and to identify some risk factors of households infested with Aedes larvae. Of 9,222 households inspected, 1,306 (14.2%) were positive for Aedes larvae. Of 38,777 wet containers examined, 2,272 (5.8%) were infested with Aedes larvae. Containers used to hold water, such as earthen jars, tanks, and drums were the most common containers for larval breeding. Tires in outdoor and rooftop locations of the households were also important for larval breeding. Although present in abundance, buckets were of less importance. Factors such as independent household, presence of a water storage system in the house, and fully/partly shaded outdoors were found to be significantly associated with household infestation of Aedes larvae. Identification and subsequent elimination of the most productive containers in a given area may potentially reduce mosquito density to below a level at which dengue transmission may be halted.
Dengue fever (DF) is one of the most important emerging diseases and serious public health concerns. It is found in tropical and sub-tropical regions around the world, predominantly in urban and semi-urban areas. The disease is now endemic in more than 100 countries in Africa, the Americas, the Eastern Mediterranean, South-east Asia and the Western Pacific. Recently the number of reported cases has continued to increase as the disease spreads to new areas [1]. In 2014, Japan experienced the first outbreak of dengue fever in almost 70 years. In Bangladesh, the first documented outbreak of dengue occurred in 1964. However, DF has become a serious public health threat in Bangladesh after the first large-scale outbreak in 2000 with 5,551 cases, among which 1,186 (37.6%) cases were dengue hemorrhagic fever. Since 2000, DF cases have been reported every year in all major cities of Bangladesh [2].
Dengue virus is transmitted to humans through the bite of infective female mosquitoes Aedes aegypti and Aedes albopictus. They breed mostly in artificial water-holding containers, but have been reported in natural containers as well [3]. In most areas there are a relatively small number of containers that consistently serve as the primary producers of Aedes larvae, with other containers playing minor roles in mosquito production. “Key containers” are the primary source of adult Aedes mosquitoes [4]. The epidemiological importance of a container class depends on the productivity and the abundance of that specific class of container in the environment. Productivity of a container type depends on a variety of factors, such as size and shape, purpose of use, location (indoors, outdoors, under vegetation, etc.), method of filling (passively/actively rain-filled, manually filled, roof runoff, etc.), lid status (covered/uncovered), material with which the container is made (plastic, metal, cement/clay, etc.), temperature, availability of food, and competition among co-species [5–11]. Moreover, each ecological setting has its own unique set of key containers [12, 13]. In Peru, for example, apparently “not useful” containers, located outdoors and passively filled with rainwater, represent the most important category for adult Ae. aegypti production [11]. In Mexico, tires and bottles were the most important class of container for the Ae. aegypti population [14], whereas in Vietnam water-holding containers for household use, such as large concrete tanks and jars, were the main source of immature Ae. aegypti development [15]. A “key container” survey for improved dengue vector surveillance and vector control was developed (1994–1997) and implemented on a regional basis in 1997 in Vietnam. This program was selected as one of the “best practices for environmental management of dengue” by USAID in 2003 [4].
By focusing on the containers that are consistent producers of larvae and houses that consistently accommodate Aedes larvae, control measures can be tailored for the specific needs of the area and populace. Once the most productive key containers are identified, targeted control of dengue vectors becomes more affordable and feasible. At the same time, targeted vector control can help minimize the use of chemicals that may be costly and have other long-term health and environment impacts. The aim of this study was to identify the containers which served as primary producers of Aedes larvae during the dengue outbreak in the year 2000 in Dhaka. This study also aimed to identify some risk factors for households infested with Aedes larvae.
A household entomological survey was conducted in Dhaka from August through October 2000, the peak epidemic period of DF. The primary purpose of the study was to identify the areas with high density of Aedes mosquitoes in order to prevent the further transmission of dengue [16]. However, no information on the Aedes larval productivity of different types of wet containers from this study was published before. Therefore, a secondary analysis of the dataset collected from this study was performed with the aim to identify the containers that served as primary producers of Aedes larvae during the dengue outbreak.
Dhaka city is situated between 23°52'49'' N to 23°41'12'' N latitude and 90°20'09'' E to 90°27'04'' E longitudes. Dhaka encompasses 347 km2 of area with an estimated population of 15.4 million. This study was conducted within the Dhaka Municipality, formerly called Dhaka City Corporation (DCC). DCC was divided into 90 small administrative units called wards. According to 2001 population census, DCC had 1,107,000 households, and a total population of 5,378,000. Bangladesh has a tropical monsoon-type climate, with a hot and rainy summer and a pronounced dry season in the cooler months. Dhaka meets all the criteria for rapid breeding of Aedes mosquito as the temperature and large rainfall with rapid urbanization and dense population [17].
For the field survey, approximately 100 households (range 100–119) were selected from each of the 90 wards. Although in a strict sense random sampling could not be done with available resources, we tried to minimize the bias in selecting houses. Households were selected considering two points: 1) frequency distribution of different house types, 2) spatial distribution of the households in the geographical area of DCC. Survey teams were equipped with the aerial view map of each ward available at icddr,b. In each ward, approximately 10 equally scattered points were marked in the map. Survey teams were then instructed to select a direction from the center of each selected point in the map by spinning a pen and to visit the required number of households in that direction. Information about distribution of housing types (i.e., independent house, multi-storied house, semi-permanent house, slum, and others) was collected from each of the ward commissioner’s office. A proportional number of households was then selected in each ward according to the distribution of housing types within that ward (e.g., if in a specific ward 10%, 20%, 30%, and 40% of the houses were of each respective housing type, then 10, 20, 30, and 40 households were selected, respectively, representing each type of house). In the survey design method, a household was defined as one separate unit of accommodation (individual home or apartment), and the immediately surrounding premises, irrespective of the number of people residing within the unit. The houses were classified as per their construction.
Independent house: These were brick-built single family homes. These houses were either single floor or duplex.
Multistoried house: These were brick-built apartment houses having two or more floors. More than one family lives in these houses.
Semi-permanent house: The walls of which are made of bricks, or cement concrete but the roofs are made of other materials, such as bamboo, tin, thatch, etc.
Slum house: Slum houses are poorly-built congested tenements, usually with inadequate infrastructure. Each slum area is designated by the local government.
Others: Others include schools, institutions, offices, factories, mosques, markets, etc.
The field survey was conducted by 46 teams comprised of two field research assistants in each team. The team interviewed the household head or other adult resident according to a pre-tested structured questionnaire to collect information on socio-demography, awareness regarding dengue and its vector control, and self-reporting dengue cases. Field research assistants also looked for containers with standing water and for Aedes larvae within the containers. All three locations of each household, i.e., indoor, outdoor, and rooftop, were inspected for potential wet containers. All larvae that could not be identified in the field were collected in labeled specimen bottles and were reared up to the adult stage to identify species. Before the field survey, field research assistants were trained on inspecting wet containers, collecting and identifying larvae, and recording data.
A total of 111 types of wet container were found in indoor, outdoor, and rooftop locations of the households. The containers were then categorized into 11 different groups: flower pots, buckets, water tanks, drums, tires, discarded appliances, plastic bowls, earthen pots, coconut shells, cans and bottles, and others. All unusual and less abundant container types that eventually were found positive were classified as “others,” such as ant guard, air conditioner drip pan, refrigerator drip pan, polythene bag, bath tub, tree hole, bamboo stump, andleaf axil. Although buckets, water tanks, drums, plastic bowls, and earthen jars mostly had a common purpose of use, i.e., water storage, we opted to keep all the varieties instead of a common category in order to obtain a detailed profile of wet containers serving as potential breeding habitats of Aedes larvae. Household infestation with Aedes larvae was defined as a household having at least one container infested with at least one Aedes larvae.
A descriptive analysis was done for the distribution of wet containers and Aedes larvae in three locations. Firstly, the number of different wet containers in the three locations was listed to identify the most abundant container categories in different locations. Secondly, the percentage of each container category was calculated to determine their larval productivity. Thirdly, the contribution of each container category to total positive containers was calculated. Finally, the percentage of Aedes larvae in each container category was calculated. The relative frequency of each container category as an Aedes larval breeding site in different locations was featured by two-dimensional presentation [18]. Slope = 1 was considered as the equality line. If the containers were equally utilized as breeding sites, all points fell on the equality line. If the percentage of positivity of any container category exceeded the percentage of contribution to total wet containers (slope > 1), the point for the container fell above the equality line. This container was then considered to be an essential container for Aedes larval breeding. Conversely, less importance was indicated for the container having a slope of < 1 (i.e., if the point falls below the equality line). Univariate logistic regression analysis was conducted to determine the risk factors associated with household infestation with Aedes larvae. The significance level was set at p < 0.05. However, variables with a p value < 0.1 (Wald chi-square test) in the univariate analysis were selected to include in the multivariate model. The purpose was to identify variables which, by themselves, were not significantly related to household infestation of Aedes larvae but would make an important contribution in the presence of other variables. IBM SPSS version 20.0 software was used for the statistical analysis.
Permission to carry out this study was provided by the of icddr,b Research and Ethical Review Committee. Signed informed consent was obtained from each household that participated in the study.
The results of the entomological survey are summarized in Table 1. Of 9,222 households inspected, 1,306 households (14.2%) were found to be positive for Aedes larvae. Multi-storey houses were the highest in number (39.6%) followed by semi-permanent houses (30.4%) and independent houses (20.5%). Household positivity rate was the highest in independent houses (18.6%) followed by slum houses (14.3%), semi-permanent houses (12.9%), and multi-storey houses (12.8%). Of 38,777 wet containers were examined, 2,272 wet containers (5.78%) were found to be infested with Aedes larvae. The mean number of wet containers per household was 4.20 (SD = 4.99, Median = 3, IQR = 5). The number of wet containers was abundant outdoors (56.5%) followed by indoors (32.2%) and rooftop (11.3%). Among the outdoor containers, 7.8% containers were found infested with Aedes larvae. Among the indoor and rooftop containers, 3.1% and 3.9% of the containers were found to be positive, respectively. The overall house index (HI) was 14.2. Breteau index (BI) was 24.6, and container index (CI) was 5.9. All of the indices showed a high level of risk for dengue transmission [19].
| Number inspected | Number positive | Percentage (%) | |
|---|---|---|---|
| House types | 9222 | 1306 | 14.2 |
| Independent houses | 1890 | 352 | 18.6 |
| Multi-storey houses | 3651 | 466 | 12.8 |
| Semi-permanent houses | 2801 | 364 | 12.9 |
| Slum houses | 771 | 110 | 14.3 |
| Others | 109 | 14 | 12.8 |
| Number of Wet Containers by location | 38 777 | 2272 | 5.8 |
| Indoor | 12 499 | 384 | 3.1 |
| Outdoor | 21 902 | 1714 | 7.8 |
| Rooftop | 4376 | 174 | 3.9 |
Figure 1a shows the number of each container category inspected in the three locations. Among the wet containers, buckets were the most abundant (n = 6580) followed by flower pots (n = 6066), cans and bottles (n = 5034), and earthen jars (n = 5018). Other water-holding containers, such as drums (n = 2945), and tanks (n = 2675) were also high in number. Buckets (29.5%), flower pots (19.4%), and drums (11.8%) were common among indoor wet containers, while earthen jars (15.2%), cans and bottles (14.9%), and miscellaneous wet containers (14.6%) were common among outdoor wet containers. Among rooftop wet containers, flower pots (33.7%), and cans and bottles (18.2%) were the most abundant.
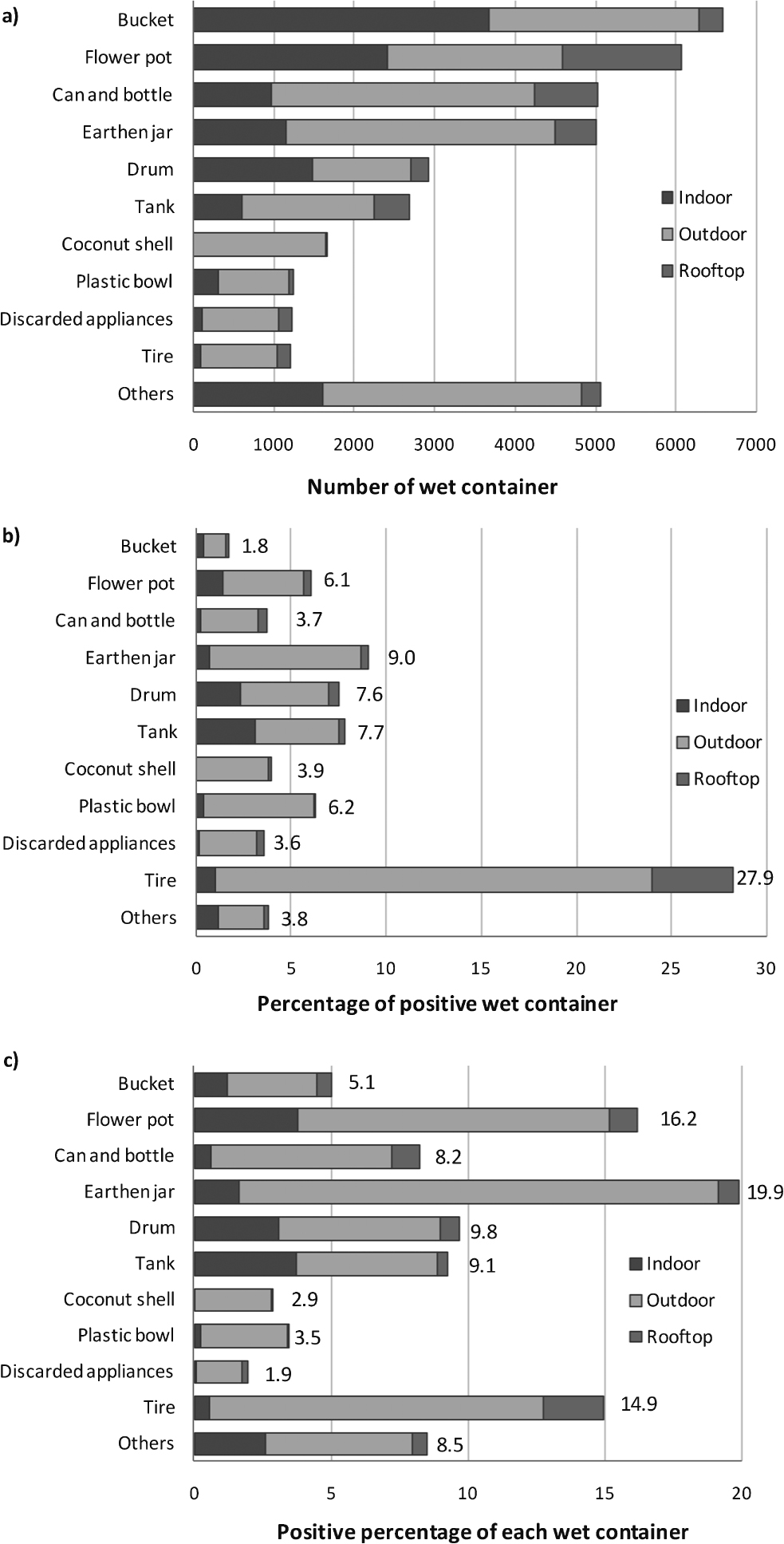
a) Number of wet containers at different locations; b) Percentage of wet containers infested with Aedes larvae; c) Positive percentage of wet container
Figure 1b shows the percentage of each container category infested with Aedes larvae. Among the tires inspected, 27.9% were found to be positive for Aedes larvae. The next three highly positive containers were earthen jars (9.0%), tanks (7.7%), and drums (7.6%). Most of the positive tires (23%), earthen jars (7.9%), tanks (4.4%) and drums (4.6%) were found outdoors. Although buckets were the most abundant, only 1.8% were found to be infested with Aedes larvae.
Figure 1c depicts the percentage contribution of each container category to total positive containers. Of the 2,272 positive containers, the most commons were earthen jars (19.9%), flower pots (16.2%), tires (14.9%), drums (9.8%), tanks (9.1%), and cans and bottles (8.2%).
Two-dimensional presentation for essential containersAmong indoor containers, tanks were found to be the most essential container for Aedes larval breeding (Fig. 2a). Tanks constituted only 4.7% of all wet containers indoors but accounted for 20.8% of all positive containers. Similarly, drums and flower pots constituted 11.8% and 19.4% of all wet containers, respectively, but accounted for 18.2% and 22.4% of all positive containers indoors. Therefore, drums and flower pots may also be considered as essential containers indoors. On the other hand, buckets represented 29.5% of all indoor containers but accounted for only 7.3% of all indoor positive containers. Therefore, buckets fall below the equality line and garner less importance.

Two-dimensional presentation for relative risk of wet containers; a) indoors, b) outdoors, c) rooftop, d) overall
Tires constituted only 4.4% of all outdoor containers but accounted for 16.2% of all outdoor positive containers. Earthen jars represented 15.2% and 23.3% of all outdoor containers and all outdoor positive containers respectively. Similarly, flower pots and drums constituted 9.9% and 5.7% of all wet containers, respectively, but accounted for 15.2% and 7.9% of all positive containers in outdoor. Therefore, tires, earthen jars, flower pots, and drums can be considered as essential containers outdoors (Fig. 2b). Buckets outdoors, as indoors, were found to be less important for Aedes larval breeding. Buckets accounted for only 4.3% of all outdoor positive containers in spite of representing 11.9% of all outdoor containers. Among the outdoor containers, less importance is also indicated for tanks as well as cans and bottles.
Tires and drums were found to be the most important containers in the rooftop location (Fig. 2c). Tires constituted 3.7% and 29.3% of all rooftop containers and all positive containers, respectively. Drums constituted 5.2% of all rooftop containers but accounted for 9.8% of all rooftop positive containers. Buckets represented 6.5% of all rooftop containers but accounted for 7.5% of all rooftop positive containers. Therefore, buckets in the rooftop location were found to be borderline essential containers. Flower pots represented 33.7% of all rooftop containers, but these constituted only 13.2% of all rooftop positive containers. Therefore, flower pots indoors and outdoors exhibited more importance as Aedes larval breeding sites than flower pots in rooftop sites.
Figure 2d shows that overall tires, earthen jars, flower pots, tanks, and drums were found to be essential containers for Aedes larval breeding. Less importance is indicated for buckets, cans and bottles, and discarded appliances.
Aedes larval populationFigure 3 shows the number of both Ae. aegypti and Ae. albopictus by the three locations, i.e., indoors, outdoors, and rooftop (in logarithm scale). A total of 3,027,867 Aedes larvae were collected, among which 1,923,648 (63.5%) were Ae. aegypti. The density of both types of larvae was higher outdoors compared to the other two locations. The ratio of the total number of Ae. aegypti larvae in the three locations was 8 : 39.6 : 1 (indoor : outdoor : rooftop) and Ae. albopictus was 0.9 : 276.7 : 1 (indoor : outdoor : rooftop). About 99% of Ae. albopictus were found outdoors. The number of Ae. aegypti was higher than the number of Ae. albopictus in all three locations (92.7 : 1, 1.4 : 1, and 9.9 :1 indoors, outdoors, and rooftop, respectively).

Number of Aedes larvae by location
Aedes larval productivity of the wet containers in different locations is listed in Table 2. Figure 4 and Figure 5 shows the container productivity for Ae. aegypti and Ae. Albopictus, respectively. Tanks showed the highest productivity for Ae. aegypti larvae both indoors (80.2%) and outdoors (46.1%). In the rooftop location, more than 70% Ae. aegypti larvae were found in buckets. Overall, around 90% of Ae. aegypti were found in water reservoirs, i.e., tanks (50.8%), earthen jars (18.8%), buckets (12.3%), and drums (8.0%). Overall, tires contained only 2.7% of Ae. aegypti. For Ae. albopictus larvae, flower pots indoors (35.6%) and rooftop (72.8%), and earthen jars outdoors (86.2%) showed the highest productivity. Overall, earthen jars alone contained about 86% of Ae. albopictus. The other three water reservoirs, tanks, drums and buckets, did not contribute much to the Ae. albopictus larval productivity (2.9%, 0.5% and 0.2% respectively). Tires contained 3.5% of Ae. albopictus.
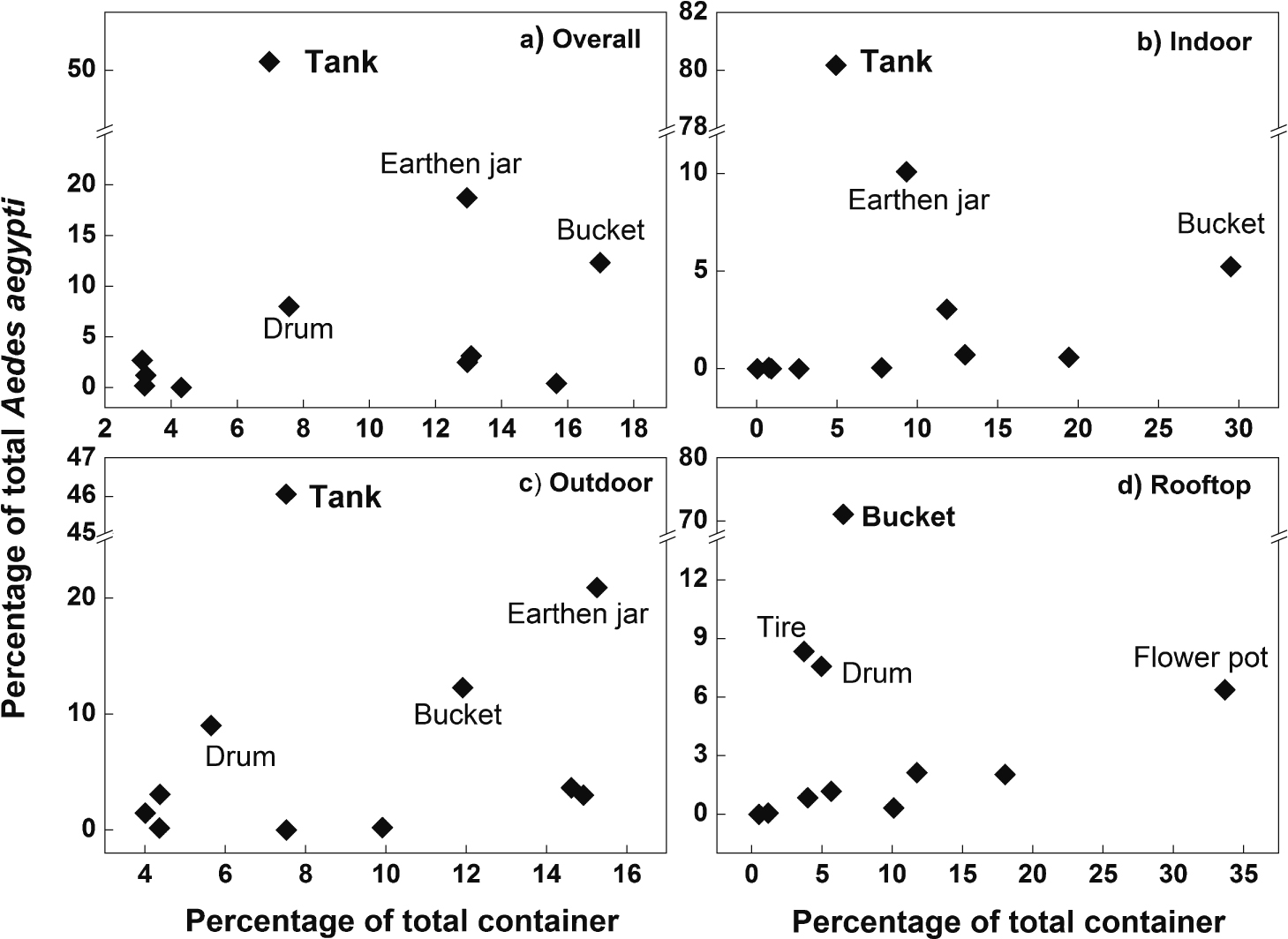
Container productivity for Aedes aegypti larvae
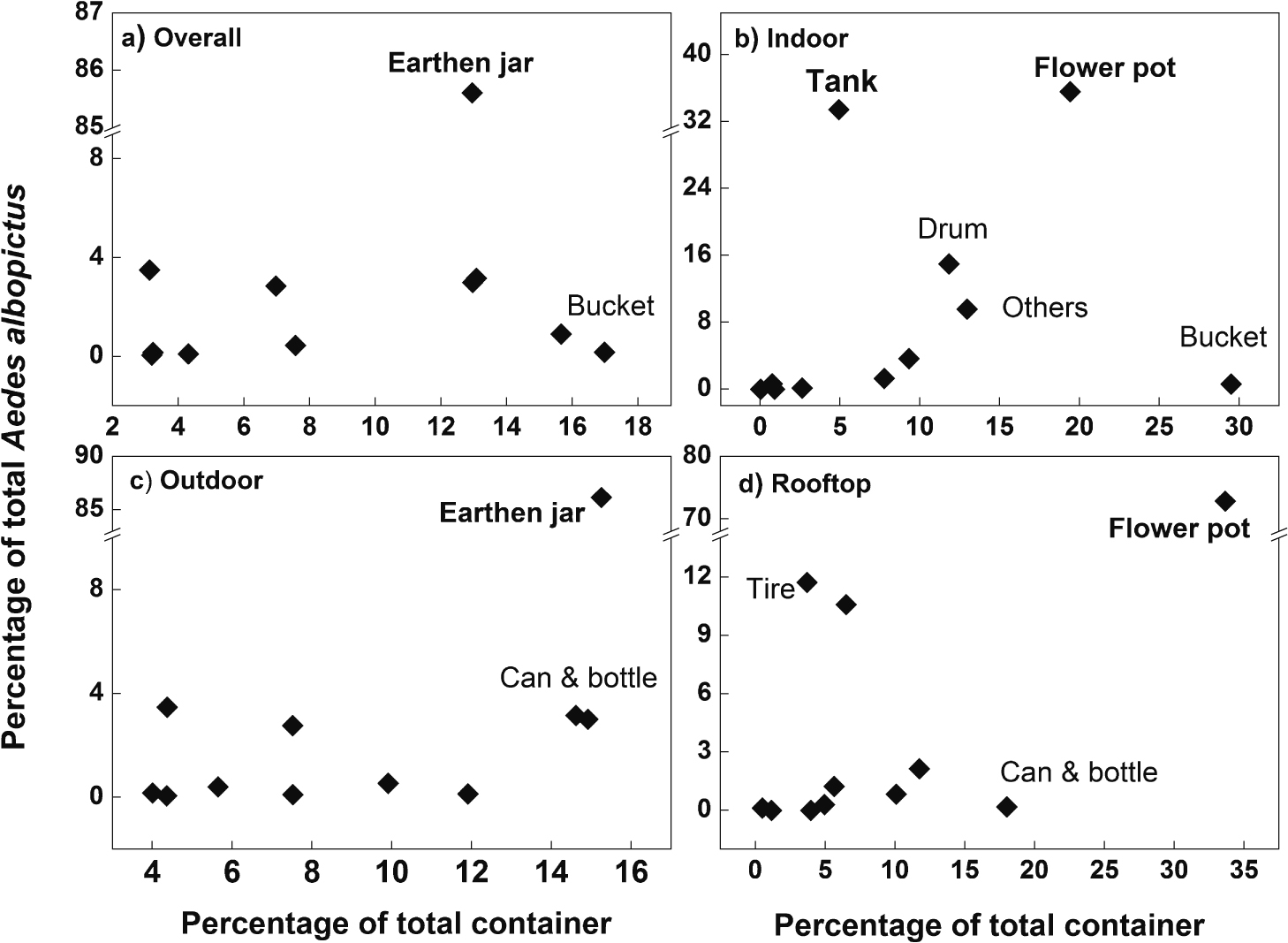
Container productivity for Aedes albopictus larvae
In Bangladesh, it is very common for schools (kindergartens), small shops, or offices to be located within the residential building categorized as a household structure in this study. The houses which were used solely for public purposes, were categorized as “others.” Among the 109 public spaces, 9 were mosques, 37 were schools/institutes, 27 were offices/factories, and the rest were marketplaces. A total of 566 containers were inspected in the public spaces, among which 23 (4.1%) were found to be infested with Aedes larvae. The total number of Aedes larvae found in public spaces was 1,232, among which 696 were Ae. aegypti (56.5%). Although the mean number of wet containers per public space (mean = 5.2, SD = 9.4) was higher than the residence households, the indices for Aedes larval population were lower than the residence households (HI = 12.8, BI = 21.1 and CI = 4.1). Like the residence households, the number of wet containers (64.5%) and number of Aedes larvae (90.7%) were the highest outdoors in public spaces. No Ae. albopictus was found in indoor positive containers (n = 7) and no Ae. aegypti was found in rooftop positive containers (n = 1). The most frequent wet containers were flower pots, tanks, buckets, cans and bottles. However, more than half of the Aedes larvae (51.3%) were found in tires, mostly outdoors (46.8%). Other productive containers were tanks indoors and flower pots outdoors.
| Aedes aegypti | Aedes albopictus | ||||||
|---|---|---|---|---|---|---|---|
| Total larvae | % of total larvae | Cumulative % of total larvae | Total larvae | % of total larvae | Cumulative % of total larvae | ||
| Indoor | |||||||
| Tank | 255161 | 80.2 | 80.2 | Flower pot | 1222 | 35.6 | 35.6 |
| Earthen jar | 32159 | 10.1 | 90.3 | Tank | 1149 | 33.5 | 69.1 |
| Bucket | 16659 | 5.2 | 95.5 | Drum | 515 | 14.9 | 84.0 |
| Drum | 9712 | 3.1 | 98.6 | Earthen jar | 126 | 3.7 | 87.7 |
| Flower pot | 1852 | 0.6 | 99.2 | Bucket | 21 | 0.6 | 88.3 |
| Outdoor | |||||||
| Tank | 721182 | 46.1 | 46.1 | Earthen jar | 945044 | 86.2 | 86.2 |
| Earthen jar | 328079 | 20.9 | 67.0 | Tire | 38151 | 3.5 | 89.7 |
| Bucket | 192673 | 12.3 | 79.3 | Can & bottle | 33059 | 3.0 | 92.7 |
| Drum | 141372 | 9.0 | 88.3 | Tank | 30380 | 2.8 | 95.5 |
| Tire | 48211 | 3.1 | 91.4 | Drum | 4464 | 0.4 | 95.9 |
| Can & bottle | 47239 | 3.0 | 94.4 | Bucket | 1452 | 0.1 | 96.0 |
| Rooftop | |||||||
| Bucket | 28113 | 71.1 | 71.1 | Flower pot | 2888 | 72.8 | 72.8 |
| Tire | 3294 | 8.3 | 79.4 | Tire | 465 | 11.7 | 84.5 |
| Drum | 2996 | 7.6 | 87.0 | Bucket | 420 | 10.6 | 95.1 |
| Flower pot | 2521 | 6.4 | 93.4 | Earthen jar | 85 | 2.1 | 97.2 |
| Earthen jar | 841 | 2.1 | 95.5 | Drum | 12 | 0.3 | 97.5 |
| Overall | |||||||
| Tank | 976473 | 50.8 | 50.8 | Earthen jar | 945255 | 85.6 | 85.6 |
| Earthen jar | 361079 | 18.8 | 69.6 | Tire | 38638 | 3.5 | 89.1 |
| Bucket | 237444 | 12.3 | 81.9 | Can & bottle | 33111 | 2.9 | 92.0 |
| Drum | 154080 | 8.0 | 89.9 | Tank | 31562 | 2.8 | 94.8 |
| Tire | 51696 | 2.7 | 92.6 | Drum | 4991 | 0.4 | 95.2 |
| Can & bottle | 48217 | 2.5 | 95.1 | Bucket | 1893 | 0.2 | 95.4 |
| Flower pot | 7788 | 0.4 | 95.5 | Flower pot | 10019 | 0.9 | 96.3 |
Table 3 shows the results of logistic regression analysis for the factors significantly related to household infestation of Aedes larvae. Multivariate analysis shows that independent household (OR = 1.57; 95% CI = 1.35–1.83, p < 0.001), having any kind of water storage system (i.e., tanks, drums, earthen jars, and buckets) in the household (OR = 1.55; 95% CI = 1.33–1.82, p < 0.001), and having fully/partly shaded outdoor premises (OR = 1.51; 95% CI = 1.34–1.70, p < 0.001) were significantly associated with household infestation of Aedes larvae. “Used mosquito spray /coil /smoke” and “Used insecticide during the last one month” were not found to be significant in the univariate analysis.
Our analyses revealed that water storage containers, such as earthen jars, tanks, and drums were consistently more likely to contain Aedes larvae. Similar results were found in previous studies [20, 21]. Indoor tanks and drums were the most productive; while outdoor earthen jars were the most productive. Rooftop drums were highly productive. Although present in abundance, buckets did not contribute much to larval production. Understanding the cultural traditions of owning and using containers is important to identify the essential containers in different locations. Dhaka city has a scarcity of domestic water supply, and 87.7% of the municipal water supply is mainly derived from groundwater [22]. Most of the city dwellers store supplied pipe water. They either send the pipe water directly to rooftop tanks or store it in underground reservoirs and pump it to the rooftop tanks. Underground reservoirs are categorized here as outdoor tanks. As the municipal water supply is not guaranteed all the time, people store water in drums, earthen jars, buckets, and indoor tanks for use in emergencies. Tanks in outdoor locations and rooftop are normally kept covered and closed; therefore, these reservoirs are protected from mosquitoes. Buckets are relatively smaller in size compared to other water storage containers and are frequently used for washing clothes, cleaning house, and transferring water from one place to another. These practices would reduce the chance of larvae breeding in buckets. Previous studies also reported that weekly cleaning of the water-holding containers was effective in the control of larval production [23, 24]. However, apparently unattractive or frequently cleaned containers, if present in large numbers, may still serve as potential breeding sites for a large portion of the Aedes population. On the other hand, drums, earthen jars, and indoor tanks are bigger in size than buckets and contain a large volume of water. Water in these containers is never emptied and is replenished periodically. A study in Rio de Janeiro found that open-mouthed and large containers are the most suitable for larval production [21]. Moreover, containers outdoors and on rooftops are not always covered, sometimes unintentionally allowing them to collect rainwater and, therefore, making them perennial breeding sites for Aedes mosquitoes [10, 25].
| Number of houses | Number of infested houses (%) | Unadjusted OR† | 95% CI | P-valueσ | Adjusted OR‡ | 95% CI | P-valueσ | |
|---|---|---|---|---|---|---|---|---|
| Type of houses | ||||||||
| Independent houses | 1890 | 352 (18.6) | 1.56 | 1.35–1.82 | < 0.001 | 1.57 | 1.35–1.83 | < 0.001 |
| Semi-permanent houses | 2801 | 364 (13.0) | 1.02 | 0.88–1.18 | 0.78 | 1.12 | 0.96–1.29 | 0.16 |
| Slum houses | 771 | 110 (14.3) | 1.01 | 0.57–1.79 | 0.96 | 0.96 | 0.54–1.70 | 0.89 |
| Multi-storey houses | 3651 | 466 (12.8) | 1 | 1 | ||||
| Had water storage system | ||||||||
| Yes | 2575 | 437 (17.0) | 1.50 | 1.29–1.75 | < 0.001 | 1.55 | 1.33–1.82 | < 0.001 |
| No | 6647 | 869 (13.1) | 1 | 1 | ||||
| Used mosquito spray/coil/smoke | ||||||||
| Yes | 3914 | 553 (14.1) | 1.01 | 0.89–1.13 | 0.94 | — | — | — |
| No | 5281 | 753 (14.3) | 1 | |||||
| Used insecticide during the last 1 month | ||||||||
| Yes | 6747 | 969 (14.4) | 1.06 | 0.93–1.27 | 0.36 | — | — | — |
| No | 2475 | 337 (13.6) | 1 | |||||
| Had fully/partly-shaded outdoor premises | ||||||||
| Yes | 4065 | 695 (17.1) | 1.55 | 1.38–1.75 | < 0.001 | 1.51 | 1.34–1.70 | < 0.001 |
| No | 2759 | 611 (22.1) | 1 | 1 | ||||
† Univariate logistic regression analysis
‡ Multivariate logistic regression analysis adjusted by the variables with p < 0.1 (i.e., type of houses, had water storage system, had fully/partly-shaded outdoor premises)
σ Wald chi-square test
Another important breeding site was tires. Around 28% of tires were found infested with Aedes larvae. They constituted 15% of all positive containers and consistently contain Aedes larvae in all three locations. In public places, more than half of the Aedes larvae were found in tires. Usually tires are left abandoned. The collected rain water in tires is an ideal source of Aedes larvae [14].
Some recent studies use different container parameters while evaluating container productivity. A study in Thailand developed a container-classification method that consists of the shape (S), use (U), and material (M) of the container (SUM-method). Size or volume of the container, exposure to sunlight, presence of abate, cover status, and filling methods of the containers were also considered to determine the container productivity for Aedes larvae and pupae [20, 21]. In our study we do not have detailed information on these container parameters.
Aedes larval populationIn our study, Ae. aegypti was found to be two times higher in number than Ae. albopictus. Moreover, Ae. aegypti was found to be the dominant indoor breeder, while Ae. albopictus showed higher affinity for outdoor containers. Previous studies on the habitation of Aedes mosquitoes showed that Ae. albopictus usually seems to be restricted to wooded areas adjacent to human habitations. Conversely, Ae. aegypti can be found in a variety of urban habitats including the highly urbanized areas without wooded vegetation [26]. Additionally, Ae. aegypti depends highly on human blood and tends to bite and rest indoors, whereas Ae. albopictus feeds on a variety of vertebrates outdoors [27]. Therefore, Ae. aegypti predominates in highly urbanized areas, specially indoor containers. Conversely, Ae. albopictus predominates in rural areas, and in outdoor containers. It seems that Ae. aegypti is better adapted than Ae. albopictus to the environment of crowded tropical cities like Dhaka. Our study found that indoor tanks were the highest productive containers for Ae. aegypti, while outdoor earthen jars were the highest productive containers (86%) for Ae. albopictus. Although a high percentage of tires was found to be positive, they did not contain large numbers of Aedes larvae. One possible reason may be that they contained less water than other water storage containers.
Factors associated with household infestation of Aedes larvaeWe have found that independent households, having water storage system in the household and having fully/partly shaded outdoor premise were significantly associated with household infestation of Aedes larvae. Usually independent households have more space than other types of household and also have both underground and rooftop water reservoir tanks. The latter two factors can easily be explained as they provide a suitable environment for Aedes larval breeding. Using insecticide or mosquito spray/coil was not found to protect against household infestation of Aedes larvae.
A previous study shows a significant variation in spatiotemporal distribution of DF since 2000 at the district level in Bangladesh [28]. However, Dhaka was constantly identified as the highest risk area for DF transmission. Chittagong and Khulna in the south were secondary clusters. Rainwater collection for domestic purposes is not a common practice in Dhaka. On the other hand, 35.5% of households in the southwest coastal region use rainwater because of the arsenic contamination and high salinity of ground water [29]. Containers used for rainwater storage might be the most productive for Aedes larvae in this costal region. Climatic factors, housing structure, types and uses of water containers may vary in different areas of Bangladesh. Thus, the result of this study cannot be generalized for other major cities in Bangladesh.
This study was conducted 15 years ago. In the interim, Dhaka, like other megacities in developing countries, has been experiencing rapid urbanization along with rapid population growth. The demand for water increases day by day along with the growth of population and development of civilization. Currently, 31.4% of households in Dhaka city do not have access to water mains and have to rely on standpipe connection or other sources to fulfill their minimum water demand [22]. Even people with access to piped water usually store water due to the uncertainty of the water supply. In our study, we found that water storage containers served as potential breeding habitats for Aedes larvae. Therefore, proper management of the water storage containers is an important strategy for the prevention of future dengue outbreaks.
Our study results suggest that proper management of water storage containers and tires may reduce a major portion of the Aedes larval population. Community-based educational programs aiming to train householders to use water containers appropriately, such as sealing of containers with lids or nets, cleaning indoor water storage containers regularly, and discarding unused containers, would be a favorable intervention program to reduce the larval breeding sites. Applying some biological agents to the water storage containers may also be an effective control tool for vector density as they are usually cheap and can be maintained by householders with minimal training [30]. Using mesocyclopes in water-holding containers was found to be an effective vector control intervention in Laos [31], Mexico [32] and Colombia [33]. Some studies also found that using larvivorus fish can be an effective biological control tool [24, 34]. However, biological control interventions need to be locally adapted and should take into account cultural practices relating to water storage and the social acceptability of keeping living organisms in containers storing drinking water. For tires, the disposal of unused tires is the best possible intervention. However, applying lime to tires was also found to be effective in reducing vector breeding in discarded tires [4].
Until a vaccine, clinical cure or genetic strategy is available, the control of dengue will continue to depend on suppression of the vector populations and interference in the vector-human interaction [20]. It is, however, a futile exercise to keep on killing mosquitoes in the presence of an almost unlimited number of breeding sites, for the larvae laid at these sites soon grow into adult mosquitoes [35]. For the same reason, generalized community clean-up campaigns to eliminate vector breeding sites have had only a transient and limited effect on disease incidence. Theoretically, the identification and subsequent elimination of most Aedes mosquito producing containers in a given area may potentially reduce mosquito density below a critical threshold, which could result in more efficient and cost-effective control campaigns.
The primary research was funded by the United States Agency for International Development (USAID); Duncan Brothers Ltd.; American Express Foundation; the Department for International Development (DFID) of the United Kingdom; and icddr,b, which is supported by countries and agencies that share concern for the health problems of developing countries. Donors providing unrestricted support included aid agencies of the Governments of Australia, Bangladesh, Belgium, Canada, Japan, the Netherlands, Sweden, Sri Lanka, Switzerland, the United Kingdom, and the United States of America; international organizations providing support included the United Nations Children’s Fund. We are grateful for the cooperation and collaboration of scientists at icddr,b. The authors are grateful to Mr. Touhid Uddin Ahmed, Professor M. Ismail Hossain, and Dr. Manjur A Chowdhury for their technical assistance in the collection of entomological data. We would also like to thank Mr. Zahirul Huq and Mr. Mamunur Rashid for their assistance in constructing the GIS database.