2013 Volume 53 Issue 10 Pages 1763-1769
2013 Volume 53 Issue 10 Pages 1763-1769
Many reactions such as the reduction of iron oxide, gasification of carbon, and so on occur simultaneously during heating of the iron oxide–carbon composites which attract attention as “microreactors”. Solid carbon and CO gas react with iron oxide via direct and indirect reductions, respectively. The rate of indirect reduction has been studied extensively. On the other hand, there are very few reports on the quantitative analysis on the direct reduction due to analytical difficulties, although it is important to understand the total reduction mechanism in detail. In this study, the contribution of the direct reduction by solid carbon in the composite while heating at a constant rate under inert gas flow was quantitatively evaluated.
The direct reduction from Fe2O3 to Fe3O4 proceeds at a low temperature during heating of the Fe2O3–graphite composite. When the reduction from Fe2O3 to Fe3O4 completes below 1000°C, the contribution ratio of the direct reduction in the total reduction is approximately 45%. However, there is no effect of the particle size of the raw materials on the contribution ratio. Further, the contribution ratio of the direct reduction for the reduction from Fe3O4 to FeO is small.
The world steel industry was responsible for approximately 10% of the anthropogenic greenhouse gas emissions,1) since a large amount of fossil fuels are utilized in the ironmaking process. Carbon is used as the heat source and reduction and carburization agents, resulting in the discharge of greenhouse gases, particularly CO2. Therefore, innovations in ironmaking to decrease CO2 emissions are highly required.
Lowering the thermal reserve zone temperature in the blast furnace by decreasing the distance between the iron oxide and carbonaceous particles may be one effective way to decrease CO2 emissions. Mixed charging of iron ore with coke in the blast furnace,2) utilization of highly reactive coke such as iron coke,3) and use of iron ore–carbon composites4,5,6) have attracted significant attention. The process involving the use of composites, which is known as a rapid ironmaking process, is commercially realized as FASTMET and ITmarkIII.7)
A number of studies have been conducted on the reduction behavior of composites containing iron oxide and carbonaceous materials from the viewpoints of utilization as a blast furnace burden,4,5,6) recycle-use of waste materials,8,9) grinding/milling effect,10,11) utilization of low-grade iron ore resources,12,13) and so on. Lowering the gasification temperature of carbon in the composite is an effective method to increase the rate of indirect reduction shown in Eq. (1), because rate-determining reaction is the gasification of carbon in most cases.
| (1) |
On the other hand, the reduction of iron oxide proceeds by not only indirect reduction but also direct reduction, which is the reaction between solid carbon and iron oxide, as described in Eq. (2).
| (2) |
The progress of this reaction is required for initiating the reduction reaction in the composite using carbon without volatile matter. Promotion of the direct reduction may help in lowering the reduction temperature. There has been no report on the quantitative analysis of the contribution of the direct reduction to the total reduction reaction. Iguchi et al. discussed this contribution on the basis of the weight change in the composite under a vacuum condition.14) However, they did not give a quantitative conclusion.
The objective of this study is to carry out a quantitative evaluation on the contribution of the direct reduction to the total reduction reaction in the composite and to clarify the mechanism of direct reduction.
The reduction reactions, Eqs. (1) and (2), and the gasification reaction, Eq. (3), proceed in the composite during heating.
| (3) |
Here, the concentrations of CO and CO2 gases at each temperature are described as XCO and XCO2, respectively. The amounts of CO and CO2 gases generated by the direct and indirect reduction reactions described in Eqs. (2) and (1), respectively, per unit time are a and b mol, and the amount of CO gas generated by the gasification reaction described in Eq. (3) is 2c mol. The following equations are obtained from the balance between the CO and CO2.
| (4) |
| (5) |
Reagents of Fe2O3, Fe3O4, and graphite and coal (F.C.:55.5%, V.M.:36.1%, Ash:8.4%) with the average particle sizes as shown in Table 1 were used. The oxide and graphite powders were mixed well. The molar ratio of fixed carbon in coal to oxygen in iron oxide, C/O, was set at 0.8. The mixed powder was press-shaped under a pressure of 9.8 × 107 Pa. A composite sample with a diameter of 10 mm and a height of 10 mm was thus prepared. Further, “mixed-ground” sample was prepared to realize good contact between iron oxide and graphite particles by grinding the iron oxide and graphite powders together in a planetary ball mill (700 rpm, 15 min × 4 times, Bessel: 40 mL (ZrO2), seven balls: 15 mmϕ (ZrO2)). The obtained powder was also press-shaped by the above manner. Composite samples with different heights were also prepared for the low-pressure experiment described below.
| Average particle size (μm), [purity] | |
|---|---|
| Fe2O3 | 2.2 [95%], 9.6[99%], 78.4 [99%], 197.0 [99%] |
| Fe3O4 | 4.4 [99%] |
| Graphite | 14.9 [99%], 18.6 [98%], 22.1 [99%], 42.5 [99%] |
| Coal | 25.2 |
The reduction experiment under atmospheric pressure was carried out by the following method. Figure 1 shows a schematic diagram of the experimental system used for the reduction of the composite. The details of this apparatus have been reported previously.12) The composite sample was placed in the holder, as shown in Fig. 1(a). After evacuating the air from the chamber, Ar–5% N2 gas was introduced at a rate of 8.33 × 10–6 Nm3/s. N2 gas was used as a tracer to estimate the amount of gas generated from the composite. The sample was then heated to 1573 K at a rate of 0.33 K/s using an infrared image furnace. The temperature was measured at a height of 1 mm from the composite sample using a Pt/Pt–13% Rh thermocouple. The concentrations of CO and CO2 of the outlet gas were continuously measured using infrared gas analyzers. The concentrations of H2, H2O, and N2 gases were also measured by gas chromatography every 90 s. Reduction degree of iron oxide in the composite was calculated based on the results of gas analyses.
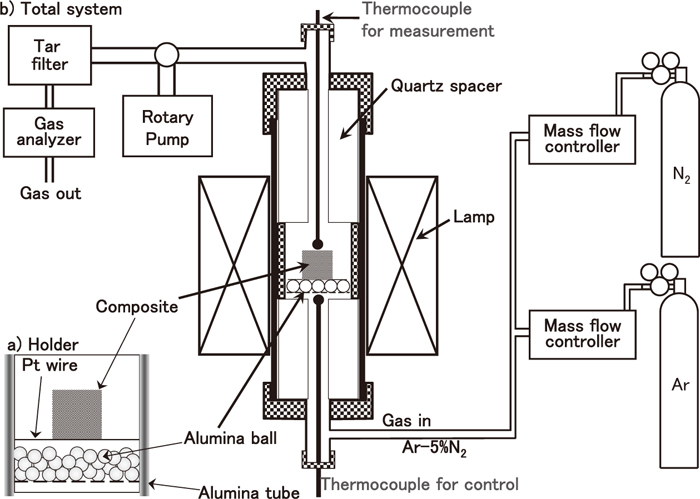
Schematic diagram of experimental apparatus for reduction of composite.11)
The low-pressure experiment was also carried out using same experimental apparatus, while the gas in the chamber was suctioned using a vacuum pump. To check the effect of pressure on the reaction behavior of the composite, two different pressures of 2 and 10 Pa were applied using different pumps. Since it was not possible to analyze the concentration of the generated gas in the case of low-pressure experiment, the reaction behavior was evaluated by weight change before and after reduction.
Figure 2 shows the changes in the rate of CO and CO2 gas generation from the Fe2O3-graphite composite during heating. The average particle sizes of Fe2O3 and graphite are 2.2 and 18.6 μm, respectively. The rate is expressed in moles per gram of Fe. At 700°C, the generation of CO2 gas begins, indicating that the reduction reaction in the composite starts at this temperature. Two peaks attributable to the generation of CO2 gas appear at 820 and 1100°C. The generation rate of CO gas starts to increase rapidly at approximately 1050°C and reaches the peak value. These rates decrease drastically above 1200°C and become 0 at 1250°C.
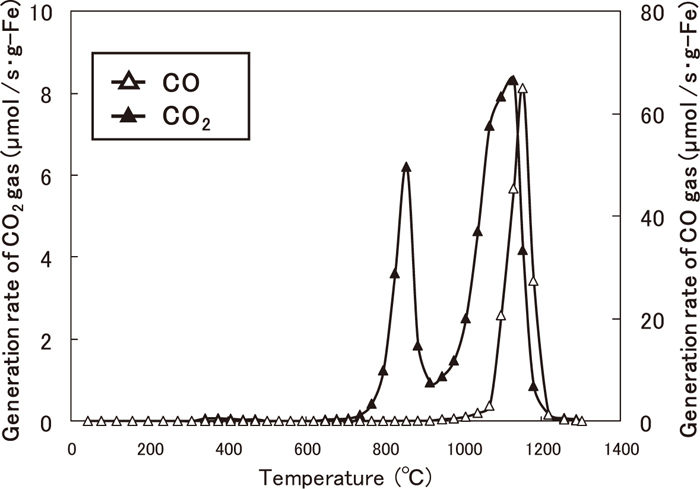
Changes in the generation rate of gases from graphite-Fe2O3 composite with temperature. (Average particle sizes of Fe2O3 and graphite are 2.2 and 18.6 μm, respectively.)
The change in the reduction degree calculated by the results of the gas analysis shown in Fig. 2 with temperature is shown in Fig. 3, together with that for the Fe2O3-coal composite, to discuss the effect of carbonaceous materials in the composite. The reduction degree of the Fe2O3-graphite composite starts to increase at approximately 700°C because of CO2 gas generated at this temperature. This increase becomes sluggish at 850°C. At this temperature, the reduction degree is 11%, indicating the complete reduction of Fe2O3 to Fe3O4, assuming that the reaction in the composite is uniform. On the other hand, the reduction of the Fe2O3–coal composite proceeds continuously without becoming sluggish. This is because volatile matter such as H2 and CH4 generated from coal can reduce iron oxide.12)
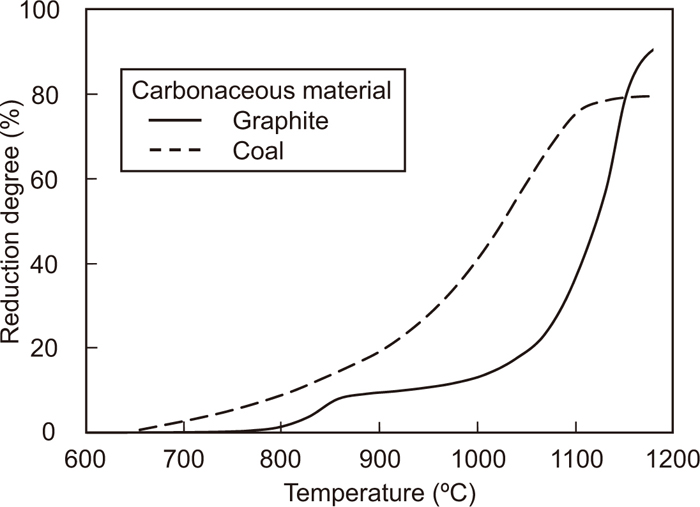
Changes in the reduction degree of Fe2O3-graphite and Fe2O3-coal composites with temperature. (Average particle sizes of Fe2O3, graphite and coal are 2.2, 18.6 and 25.2 μm, respectively.)
Further, a rapid increase in the reduction degree of the Fe2O3–graphite composite is observed at approximately 1050°C. This behavior corresponds to the rapid increase in the generation rate of CO gas, as shown in Fig. 2. It is well known that the gasification reaction described in Eq. (3) is accelerated by the catalytic effect of metallic iron. Ohtsuka et al. have reported on the catalytic effect of iron oxide on the gasification reaction of graphite.17) They suggested that the gasification rate of graphite below 1000°C in the absence of metallic iron and iron oxide is very low.
Under these experimental conditions, metallic iron does not form below 1000°C. The catalytic effect by the redox reaction between Fe2O3 and Fe3O4 may be small17) in the temperature range 700 to 900°C, where the first peak for CO2 gas generation is observed. Therefore, it can be concluded that the effect of the gasification reaction on the gas generation behavior is insignificant below 1000°C. On the other hand, the rapid increase in CO gas generation is caused by the increase in the gasification rate brought about by the catalytic effect of metallic iron.
3.2. Mechanism of Reduction of the Fe2O3–graphite CompositeThe reduction reaction of the Fe2O3-coal composite starts at lower temperature than that of the Fe2O3-graphite composite and reduction degree increases with increasing temperature as shown in Fig. 3. This is due to the indirect reduction by H2 and hydrocarbon gases generated from coal. On the other hand, this reduction in the case of graphite becomes sluggish because direct reduction is required due to lower gasification rate of graphite below 1000°C. Namely, the direct contact between iron oxide and graphite is important for the progress of the reduction reaction at lower temperature than the gasification temperature of carbon, because iron oxide gets off from graphite when the direct reduction reaction proceeds. Brill-Edwards et al. reported on the apparent volume expansion of iron oxide reduced by hydrogen.18) The values for the reduction from Fe2O3 to Fe3O4 and from Fe2O3 to FeO at 825°C were 16.2% and 18.0%, respectively, implying that the volume change from Fe3O4 to FeO is 9 times smaller than that from Fe2O3 to Fe3O4. Therefore, it can be expected that direct reduction from Fe2O3 to Fe3O4 proceeds by continuous contact between iron oxide and graphite, while the reduction from Fe3O4 to FeO is suppressed because of losing the direct contact.
The following two experiments were carried out to confirm such a hypothesis. One is the reduction experiment using the Fe3O4–graphite composite. It is expected that a high reduction rates at 700°C and suppression at 820°C are not observed because it is difficult to achieve continuous contact between iron oxide and graphite particles. Another is the reduction experiment using the composite of iron oxide and graphite powders mix-ground by a planetary mill (mixed-grinding), as described in section 2.2, to increase the contact area between iron oxide and graphite. It is known that the reduction reaction tends to proceed at a lower temperature when using materials with milling treatment.9) If the above hypothesis is correct, the reduction rate will start to decrease when the expansion of iron oxide by the reduction from Fe2O3 to Fe3O4 is completed and the reduction rate of Fe3O4 is much lower than that of Fe2O3.
Figure 4 shows the change in the reduction degree of the composites with temperature. The reduction degree of the Fe3O4–graphite composite is 11% at the start of the experiment. The reduction reaction of the composite using the mixed-grinding sample proceeds even below 400°C. Accordingly, the reduction temperature of this composite was approximately 200°C lower than that of the Fe2O3–graphite composite. However, the reduction rate also decreases at a reduction degree of 11%. A rapid increase in the reduction degree is observed above approximately 900°C. This is because the gasification temperature becomes lower than that of the Fe2O3-graphite composite with increasing contact area between iron oxide and carbonaceous material particles by the mixed-grinding.
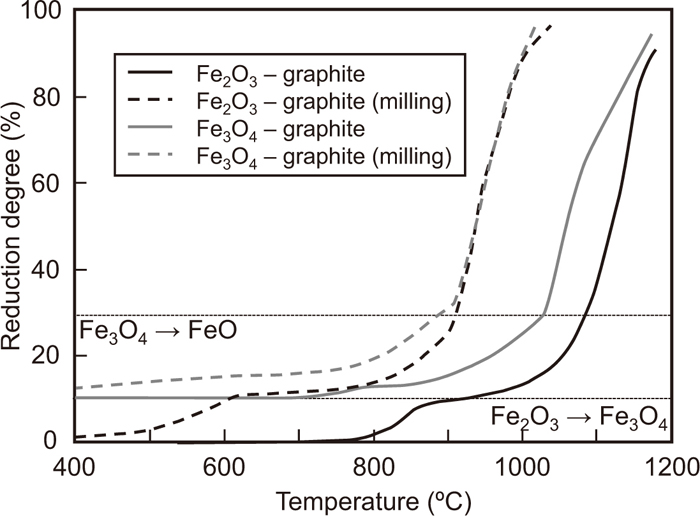
Changes in the reduction degree of Fe2O3-graphite and Fe3O4-graphite composites and composite prepared by milling powder of graphite and Fe2O3 with temperature. (Average particle sizes of Fe2O3, Fe3O4 and graphite are 2.2, 4.2 and 18.6 μm, respectively.)
On the other hand, the reduction from Fe3O4 to FeO in the Fe3O4-graphite composite starts at approximately 700°C, just like that from Fe2O3 to Fe3O4 in the Fe2O3-graphite composite. However, the progress of former reduction is much smaller than that of the latter. Further, the reduction degree is still below 30%, even at a temperature of 1000°C. At last, reduction degree rapidly increases above 1030°C. A lower reduction temperature is achieved for the mixed-grinding sample of Fe3O4 and graphite, compared to the case of the Fe3O4–graphite composite.
Figure 5 shows a schematic image of the reduction model of iron oxide by solid carbon in the composite, which can be expected considering the above results. First, upon heating the composite, the direct reduction from Fe2O3 to Fe3O4, as described in Eq. (1), proceeds at the contact point between Fe2O3 and graphite, and the indirect reduction described in Eq. (2) successively proceeds by CO gas generated through the direct reduction. Since this reaction involves a large volume expansion of iron oxide, as shown in Fig. 5(a), contact points are maintained and/or renewed continuously. Therefore, the reduction from Fe2O3 to Fe3O4 proceeds rapidly even at lower temperatures. However, the reduction from Fe3O4 to FeO involves only little expansion, as shown in Fig. 5(b), and becomes sluggish at a reduction degree of 11%. When the composite is heated up to the gasification temperature, the reduction by CO gas proceeds again. It can be concluded that the direct reduction from Fe2O3 to Fe3O4 proceeds below the gasification temperature.
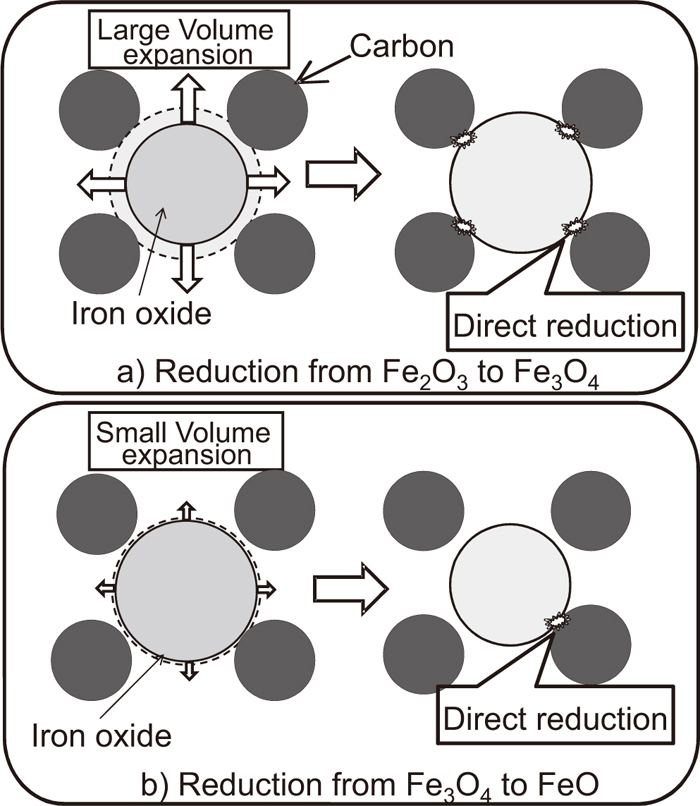
Schematic drawings of composite when iron oxide is reduced from a) Fe2O3 to Fe3O4 and b) Fe3O4 to FeO.
The change in the weight decrease ratio of the composite reduced at 900°C under both inert gas atmosphere and low pressure is shown in Fig. 6 as a function of the thickness of the composite, to discuss the contribution of the direct reduction to the total reaction in the composite. The total pressures are 105 (atmospheric pressure), 10, and 2 Pa. The average particle sizes of Fe2O3 and graphite are 2.2 and 18.6 μm, respectively.
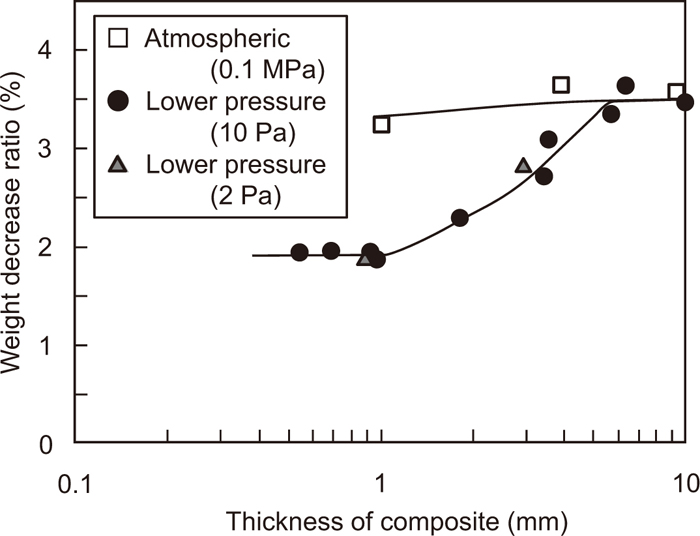
Relation between weight decrease ratio of the composite heated up to 900°C and thickness of the composite. (Average particle sizes of Fe2O3 and graphite are 2.2 and 18.6 μm, respectively.)
In the case of the low-pressure experiment, the weight decrease ratio of the composite with a thickness above 6 mm shows a constant value of approximately 3.5%, which corresponds to that under atmospheric pressure. The reason for these equal reduction degrees is that indirect reduction is fast, and the distance from the point at which CO gas is generated in the composite to the outside of the composite (diffusion distance) is adequately long to cause the reaction with iron oxide. The weight decrease ratio of the composite reduced under atmospheric pressure slightly decreases with a decrease in the composite thickness. On the other hand, the weight decrease ratio of the low-pressure experiment decreases at thicknesses below 6 mm. This is because the contribution of indirect reduction decreases with a decrease in the diffusion distance. Further, when the thickness of the composite is below 1 mm, the weight decrease ratio shows a constant value of 1.9%. This indicates that CO gas generated by direct reduction diffuses to the outside of the composite without being utilized for indirect reduction. Further, decreasing the pressure from 10 to 2 Pa does not lead to any change in the behavior of the weight decrease ratio. Consequently, this value is the weight change of the composite due to the direct reduction at 900°C. Using this value, the contribution ratio of the direct reduction in total reduction reaction is calculated.
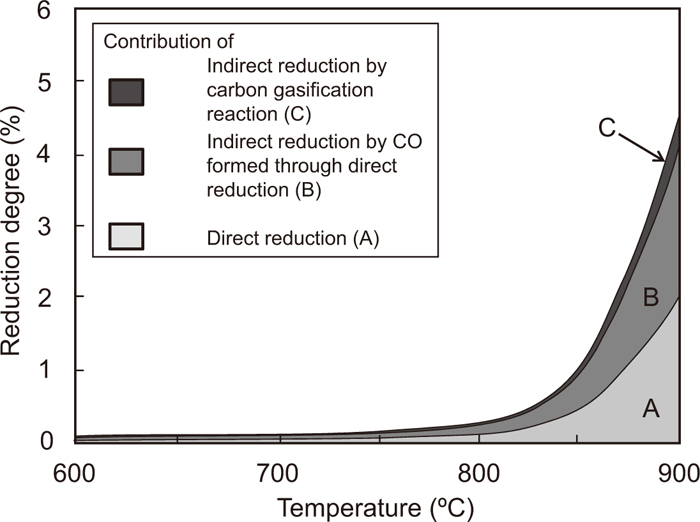
Using the same method, the contribution ratio of the direct reduction at 850, 950, and 1000°C was calculated. The thickness of the Fe2O3-graphite composite, which was used for the calculation, was less than 1 mm. When the target temperature is higher than 950°C, the possibility affected to the phenomena except for direct reduction reaction should be considered. In this study, therefore, the contribution was mainly discussed below 950°C. Figure 7 shows the change in the calculated contribution ratios of the direct reduction and the indirect reduction in the total reduction degree with temperature. Below 800°C, it was difficult to measure the weight decrease ratio of the composite because of the very small weight change. Therefore, it was assumed that the contribution ratio of the direct reduction was 50% since the effect of the gasification reaction can be neglected.17) Area A in Fig. 7 is the contribution of the direct reduction, calculated from the weight decrease ratio in the low-pressure experiment. Areas B and C are those of the indirect reduction by CO gas generated from the direct reduction and the gasification reaction of graphite, respectively.
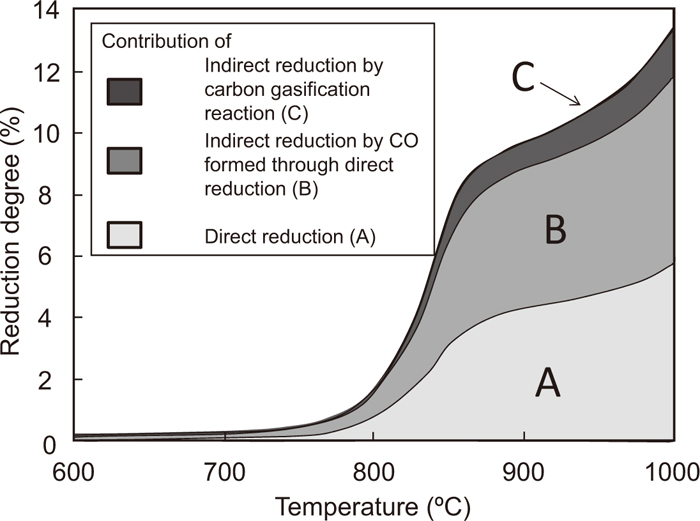
Temperature dependence of the contribution of direct reduction and indirect reduction by CO formed through direct reduction and carbon gasification reactions. (Average particle sizes of Fe2O3 and graphite are 2.2 and 18.6 μm, respectively.)
The reduction degree of the composite reaches 11% at 955°C. The contribution ratios of A, B, and C at this temperature were 44.0%, 45.9%, and 10.1%, respectively. Essentially, the value of A should not be lower than that of B, because the reducing gas of B is attributable to the direct reduction. However, the calculated value of A is slightly lower than that of B. This may be due to experimental error, as the composite with a thickness of less than 1 mm is very lightweight. Accordingly, it is estimated that the calculated contributions include experimental error of up to 2%. Therefore, it can be concluded that the contribution of A is the same as that of B.
3.4. Particle Size Dependence on the Reduction ReactionFigure 8 shows the change in the reduction degree of the composites as a function of temperature, with hematite particle sizes of 2.2, 78.4, and 197.0 μm. The reduction temperature from Fe2O3 to Fe3O4 decreases with decreasing particle size of hematite. This is caused by an increase in the contact points between Fe2O3 and graphite with decreasing particle size. The change in the contribution ratios of the direct and indirect reductions of the composite using Fe2O3 and graphite with temperature is calculated by the same method, and the result is shown in Fig. 9. The particle sizes of Fe2O3 and graphite are 197.0 and 18.6 μm, respectively. The reduction degree does not reach 11% at 900°C, because of the large particle size of hematite. At this temperature, the contribution ratios of A, B, and C are 45.1%, 46.7%, and 8.2%, respectively. It means that the contact point of Fe2O3 and graphite is important factor that the direct reduction from Fe2O3 to Fe3O4 proceeds below the gasification temperature, and the direct reduction is major reaction at this temperature range.
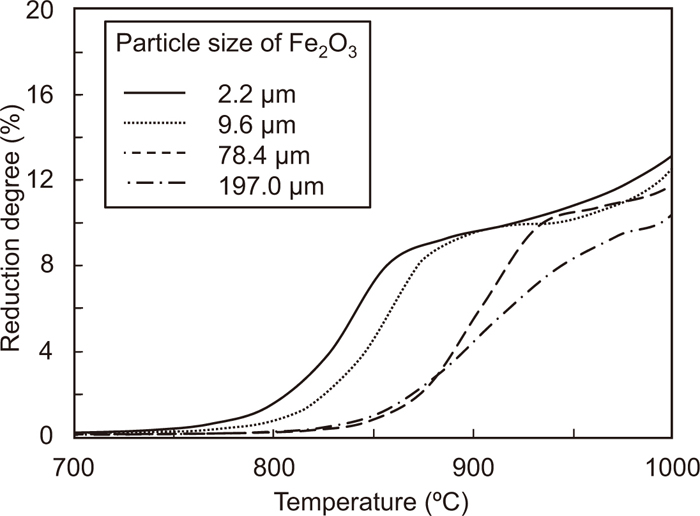
Changes in the reduction degree of Fe2O3-graphite composites with different Fe2O3 size with temperature. (Average particle size of graphite is 18.6 μm.)
Temperature dependence of the contribution of direct reduction and indirect reduction by CO formed through direct reduction and carbon gasification reactions. (Average particle sizes of Fe2O3 and graphite are 197.0 and 18.6 μm, respectively.)
With Fe2O3 particle sizes of 78.4 and 197.0 μm, the results of the low-pressure experiment could not be applied at 950 and 1000°C because the weight decrease ratio caused by the direct reduction could not be calculated. The reason is that the preparation of the composite with a thickness of 1.0 mm was impossible. The weight decrease ratio was calculated using the results obtained with a composite thickness of 1.4 mm. Therefore, the effect of the particle size of hematite will be discussed using the value at 900°C, while the values of reduction degree are not same.
Figure 10 shows the effect of the average particle size of Fe2O3 on the reduction degree and the contribution ratio of the direct reduction at 900°C. The particle size of graphite was 18.6 μm. The contribution ratio of the direct reduction is constant with increasing average particle size of hematite, while the reduction degree at 900°C decreases.
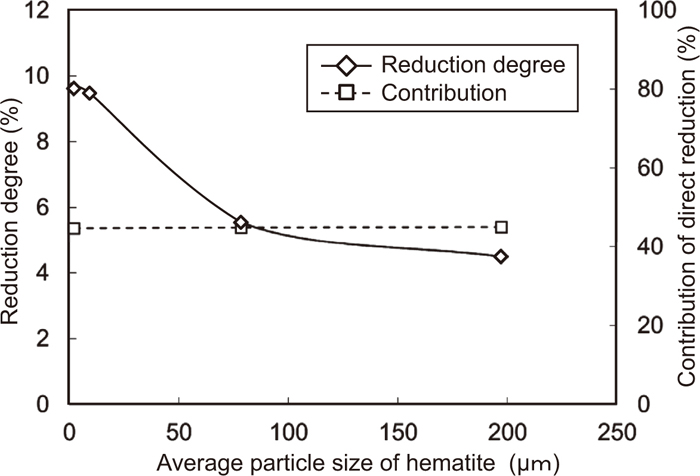
Effect of the average particle size of Fe2O3 on reduction degree and contribution ratio of direct reduction of the composite heated up to 900°C. (Particle size of graphite is 18.6 μm.)
The change in the reduction degree of the composite of Fe2O3 with an average particle size of 2.2 μm and graphite with particle sizes of 14.9, 18.6, and 22.1 μm with temperature is shown in Fig. 11. In all particle size of graphite, the reduction reaction becomes sluggish at the reduction degree of 11%, indicating the complete reduction of Fe2O3 to Fe3O4. However, the temperature of the complete reduction decreases upon decreasing the average particle size of graphite. This is because the number of contact points increases with decreasing particle size. Above 1000°C, the temperature at which the reduction degree starts to increase rapidly decreases with increasing particle size. For the composite of Fe2O3 and graphite with the particle sizes of 2.2 and 14.9 μm, respectively, the change in the contribution ratios of the direct reduction and the indirect reduction in the total reduction degree with temperature is calculated by the same method as that shown in Fig. 7; the result is shown in Fig. 12. The contribution ratios of these reactions show similar behavior as that depicted in Fig. 7, even as the particle size of graphite decreases. Figure 13 shows the contribution ratio of these reactions and the temperature at which the reduction degree reaches 11%. The particle size of hematite was 2.2 μm. The contribution ratio is constant, even if the particle size of graphite is changed. This is because the gasification rate of graphite is very small below these temperatures.
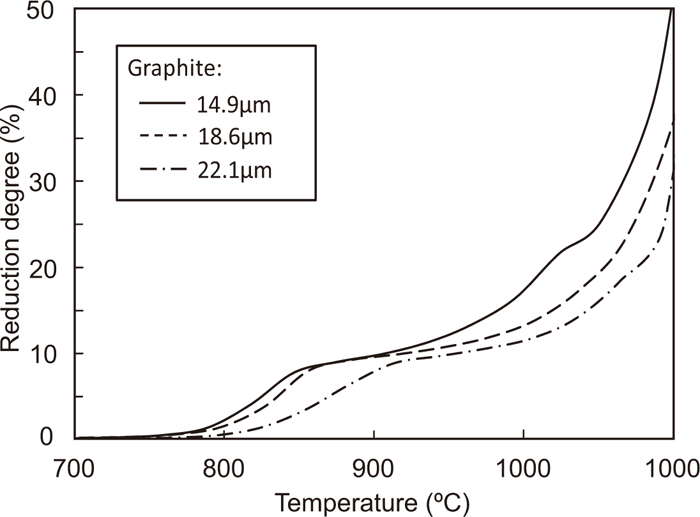
Changes in the reduction degree of Fe2O3-graphite composites having different graphite sizes with temperature. (Average particle size of Fe2O3 is 2.2 μm.)
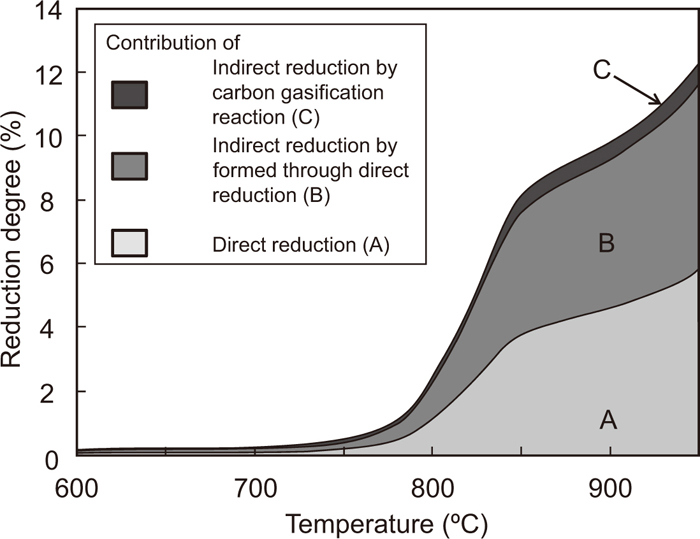
Temperature dependence of the contribution of direct reduction and indirect reduction by CO generated from direct reduction and gasification reaction. (Average particle sizes of Fe2O3 and graphite are 2.2 and 14.8 μm, respectively.)
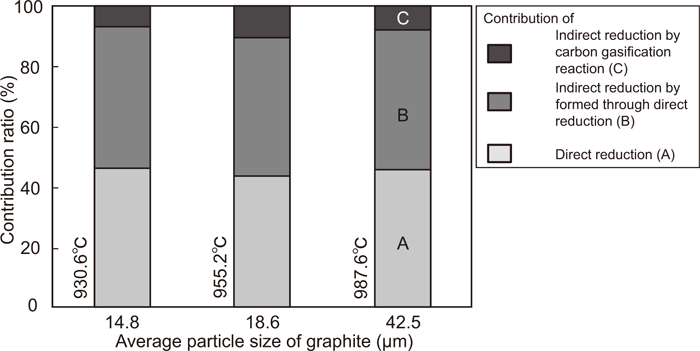
Effect of average size of graphite on the contribution ratio of each reactions and temperature when reduction degree reached at 11.1%. (Average particle size of Fe2O3 is 2.2 μm.)
In this study, the temperature range at which direct reduction progresses is different from that for the gasification reaction because graphite possesses rather higher gasification temperature. Therefore, the effect of particle size on the contribution ratios of these reactions is insignificant. If carbonaceous material with a high gasification temperature such as coke is used, similar results will be obtained. On the other hand, if a material with a low gasification temperature such as charcoal is used, the contribution of direct reduction will decrease. For the effective utilization of direct reduction, it is very important to increase the contact area between iron oxide and the carbonaceous material particles. In fact, mixed-grinding leads to lowering the temperature at which the reduction from Fe2O3 to Fe3O4 occurs. However, this effect is not significant in the case of the reduction from Fe3O4 to FeO, indicating that not only mixed-grinding but also other innovative methods are required to promote the direct reduction.
The reduction mechanism for an iron oxide-graphite composite below 1000°C, where graphite shows a low gasification rate, is discussed. The contribution ratios of direct reduction by solid carbon and indirect reduction by CO gas (generated from the direct reduction and carbon gasification) were estimated through reduction experiment of the composite under inert and different pressure atmospheres. The following results are obtained.
(1) The reduction from Fe2O3 to Fe3O4 proceeds at low temperatures, at which the gasification rate is very small. During this period, direct reduction proceeds because new contact points between Fe2O3 and graphite are formed continuously due to the large volume expansion of iron oxide. The contribution ratio of the direct reduction is approximately 45% during the reduction from Fe2O3 to Fe3O4.
(2) The contribution ratio of the direct reduction in the total reduction reaction during the reduction from Fe3O4 to FeO is small, because it does not lead to a large volume expansion. Therefore, the rate of the direct reduction remains at a lower value until temperature reaches the point at which the gasification reaction becomes active.
(3) Decreasing the average particle sizes of Fe2O3 and graphite leads to lowering the reduction temperature owing to the increased contact between these particles. Below 1000°C, however, the contribution ratio of the direct reduction in the total reduction reaction is constant.
The authors are grateful for the financial support to the present study from the research promotion grant from the Iron and Steel Institute of Japan, by the Steel Industry Foundation for the Advancement of Environmental Protection Technology.