2023 Volume 73 Issue 3 Pages 343-348
2023 Volume 73 Issue 3 Pages 343-348
Golden cyst nematodes have threatened the cultivation of ‘Toyoshiro’, a major potato variety used for chip processing in Japan. Common scab is a soilborne disease that occurs in potato fields worldwide. To solve these problems, we crossed two US varieties and selected a clone that showed a slightly higher marketable yield and a significantly higher yield rate, compared with ‘Toyoshiro’, and had good chip processing quality, extreme resistance to cyst nematodes, and moderately high resistance to common scab. This clone was named ‘Poroshiri’; it is the first variety released from the Calbee Potato breeding program.
Potato chips have been popular in Japan since the 1970s. The chip processing varieties ‘Waseshiro’ (early maturity) and ‘Toyoshiro’ (medium-early maturity) were released in 1974 and 1976, respectively. The high yield and good quality of ‘Toyoshiro’ promoted the rapid growth of the potato chipping industry. ‘Toyoshiro’ is still widely grown in Hokkaido (6,044.5 ha in 2021). Golden cyst nematodes (Globodera rostochiensis) were first found in Hokkaido in 1972 and spread rapidly; they have threatened potato cultivation because none of the top four varieties, ‘Irish Cobbler’, ‘May Queen’ (both for table stock), ‘Toyoshiro’ (for chipping), and ‘Konafubuki’ (for starch production), in Japan are resistant to golden cyst nematodes. Cyst nematodes significantly reduce yields. Furthermore, once the field is infested, it can never be used for seed tuber production. Thus, over the last three to four decades, there has been constant struggle against cyst nematodes (Mori et al. 2007). Common scab is a soilborne disease that occurs in potato worldwide. Scab is usually superficially present on the tuber surface and rarely invades internal tissues. However, severe infestations occur during warm, dry seasons in soils with a relatively high pH (Braun et al. 2017, Loria 2001) and often reduce tuber quality.
Calbee Potato, Inc. (hereafter abbreviated ‘CP’), a subsidiary company of Calbee, Inc., started introducing chip processing varieties from the United States of America (US) in 1984. Most of their introductions were resistant to golden cyst nematodes and/or common scab. The company started intervarietal cross-breeding of these varieties in 2003. ‘Poroshiri’ is the first variety released from the CP potato breeding program. It is a chip processing variety with extreme resistance to cyst nematodes and moderately high resistance to common scab.
The pedigree of ‘Poroshiri’ is shown in Fig. 1. The female parent ‘NorKing Russet’ is a medium-maturing russet variety with common scab resistance released from the North Dakota potato breeding program in 1986 (Johansen et al. 1986). It was introduced by CP and officially filed in 2002. The male parent ‘Pike’ is a common scab-resistant variety carrying a cyst nematode resistance gene (H1) with low reducing sugar accumulation released by Cornell University and the Pennsylvania Agricultural Experiment Station in 1996 (Plaisted et al. 1998).
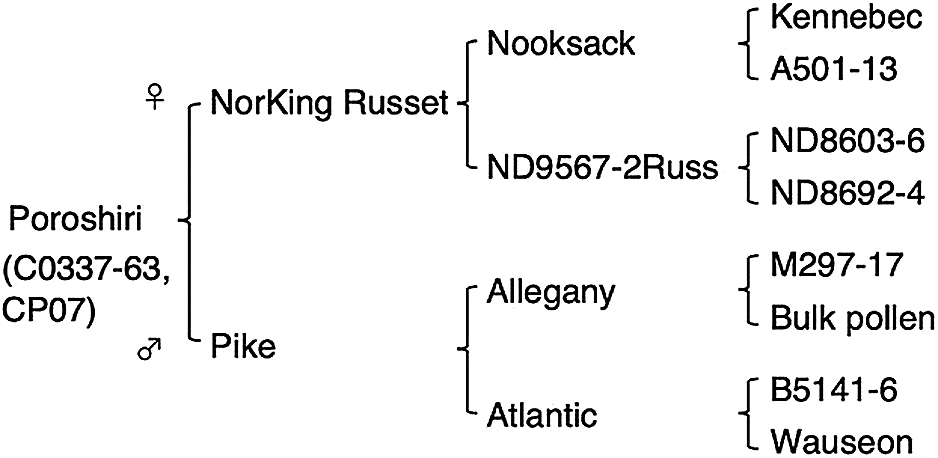
Pedigree of ‘Poroshiri’.
Hybrid seeds were obtained by artificial pollination. Seedlings were raised in pots (12 cm diameter), and one tuber from each pot was harvested. In the following year (first clonal selection), these tubers were planted into rows with 75 cm between rows in the field. Since each hill was a genetically different plant, a wide distance between hills (90 cm) was applied to separate neighboring genotypes. At harvest, superior genotypes were visually selected based on tuber size, number and shape in the field. After harvest, further selection was made based on glucose content and chip color after 5 months of storage at 6°C. Selected genotypes were subjected to line selection: 10 hills per genotype planted in one row with 75 cm between rows and 27 cm between hills. At harvest, high-yielding genotypes producing ovate- or round-shaped tubers with shallow eyes and a uniform size distribution were visually selected in the field. Chip color tests were performed just after harvest in mid-October and in late February after 4 months of storage at 6°C. A bioassay for resistance to cyst nematodes was conducted during the winter season using a plastic cup method (https://www.naro.affrc.go.jp/org/harc/seika/h19/304.html), and susceptible genotypes were discarded. The selected genotypes were subjected to a preliminary yield trial: for each genotype, there were 10 hills in 4 rows with 75 cm between rows and 27 cm between hills. Tuber yields were measured at harvest, and chip color and glucose and sucrose contents were measured at harvest and after 4 months and 6 months of storage under four storage conditions: 6°C storage, reconditioned at 15°C for 2 weeks after 6°C storage, 9°C storage, and reconditioned at 15°C for 2 weeks after 9°C storage. Simultaneously, resistance to common scab was evaluated by growing some tubers in an infested field. For several years, yield trials were conducted: for each genotype, there were 10 hills in 4 rows with 75 cm between rows and 27 cm between hills with 3 replications. In addition to the measurements performed in the preliminary yield trial, chip color and glucose and sucrose contents were measured after 8 months of storage at 6°C and after reconditioning at 15°C for 2 weeks after 4, 6, and 8 months of storage at 4°C. Cyst nematode resistance assays, common scab resistance assays, tuber bruising tolerance assays, and culinary tests were conducted every year during yield trials. Fields were fertilized in the range of 1–8 kg 10 a–1 N, 17.6–28.5 kg 10 a–1 P, 6–10 kg 10 a–1 K, and 0–7.5 kg 10 a–1 Mg depending on yearly soil nutrient tests. Seed tubers were usually planted in the field in mid-May. Tubers were harvested in late August for first clonal selection and line selection and in mid-September for the preliminary yield trial and subsequent yield trials.
Bruising tolerance assayTubers were chilled at 6°C before testing. Thirty chilled tubers for each genotype were placed together in a drum chamber and rotated electrically at a speed of 8.75 rotations per minute for 4 minutes. After this treatment, the tubers were placed at approximately 10°C for a few days for curing and then placed at 30°C for 72 h to promote bruising damage. Tuber skins were peeled off, and black spots >8 mm in diameter were counted.
Common scab resistance assayCommon scab resistance assays were conducted in infested fields at two locations, the Potato Research Center of CP and the Hokkaido Agricultural Research Center (HARC) at Memuro. Both fields were infested predominantly with Streptomyces turgidiscabies, which is a major causal agent of common scab in eastern Hokkaido (Miyajima et al. 1998). Three plants at the CP and five plants at the HARC locations were planted per genotype in accordance with a randomized complete block design, with two replications. All mature tubers (>30 g at the CP and >20 g at the HARC locations) were harvested, washed, and assessed on the basis of the surface area covered by scabs (0, no scab symptoms; 1, <3% coverage or one to three scabs; 2, 4–13% coverage or 4 to 10 scabs; 3, 14–25% coverage or 11 to 20 scabs; and 4, >26% coverage or >21 scabs). The genotype means were readjusted to a disease rating scale of 1–7 every year using the following standard varieties: Yukirasha (resistant, score of 7), NorKing Russet (score of 6), Pike (score of 5), Snowden (medium, score of 4 or 5), and Toyoshiro (susceptible, score of 1 or 2) (Koizumi et al. 2021).
In 2003, ‘NorKing Russet’ was pollinated with the pollen of ‘Pike’, and 1,232 hybrid seeds were obtained. In 2004, 1,000 seedlings with the family name ‘C0337’ were raised in pots, from which 793 genotypes were selected. In 2005, the 793 genotypes were subjected to first clonal selection in the field. Eighty genotypes were visually selected in the field, from which 53 genotypes were selected after chip quality tests such as chip color and glucose content (the line ‘Poroshiri’ was originally called ‘C0337-63’). In 2006, 53 genotypes were planted for line selection, of which 17 were visually selected in the field and seven were selected after harvest. In 2007, the seven genotypes were subjected to a preliminary yield trial, from which three were selected. After four years of yield trials from 2008 to 2011, one genotype was selected and named ‘CP07’. To test regional adaptability, yield trials were continued from 2012 to 2014 in seven farmer’s fields distributed throughout Hokkaido. The results showed that ‘CP07’ was a medium-maturing genotype with equivalent processing quality and slightly higher yield than ‘Toyoshiro’. It also had extremely high resistance to cyst nematodes and moderately high resistance to common scab.
Thus, ‘CP07’ was named ‘Poroshiri’, and official registration paperwork for this clone was filed on June 12, 2013. ‘Poroshiri’ was officially registered on February 28, 2017 and announced as a new variety on March 8, 2017. ‘Poroshiri’ was named after ‘Mt. Poroshiri’ (meaning ‘a big mountain’ in Ainu), the highest peak among the Hidaka mountains that surround the Tokachi Plain where the CP breeding field is located. We hope that ‘Poroshiri’ becomes a popular variety.
Morphological descriptionsThe plant, leaf, flower, tuber and sprouting characteristics of ‘Poroshiri’ are shown in Fig. 2. The growth habit of ‘Poroshiri’ is semierect, similar to ‘Toyoshiro’. Stems are more pigmented with anthocyanin and are slightly shorter and thicker than those of ‘Toyoshiro’. Flowers are light pinkish purple, and there are fewer flowers produced than ‘Toyoshiro’. Tubers are ovate with very shallow eyes. Tuber skins are slightly russeted with a light beige color, and the flesh is white.

Plant, leaf, flower, tuber and sprout characteristics of ‘Poroshiri’.
‘Poroshiri’ grows vigorously at the early growing stage, similar to ‘Toyoshiro’, and is a medium-maturing variety with an approximately seven-day longer maturity period than ‘Toyoshiro’ (Table 1). The average tuber weight and total marketable yield of ‘Poroshiri’ were slightly higher than those of ‘Toyoshiro’; the 7-year averages (2008–2014) were 122 g and 4,684 kg 10 a–1, respectively, for ‘Poroshiri’ and 117 g and 4,311 kg 10 a–1, respectively, for ‘Toyoshiro’ (Table 1). The yield rate (=100 × marketable yield/total yield) of ‘Poroshiri’ was 88.4%, which was significantly higher (p < 0.01 by t test) than that of ‘Toyoshiro’ (84.0%). External disorders such as cracks and secondary growth occurred to the same level as ‘Toyoshiro’, whereas the occurrence of common scab was significantly lower in ‘Poroshiri’ (0.1%) than ‘Toyoshiro’ (7.5%) (Table 2). There was no significant difference between the two varieties in the occurrence of tuber rotting in the Tokachi area, although a higher occurrence after the incidence of late blight was reported for ‘Poroshiri’ compared with ‘Toyoshiro’ in the other areas (https://www.hro.or.jp/list/agricultural/center/kenkyuseika/gaiyosho/27/f0/03.pdf). Internal defects such as hollow heart and internal brown spots occurred to the same level as ‘Toyoshiro’ (Supplemental Table 1). The bruising tolerance (Table 3) and length of the tuber dormancy period (data not shown) of ‘Poroshiri’ were similar to those of ‘Toyoshiro’.
| Year | Tuber number (no./hill) | Tuber weight (g/hill) | Mean tuber weight (g) | Total yield (kg 10 a–1)a | Marketable yield (kg 10 a–1)b | Yield rate (%)c | Maturityd |
|---|---|---|---|---|---|---|---|
| 2008 | 8.4 ± 0.29 ns | 976 ± 39.0 ns | 116 ± 0.6 ns | 5,518 ± 363.4 ns | 4,822 ± 192.5 ns | 87.5 ± 2.36 ns | August 16 |
| 8.0 ± 0.59 | 846 ± 114.1 | 105 ± 8.2 | 4,993 ± 497.9 | 4,176 ± 563.3 | 83.4 ± 3.42 | August 9 | |
| 2009 | 6.1 ± 0.28*** | 774 ± 73.1 ns | 126 ± 7.8 ns | 4,076 ± 327.5* | 3,820 ± 361.2 ns | 93.7 ± 1.32 ns | August 23 |
| 7.2 ± 0.14 | 867 ± 105.0 | 121 ± 14.3 | 4,734 ± 400.0 | 4,280 ± 518.7 | 90.2 ± 3.95 | August 16 | |
| 2010 | 5.6 ± 0.58* | 777 ± 52.8 ns | 141 ± 16.8 ns | 4,176 ± 299.4 ns | 3,839 ± 260.6 ns | 91.9 ± 1.35 ns | August 24 |
| 4.6 ± 0.55 | 711 ± 82.1 | 155 ± 7.9 | 4,160 ± 229.0 | 3,509 ± 405.2 | 84.2 ± 7.12 | August 23 | |
| 2011 | 9.4 ± 0.82* | 1,236 ± 78.7** | 132 ± 3.2 ns | 6,836 ± 546.4** | 6,101 ± 388.4** | 89.3 ± 1.55 ns | August 24 |
| 8.0 ± 0.65 | 1,007 ± 83.3 | 126 ± 6.7 | 5,615 ± 370.8 | 4,972 ± 411.2 | 88.5 ± 2.76 | August 16 | |
| 2012 | 7.6 ± 1.52 ns | 729 ± 202.0 ns | 95 ± 8.6 ns | 4,681 ± 622.5 ns | 3,599 ± 997.6 ns | 75.8 ± 12.03 ns | August 22 |
| 7.0 ± 0.96 | 637 ± 123.4 | 90 ± 5.94 | 4,502 ± 477.1 | 3,144 ± 609.1 | 69.3 ± 7.08 | August 17 | |
| 2013 | 8.4 ± 0.31* | 980 ± 49.4 ns | 117 ± 7.3*** | 5,442 ± 251.2 ns | 4,839 ± 243.8 ns | 88.9 ± 0.66* | August 28 |
| 9.2 ± 0.40 | 905 ± 65.6 | 98 ± 3.7 | 5,476 ± 218.6 | 4,467 ± 324.2 | 81.5 ± 3.57 | August 15 | |
| 2014 | 9.1 ± 0.13 ns | 1,168 ± 89.2 ns | 128 ± 8.2 ns | 6,308 ± 406.8 ns | 5,766 ± 440.3 ns | 91.4 ± 1.63 ns | August 19 |
| 9.5 ± 0.86 | 1,140 ± 124.1 | 120 ± 7.6 | 6,212 ± 637.5 | 5,629 ± 613.0 | 90.7 ± 4.79 | August 8 | |
| Mean | 7.8 ± 1.45 ns | 949 ± 200.1 ns | 122 ± 14.5 ns | 5,291 ± 1047.7 ns | 4,684 ± 988.1 ns | 88.4 ± 5.90** | August 22** |
| 7.6 ± 1.64 | 873 ± 169.9 | 117 ± 21.6 | 5,099 ± 710.5 | 4,311 ± 839.1 | 84.0 ± 7.37 | August 14 |
a Total weight of tubers weighing ≥10 g.
b Total weight of tubers weighing 60 g≤ but <340 g.
c Yield rate = 100 × marketable yield/total yield.
d Maturity was indicated by the date when half of the leaves turned yellow.
Significant differences were tested by t test: ns, not significant, * p < 0.05, ** p < 0.01, and *** p < 0.001.
| Cultivar | Mean number of examined tubers | Normal (%) | Rotting (%) | Common scab (%) | Powdery scab (%) | Malformation or cracking (%) | Secondary growth (%) | Greening (%) |
|---|---|---|---|---|---|---|---|---|
| Poroshiri | 145 ± 38.7 | 96.3 ± 0.03* | 0.2 ± 0.01 ns | 0.1 ± 0.00* | 0.1 ± 0.00 ns | 0.1 ± 0.00 ns | 0.6 ± 0.01 ns | 2.6 ± 0.03 ns |
| Toyoshiro | 274 ± 108.2 | 82.0 ± 0.13 | 0.2 ± 0.00 | 7.5 ± 0.06 | 5.2 ± 0.13 | 0.2 ± 0.01 | 0.2 ± 0.00 | 4.8 ± 0.05 |
Significant differences were tested by t test: ns, not significant, and * p < 0.05.
| Year | Poroshiri | Toyoshiro |
|---|---|---|
| 2008 | 1.9 | 1.6 |
| 2009 | 2.0 | 2.1 |
| 2010 | 2.6 | 2.7 |
| 2011 | 2.2 | 2.2 |
| 2012 | 2.3 | 2.9 |
| 2013 | 2.5 | 2.7 |
| 2014 | 1.6 | 2.8 |
| Mean | 2.1 ± 0.36 ns | 2.4 ± 0.46 |
Significant differences were tested by t test: ns, not significant at a 5% level.
The starch content of ‘Poroshiri’ was slightly lower than that of ‘Toyoshiro’ (15.8% and 16.0%, respectively) (Table 4). Chip colors were measured using an Agtron meter (green): Agtron values of ≥40 are acceptable for chip processing. The chip color of ‘Poroshiri’ at harvest was similar to that of ‘Toyoshiro’ (Agtron values of 40.3 and 38.8 respectively). However, their Agtron values were reduced to 27.5 and 31.9, respectively, after 4 months of storage at 9°C (Supplemental Fig. 1). Tubers of ‘Poroshiri’ at harvest contained significantly lower glucose and sucrose contents (0.49 and 1.36 mg/g, respectively) than those of ‘Toyoshiro’ (0.62 and 1.72 mg/g, respectively). However, after 4 months of storage at 9°C, the two varieties increased their sugar contents to higher levels that were not suitable for long storage. The sprout length of ‘Poroshiri’ after storage at 9°C was significantly shorter (4.8 mm) (p < 0.01 by t test) than that of ‘Toyoshiro’ (14.1 mm) (Table 4).
| Year | Specific gravity | Starch content (%)a | Chip colorb | Glucose (mg/g) | Sucrose (mg/g) | Sprout length (mm)c | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| At harvest | After storagec | At harvest | After storagec | At harvest | After storagec | ||||||
| 2008 | 1.089 | 15.9 | 28.8 | 32.6 | 1.49 | 1.29 | 2.16 | 2.64 | 1.1 | ||
| 1.086 | 15.2 | 25.8 | 27.4 | 1.77 | 2.46 | 1.86 | 1.78 | 14.3 | |||
| 2009 | 1.093 | 16.7 | 45.0 | 29.4 | 0.40 | 1.05 | 1.42 | 2.36 | 4.4 | ||
| 1.092 | 16.5 | 43.4 | 33.6 | 0.54 | 0.99 | 2.16 | 1.82 | 9.3 | |||
| 2010 | 1.084 | 14.8 | 47.2 | 30.6 | 0.16 | 1.19 | 0.94 | 2.04 | 15.1 | ||
| 1.088 | 15.7 | 43.2 | 34.0 | 0.34 | 0.69 | 1.52 | 1.70 | 15.6 | |||
| 2011 | 1.082 | 14.4 | 34.4 | 22.2 | 0.65 | 1.55 | 1.46 | 1.34 | 2.4 | ||
| 1.083 | 14.6 | 34.8 | 26.4 | 0.62 | 0.95 | 2.10 | 1.38 | 19.3 | |||
| 2012 | 1.095 | 17.2 | 45.4 | 24.2 | 0.27 | 1.67 | 1.52 | 3.52 | 2.9 | ||
| 1.095 | 17.2 | 47.6 | 32.4 | 0.23 | 1.04 | 1.54 | 1.76 | 12.4 | |||
| 2013 | 1.093 | 16.7 | 40.0 | 26.2 | 0.29 | 1.59 | 1.06 | 1.58 | 4.6 | ||
| 1.099 | 18.0 | 39.2 | 33.6 | 0.54 | 0.69 | 1.42 | 1.48 | 18.7 | |||
| 2014 | 1.083 | 14.6 | 41.4 | 27.6 | 0.19 | 1.29 | 0.94 | 1.32 | 2.9 | ||
| 1.085 | 15.0 | 37.4 | 35.6 | 0.29 | 0.80 | 1.46 | 1.36 | 9.1 | |||
| Mean ± SD | 1.088 ± 0.0054 ns | 15.8 ± 1.15 ns | 40.3 ± 6.63 ns | 27.5 ± 3.65* | 0.49 ± 0.469* | 1.38 ± 0.230 ns | 1.36 ± 0.432* | 2.11 ± 0.799 ns | 4.8 ± 4.71** | ||
| 1.090 ± 0.0058 | 16.0 ± 1.25 | 38.8 ± 7.13 | 31.9 ± 3.53 | 0.62 ± 0.528 | 1.09 ± 0.62 | 1.72 ± 0.313 | 1.61 ± 0.198 | 14.1 ± 4.11 | |||
a Starch content (%) = (specific gravity–1.05) × 214.5 + 7.5 (Remy 1928).
b Agtron values (green) obtained using a photoelectric color meter (Agtron M-35, Magnuson Corporation, Reno, USA). Larger Agtron values indicate lighter and whiter colors. Agtron values ≥40 are acceptable for chips.
c Stored for 4 months at 9°C.
Significant differences were tested by t test: ns, not significant, * p < 0.05, and ** p < 0.01.
‘Poroshiri’, carrying the H1 gene inherited from ‘Pike’, showed extreme resistance to golden cyst nematodes (pathotype Ro1) (Supplemental Table 2). The common scab resistance was moderately high with a score of 6, which was the same resistance level as the ‘NorKing Russet’ parent (Table 5). According to resistance evaluation tests against late blight (Phytophthora infestans) and Potato virus Y, conducted at the HARC and the Hokkaido Research Organization, respectively, ‘Poroshiri’ was regarded as susceptible for them, similar to ‘Toyoshiro’.
| Year | Location | Poroshiri | Yukirasha | NorKing Russet | Pike | Snowden | Toyoshiro | Irish Cobbler |
|---|---|---|---|---|---|---|---|---|
| 2008 | HARC | 6 | – | – | – | 5 | 1 | 2 |
| 2009 | HARC | 6 | 7 | – | – | 5 | 1 | 1 |
| 2010 | CP | 6 | 7 | 6 | 5 | – | 2 | – |
| 2011 | HARC | 5 | – | – | – | – | 1 | 1 |
| 2020 | HARC | 6 | – | – | – | – | 2 | – |
| CP | 6 | 7 | 6 | 5 | 4 | 2 | – | |
| 2021 | CP | 6 | 7 | 6 | 5 | 4 | 2 | – |
Scored on a 1–7 scale, where 1 indicates the worst disease symptoms on the tubers and 7 indicates no disease present on the surface of tubers.
We bred ‘Poroshiri’, which is a slightly better chip processing variety than ‘Toyoshiro’ in terms of tuber yield and yield rate and possesses extreme resistance to golden cyst nematodes and moderately high resistance to common scab. The lower occurrence of scabby tubers in ‘Poroshiri’ is preferred by chip-processing companies because labor intensity in hand-trimming of scab is significantly reduced. Thus, ‘Poroshiri’ can replace ‘Toyoshiro’, particularly in fields infested with golden cyst nematodes and/or in fields with a high occurrence of common scab. According to the latest record, the cultivation area of ‘Poroshiri’ increased to 800.9 ha in 2019 (https://www.maff.go.jp/j/seisan/tokusan/imo/r3shiryou.html). However, a longer storage period of ‘Poroshiri’ increases the reducing sugar content to an extent similar to ‘Toyoshiro’, which results in unacceptably dark chip colors. Thus, ‘Poroshiri’ is a chip processing variety for short-term storage and can be used until the end of January. For a year-round supply, low reducing sugar accumulation after long, cold storage is highly desirable for future breeding efforts.
According to the genome-wide association analysis of common scab resistance (Koizumi et al. 2021), the resistance phenotype of ‘Poroshiri’ is likely associated with the quantitative trait loci (QTLs) identified at 74.4 Mb on chromosome 1 and at 27.4 Mb on chromosome 2 (Kaiser et al. 2020). Since the resistance QTL on chromosome 1 is recessive, it could be difficult to use in tetraploid breeding (Koizumi et al. 2021). However, the QTL on chromosome 2 is dominant and has been identified repeatedly (Bradshaw et al. 2008, Kaiser et al. 2020, Yuan et al. 2020). Therefore, ‘Poroshiri’ could be an important source for breeding common scab-resistant varieties.
MT, SO, KO, TI, and EK were deeply involved in all the breeding process, and EK wrote the manuscript with the aid of TI.
‘Poroshiri’ was officially registered under the Plant Variety Protection and Seed Act with the applicants Hiroyuki Uemura, Seiya Ohashi, Shoji Tsumiyama, Takashi Murata, Keiichi Ogawa, Shogo Ogawa, Mutsuo Tsuyama, Toshiya Igarashi, Masahiro Nishimoto, and Emiko Koizumi. We thank the staff at the Hokkaido Agricultural Research Center, NARO, for the common scab resistance assay and the many staff at the agricultural experiment stations and extension centers of Hokkaido Research Organization and local farmers, who conducted regional adaptability tests. We also thank Dr. Kazuyoshi Hosaka, Obihiro University of Agriculture and Veterinary Medicine, for improving the manuscript and Kayoko Fujiwara for technical assistance.