Article ID: 2020-010
Article ID: 2020-010
The development of oligonucleotide therapeutics (ONTs) has advanced recently. Various ONTs (e.g., antisense oligonucleotides, small-interfering RNA, and microRNA) exert their pharmacological effects via hybridization with mRNA sequences, and they can also bind to unintended mRNA sequences owing to sequence homology. For this reason, the safety of ONTs should be evaluated by judging hybridization-dependent on- and off-target toxicity in preclinical studies. As the off-target toxicity is unique to ONTs, it is difficult to assess their safety with the current guidelines established for small molecules and biotechnology-derived pharmaceuticals; thus, several research groups, such as the Oligonucleotide Safety Working Group in the Drug Information Association (DIA), have proposed concepts for the preclinical safety evaluation of ONTs. Although there are currently no specific International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) guidelines for ONTs, the ICH S6 guideline states that “the principles outlined in this guidance may also be applicable to oligonucleotide drugs.” Recently, a preclinical safety guideline for ONTs has been developed by a Japanese working group to address the issues associated with the ICH S6. Here, the preclinical safety assessments of mRNA-targeting ONTs are discussed based on this guidance.
Oligonucleotide therapeutics (ONTs) are a groundbreaking therapy, for which more than 100 clinical trials are currently being conducted worldwide. To date, ten ONTs have been approved for marketing by regulatory agencies, most of which are either antisense oligonucleotides or small interfering RNA (siRNA) that target mRNA (Table 1). In Japan, Spinraza® (nusinersen), which treats a rare disorder called spinal muscular atrophy, was the first antisense ONT approved for marketing in July 2018. Last year 2019, Onpattro® (patisiran) was approved as the first siRNA ONT, indicated for the treatment of polyneuropathy caused by hereditary transthyretin-mediated amyloidosis. Although the knowledge required for ONT development is gradually accumulating, there are still no standardized procedures for the preclinical safety evaluation of ONTs. ONTs are highly target-specific, similar to biotechnology-derived pharmaceuticals (biopharmaceuticals), but have the toxicological characteristics of new chemical entities (NCEs) because most ONTs are composed of nucleotides with various chemical modifications (e.g., 2’-methoxyethyl-modified nucleotides). Therefore, it is not appropriate to automatically apply the conventional guidelines for biopharmaceuticals or small molecule pharmaceuticals to evaluate the safety of ONTs. However, currently, the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) S6 guideline states that “the principles outlined in this guidance may also be applicable to oligonucleotide drugs”.
| Generic name | Trade name | Type | Target | Indication | First approved |
|---|---|---|---|---|---|
| Fomivirsen | Vitravene | Antisense | CVM IE2 mRNA | Cytomegalovirus retinitis in patient with AIDS | US: 1998 |
| EU: 1999 | |||||
| Pegaptanib | Macugen | Aptamer | VEGF protein | Neovascular (wet) age-related macular degeneration | US: 2004 |
| EU: 2006 | |||||
| JP: 2008 | |||||
| Mipomersen sodium | Kynamro | Antisense | ApoB-100 mRNA | Homozygous familial hypercholesterolemia | US: 2013 |
| Eteplirsen | Exondys 51 | Antisense | Dystrophin pre-mRNA | Duchenne muscular dystrophy | US: 2016 |
| Nusinersen | Spinraza | Antisense | SMN2 pre-mRNA | Spinal muscular atrophy | US: 2016 |
| EU: 2017 | |||||
| JP: 2017 | |||||
| Inotersen | Tegsedi | Antisense | TTR mRNA | Polyneuropathy of hereditary transthyretin mediated amyloidosis | US: 2018 |
| EU: 2018 | |||||
| Patisiran | Onpattro | siRNA | TTR mRNA | Polyneuropathy of hereditary transthyretin mediated amyloidosis | US: 2018 |
| EU: 2018 | |||||
| JP: 2019 | |||||
| Volanesorsen | Waylivra | Antisense | Apo-CIII mRNA | Familial chylomicronemia syndrome | EU: 2019 |
| Hepatitis B Vaccine (Recombinant), Adjuvanted | HEPLISAV-B | Vaccine | Hepatitis B surface antigen | Prevention of infection caused by all known subtypes of hepatitis B virus | US: 2019 |
| Golodirsen | Vyondys 53 | Antisense | Dystrophin pre-mRNA | Duchenne muscular dystrophy | US: 2020 |
The preclinical safety assessment for ONTs has been discussed in Europe and the United States [1,2,3,4,5,6,7,8]. In Japan, the Ministry of Health, Labor and Welfare recently issued a preclinical safety guideline for ONTs [9]. This guideline applies to ONTs that induce a biological reaction by hybridization with the target sequence of DNA or RNA in the absence of novel protein synthesis. Specifically, antisense oligonucleotides, siRNAs, and microRNAs are applicable. The principles outlined in this guidance is also applicable to aptamer and decoy. This review will provide an overview of the considerations for the preclinical safety assessment of the mRNA-targeting ONTs described in this guideline.
To evaluate the preclinical safety of ONTs, it is necessary to understand the similarities and differences between ONTs and other pharmaceuticals, such as NCEs and biopharmaceuticals. Generally, biopharmaceuticals are highly specific to their target and exert mechanism-based toxicity (known as on-target toxicity), whereas NCEs sometimes cause unexpected biological functions (known as off-target toxicity), which are difficult to predict based on their properties. As ONTs have the properties of both NCEs and biopharmaceuticals, it is necessary to evaluate their preclinical safety regarding on- and off-target toxicity (Table 2).
| Chemical entities | Oligonucleotides | Biopharmaceuticals | |
|---|---|---|---|
| Modality | Synthetic chemicals | Synthetic chemicals | Monoclonal antibodies, Fusion proteins etc. |
| Synthesis | Chemical | Chemical | Biotechnological |
| Molecular weight | <1,000 Da | 500–15,000 Da | >1,000 Da |
| Target selectivity | Low | Low -High | High |
| Species Specificity | Non-specific | Non-specific / Specific | Specific |
| Typical animal | Rodent and non-rodent | Rodent and non-rodent | Non-human primate |
| Metabolism | Inactive and active metabolites | Inactive and active metabolites | Amino acids |
| Immunogenicity | Rare | Possible | Possible |
| Toxicity | Off-target | Off-target/On-target | On-target |
Various ONTs, such as antisense oligonucleotide, siRNA, aptamer, and decoy, hybridize with specific sequences of mRNA and induce on-target toxicity owing to exaggerated pharmacology. As the on-target toxicity of ONTs is similar to that of biopharmaceuticals, the principles described in the ICH S6 (R1) guideline could be applied to evaluate the on-target toxicity of ONTs [10]. For example, in terms of species selection, it is appropriate to compare target sequences between animal species and choose pharmacologically relevant animals for toxicity studies. When no pharmacologically relevant animal species is available, a surrogate evaluation would be recommended as described in the ICH S6 (R1) guideline. However, although a surrogate is useful for hazard detection, it is not for quantitative risk assessment, as in the case of biopharmaceuticals.
2) Hybridization-dependent off-target toxicityOwing to the high homology between RNA sequences, ONTs can also bind to unintended mRNA sequences and cause hybridization-dependent off-target toxicity. Although hybridization-dependent off-target toxicity studies are crucial for human safety evaluation, those using laboratory animals are of limited value because of genetic differences between humans and animals. To identify hybridization-dependent off-target toxicity, the Pharmaceuticals and Medical Devices Agency generally recommends in silico screening with a representative public database (e.g., RefSeq and GENCODE) and algorithm (e.g., GGGenome, and siDirect), then analyzing the results with an in vitro array (e.g. quantitative-polymerase chain reaction (PCR), microarray, reporter assay, next-gen transcriptome profiling methods such as RNAseq) using human cells (Fig. 1). When candidates are isolated, various data, such as information on the knock-out animal, human genetic disease, target biology, and mechanism of action, should be utilized to evaluate the risk to humans. If the weight of evidence approach suggests any clinical concern, an appropriate risk management plan or optimized oligonucleotide sequence should be considered to avoid the risk.
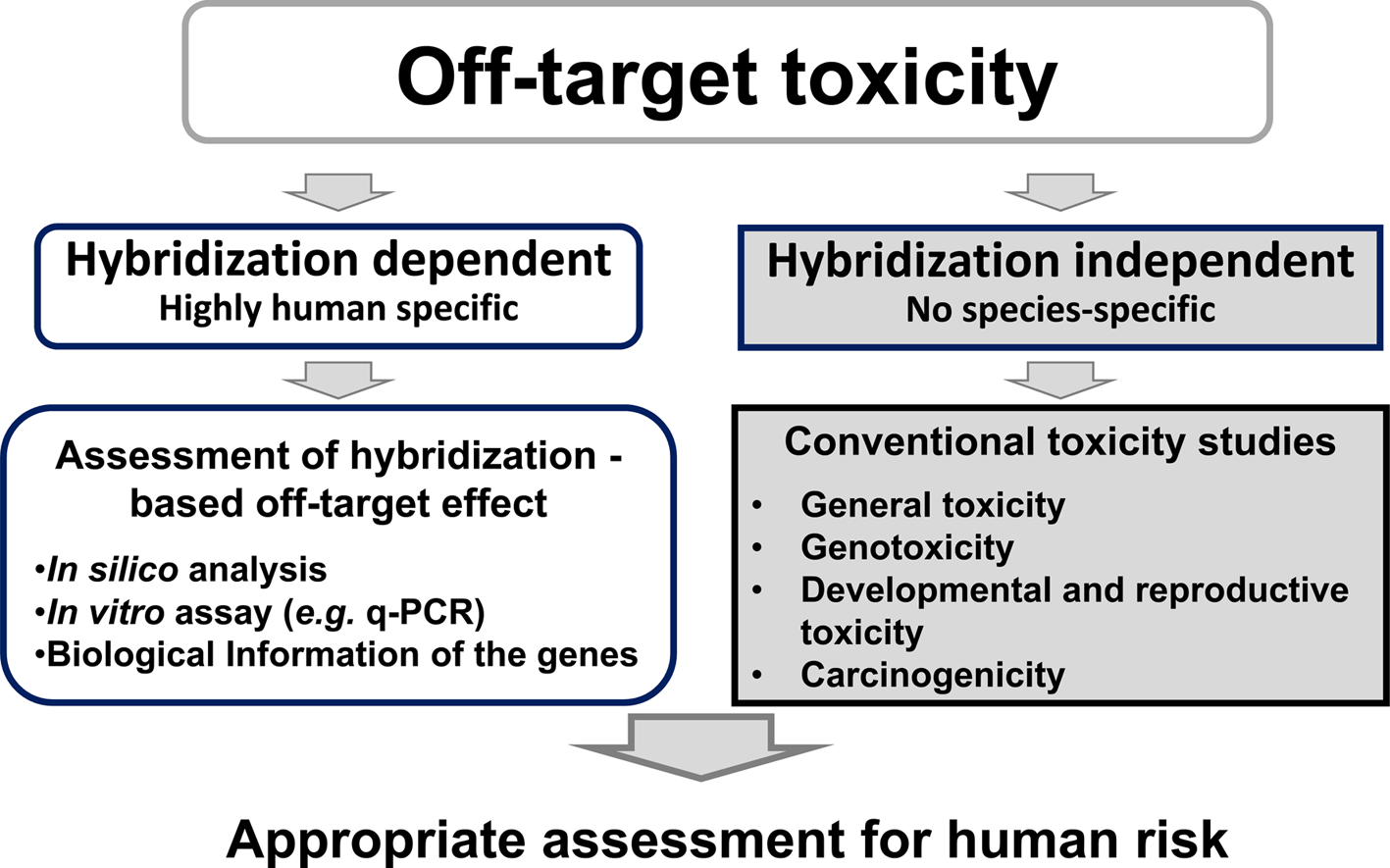
Safety assessment for Off-target toxicity of oligonucleotide therapeutics.
Off-target toxicity caused by any physicochemical property of ONTs, such as class effects which caused by certain chemical structure (e.g., Toll-like receptor-mediated innate immunity, prolonged activated partial thromboplastin time (aPPT), hepatotoxicity, and nephrotoxicity) or toxicity from chemical modifications, can be detected with a conventional toxicity study using laboratory animals (Fig. 1).
To determine the animal species, dosing route, duration of treatment, and frequency of administration for the preclinical safety evaluation, it is necessary to consider the predicted human pharmacokinetic profile of the ONT [11,12,13]. The route and frequency of administration should be as close as possible to those proposed for clinical use. When the pharmacokinetic profiles differ greatly between humans and animals, it may be appropriate to consider other study designs to maintain the level of exposure appropriate for the assessment of toxicity (e.g., frequent dosing).
1) High dose selectionTo detect the off-target toxicity of chemically modified ONTs, it is necessary to follow the ICH M3 guideline for NCEs [14]. In a repeated-dose toxicity study, this guideline recommends a high dose that provides the maximum tolerated dose, limiting dose (1,000 mg/kg/day), exposure saturation, and maximum feasible dose or a dose that is 50-fold greater than the clinical exposure (see the ICH M3 guideline).
2) Species selectionIn principle, two animal species should be used for the general toxicity testing of ONTs, as in the case of NCEs and biopharmaceuticals. This strategy is intended to enhance the prediction of human safety because chemically modified ONTs often show off-target toxicity and their sensitivity and specificity are different among animal species. To evaluate on-target toxicity, it is necessary to use the species in which the pharmacological effect of the clinical candidate can be expressed. When at least one of the two species used for the toxicity study is pharmacologically relevant for the clinical candidate, on-target toxicity can be assessed. However, if no pharmacologically relevant species is available for the clinical candidate, a safety assessment with surrogates should be considered.
3) Use of surrogatesONTs are often species-specific because of their high target-specificity and preclinical safety assessments using surrogates may be performed if relevant animal species with pharmacological effects cannot be obtained with clinical candidates. However, as the surrogate is never identical to the clinical candidate, it could induce surrogate-specific off-target toxicity. Therefore, toxicological results using surrogates should be carefully interpreted in the context of the overall pharmacology and safety assessment.
Most ONTs contain various impurities, such as product-related substances [e.g., n+1, n-1 mer impurities], small organic impurities, residual solvents, and elemental impurities. As it is difficult to characterize each product-related substance, their safety should be assessed in preclinical studies with ONTs. The safety of other impurities can be evaluated with reference to ICH Q3A, ICH Q3B, and ICH M7 for small organic impurities, ICH Q3C for residual solvents, and ICH Q3D for elemental impurities [15,16,17,18,19].
Based on the principles of the “case-by-case basis assessment” and “welfare of animals”, the Japanese guideline provides points to consider to improve the preclinical safety assessment of ONTs. However, continuous efforts are required to identify the limitations of conventional toxicological methods and advance the proper safety evaluation of ONTs.
The author declares no conflict of interest to this report. The views expressed herein are the result of independent work and do not necessarily represent the views and findings of the Pharmaceutical and Medical Devices Agency.
I am grateful to colleagues of the Japanese working group for ICH S6 and related issues: Drs. Yoko Hirabayashi, Osamu Fueki, Misaki Naota, Kazuto Watanabe, Kiyoshi Kinoshita, Mutsumi Suzuki, Takahiro Nakazawa, Satoshi Obika, Teruyo Arato, Aki Fujisaka, Kosuke Ito and Hiroshi Onodera for valuable advice. I would like to thank Ms. Tamara Black for English language editing. This work was supported by the Japan Agency for Medical Research and Development (AMED, Promotion Project “Pharmaceutical Regulations Harmonization and Evaluation Research Project”).