2016 Volume 39 Issue 8 Pages 1273-1283
2016 Volume 39 Issue 8 Pages 1273-1283
This study was conducted to evaluate the effects of Sargassum muticum extract and apo-9′-fucoxanthinone, a principal component of S. muticum, on hair growth. When rat vibrissa follicles were treated with S. muticum extract for 21 d, the hair-fiber lengths for the vibrissa follicles increased significantly. Treatment with the S. muticum extract and the EtOAc fraction of the S. muticum extract markedly increased the proliferation of dermal papilla cells (DPCs) and decreased the 5α-reductase activity. In addition, the EtOAc fraction of the S. muticum extract significantly promoted anagen initiation in C57BL/6 mice. Especially, apo-9′-fucoxanthinone, an active constituent from the S. muticum extract, caused an increase in DPC proliferation and a decrease in 5α-reductase activity. To elucidate the molecular mechanisms of apo-9′-fucoxanthinone on the proliferation of DPCs, we examined the level of various signaling proteins. Apo-9′-fucoxanthinone increased the level of vascular endothelial growth factor receptor-2 (VEGF-R2), Wnt/β-catenin signaling proteins such as phospho(ser9)-glycogen synthase kinase-3β (GSK-3β) and phospho(ser552)-β-catenin, whereas apo-9′-fucoxanthinone did not affect the transforming growth factor-β (TGF-β) signaling proteins such as Smad2/3. These results suggest that apo-9′-fucoxanthinone from S. muticum could have the potential for hair growth with DPC proliferation via the activation of Wnt/β-catenin signaling and the VEGF-R2 pathway.
Hair loss (known as alopecia) is currently highlighted as an increasing disorder in both men and women1,2) with complex causes that cannot be solved easily. The imbalances of hormones such as thyroid, estrogen and androgen, as well as autoimmune diseases, stress, shock, poor nutrition and so on have been known to induce alopecia.3–6) Alopecia can also occur due to chemotherapy and fungal infections.7,8) Even though demand for alopecia therapy is increasing, only two hair-growth agents, i.e., finasteride (Propecia®) and minoxidil (Rogaine®), have been approved by the Food and Drug Administration for the treatment of alopecia.7,9,10) Finasteride, a type II 5α-reductase inhibitor, can be used in prostatic hypertrophy as well as androgenetic alopecia.9) 5α-Reductase enzyme converts testosterone to dihydrotestosetrone (DHT) and is responsible for androgenetic alopecia.7,9) DHT, a potent form of testosterone, can cause shortening of the anagen phase, resulting in the miniaturization of hair follicles.7,9)
Minoxidil, an ATP-sensitive K+-channel opener, was developed as an anti-hypertensive.10) It has a desirable hair-growth promoting side effect and can be used for alopecia treatment. The action mechanisms of minoxidil on hair growth include the increase of ATP-sensitive K+-channel opening, the up-regulation of vascular endothelial growth factor (VEGF) and the activation of the β-catenin pathway in dermal papilla cells (DPCs).11–13) However, because of unwanted side effects and transient action, dissatisfied responses to finasteride and minoxidil in alopecia have been reported.14,15) Therefore, the demand for new therapies for alopecia is on the rise.
DPCs, fibroblasts of mesenchymal origin, regulate hair growth via the activation of hair germ cells.16–18) Previous studies demonstrated that minoxidil increased the proliferation of DPCs by regulating apoptosis-related proteins and extracellular signal-regulated kinase (ERK).18) Indeed, minoxidil extends the anagen (growth) phase of the hair cycle via the activation of Wnt/β-catenin in DPCs.13) The expression of Wnt/β-catenin proteins has been observed in stem cells,19) cancer cells20) and hair follicle cells21) and several studies suggest that the activation of β-catenin, the main component of Wnt/β-catenin, regulates stem cell function,19) cell survival20) and proliferation.22) The level of β-catenin is regulated by the disruption complex, including axin, adenomatous polyposis coli, casein kinase I and glycogen synthase kinase-3β (GSK-3β). GSK-3β inhibitor increases hair growth resulting from the proliferation of DPCs by increasing the β-catenin level.13,23) VEGF is an important regulator in angiogenesis and tumorigenesis.24–26) The effect of VEGF is mediated by the binding of VEGF receptor-2 (VEGF-R2) and recent studies showed that VEGF-mediated proliferation effects have been observed in DPCs and outer root sheaths (ORS).27,28) These studies suggest that the VEGF-R2 pathway may act as an activator in the proliferation of hair follicles cells. In addition, several growth factors, such as insulin-like growth factor-1,29) fibroblast growth factor-5,30) -731) and transforming growth factor-β (TGF-β)32,33) have also been known to positively or negatively regulate hair growth. Especially, TGF-β is the most important regulator in hair growth and the hair cycle.33) Several studies reported that TGF-β could induce the transition of the catagen (regression) phase in the hair cycle,34,35) indicating that the inhibition of the TGF-β pathway is associated with the promotion of hair growth.
Algae are known to improve human health because they contain a lot of bioactive materials including minerals, vitamins, polysaccharides and polyphenols.36) Sargassum muticum, a brown alga, is used as food such as soup and seasoned seaweed in Jeju Island. There are several reports on the biological activities of S. muticum including anti-inflammatory, antioxidant and antiangiogenic effects.36,37) However, the effects of S. muticum and apo-9′-fucoxanthinone, a bioactive compound from S. muticum, on the regulation of hair growth have not yet been performed. Moreover, the action mechanisms of S. muticum and apo-9′-fucoxanthinone on hair growth have not been revealed. This study was carried out to investigate the promotion effect of S. muticum extract and apo-9′-fucoxanthinone as active components for hair growth.
Dimethyl sulfoxide (DMSO), 3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyltetrazolium bromide (MTT), Earle’s balanced salts solution (EBSS), insulin, hydrocortisone, minoxidil, dithiothreitol (DTT), phenylmethylsulfonylfluoride (PMSF), ethylenediaminetetraacetic acid (EDTA), ethylene glycol tetraacetic acid (EGTA), sodium orthovanadate (Na3VO4) and mouse monoclonal anti-β-actin antibody were purchased from Sigma-Aldrich, Inc. (St. Louis, MO, U.S.A.). Aprotinin and leupeptin were purchased from Calbiochem (LaJolla, CA, U.S.A.). Nonidet-P40 (NP-40) was purchased from Roche Diagnostics (Mannheim, Germany). [1,2,6,7-3H] testosterone was purchased from American Radiolabeled Chemicals, Inc. (St. Louis). Testosterone and DHT were purchased from Tokyo Chemical Industry Co., Ltd. (Tokyo, Japan). Williams medium E and L-glutamine were purchased from Gibco BRL (Grand Island, NY, U.S.A.). Penicillin/streptomycin solution and trypsin–EDTA solution were purchased from Invitrogen-Life Technologies (Carlsbad, CA, U.S.A.). Fetal bovine serum (FBS) and Dulbecco’s modification of Eagle’s medium (DMEM) were purchased from Hyclone Inc. (Logan, UT, U.S.A.). Finasteride was purchased from Merck-Sharpe-Dohme (Whitehouse Station, NJ, U.S.A.). Silica gel 60 F254 TLC plate was purchased from Merck (Darmstadt, Germany). ULTIMA GOLD™ Cocktail was purchased from PerkinElmer, Inc. (Waltham, MA, U.S.A.). Five percent Minoxidil (MINOXYL™) was purchased from Hyundai Pharmaceutical Inc. (Chungnam, Korea). Polyvinylidene fluoride (PVDF) membranes and Protein Assay Dye Reagent Concentrate were purchased from Bio-Rad (Hercules, CA, U.S.A.). Mouse-monoclonal anti-VEGF-R2, α-tubulin, rabbit-polyclonal anti-β-catenin and Smad2/3 were purchased from Santa Cruz Biotechnology (Santa Cruz, CA, U.S.A.). Rabbit-polyclonal anti-phospho-(ser552)-β-catenin, phospho-(ser675)-β-catenin, phospho(ser9)-GSK3β and GSK3β were purchased from Cell Signaling Technology (Beverly, MA, U.S.A.). Rabbit-polyclonal anti-Lamin B1 was purchased from Abcam (Cambridge, U.K.). NE-PER nuclear extraction kit was purchased from Pierce (Rockford, IL, U.S.A.). Enhanced chemiluminescence solution (West-zol™ Plus) was purchased from iNtRon Biotechnology (Seoul, Korea).
The Preparation of S. muticum Extract and Isolation of Apo-9′-fucoxanthinoneS. muticum were collected from Udo in Jeju Island, Korea and identified by Dr. Dong Sam Kim. A voucher specimen (A10-0000107) has been deposited at the herbarium of Jeju Biodiversity Research Institute. S. muticum was dried in the well-ventilated shade and then 2 kg of S. muticum was extracted with 80% aqueous methanol (MeOH) for 2 d at room temperature. The 80% MeOH extract was concentrated using a vacuum evaporator and dried to produce powder 200 g. The powdered extract was suspended in water and partitioned into n-hexane, methylene chloride (CH2Cl2), ethyl acetate (EtOAc), n-butanol (BuOH) and aqueous fractions. The CH2Cl2 fraction (5.4 g) was chromatographed over celite with n-hexane, CH2Cl2, EtOAc and MeOH successively (hexane–CH2Cl2 1 : 0, 10 : 1, 5 : 1, 2 : 1, CH2Cl2, EtOAc, MeOH). The (hexane–CH2Cl2 1 : 0) fraction was chromatographed with normal phase silica gel (4×50 cm) and eluted with hexane–EtOAc–MeOH (2 : 1 : 0.1). The fraction yielded apo-9′-fucoxanthinone (1.8 mg). The purified compound was identified by comparing its 1H- and 13C-NMR data with those reported in the literature.38) Apo-9′-fucoxanthinone (Fig. 1) was dissolved in DMSO (Amresco, Solon, OH, U.S.A.).

Male Wistar rats (3 weeks of age), female C57BL/6 mice (6 weeks of age) and male Sprague-Dawley (SD) rats (8 weeks of age) were supplied by Orient Bio (Seongnam, Gyeonggi, Korea). All animals were provided with a standard laboratory diet and water ad libitum. All animals were cared for according to protocols (20110014) approved by the Institutional Animal Care and Use Committee (IACUC) of the Jeju National University.
Isolation and Culture of Rat Vibrissa FolliclesIsolation of rat vibrissa follicles was performed as described previously.39) Briefly, rat vibrissa follicles were harvested from male Wistar rats (23 d of age). First of all, the rats were sacrificed under carbon dioxide (CO2). Both mystacial pads were removed from the rats and placed in a 1 : 1 mixture of solution of EBSS and phosphate buffered saline (PBS) containing 100 units/mL of penicillin and 100 µg/mL of streptomycin. Anagen vibrissa follicles were then carefully dissected using sterile dissecting forceps and blade under a stereomicroscope (Olympus, Tokyo, Japan) from the mystacial pads. The isolated follicles were cultured in 24-well plates containing Williams E medium supplemented with 2 mM L-glutamine, 10 µg/mL insulin, 50 nM hydrocortisone, 100 units/mL penicillin and 100 µg/mL streptomycin at 37°C in an atmosphere comprised of 5% CO2 and 95% air. The isolated follicles were treated with S. muticum extract (0.1, 1, 10 µg/mL) or minoxidil (10 µM) as a positivie control.40) The culture medium containing S. muticum extract or minoxidil was changed every 3 d and the length of the vibrissa follicles was measured using a DP controller (Olympus).
Assay for Prostatic 5α-Reductase ActivityMale SD rats (8-week-old) were sacrificed with CO2. The rat prostates were removed from their capsules, washed with PBS before stored at −80°C. Frozen tissues were placed in a culture dish. The tissues were homogenized with a tacoTM Prep Bead Beater (GeneReach Corp., Taichung, Taiwan) in 5 tissue volumes of homogenizing buffer [20 mM potassium phosphate buffer (pH 6.6), 0.32 M sucrose, 25 µg/mL leupeptin, 25 µg/mL aprotinin, 1 mM DTT and 0.2 mM PMSF]. The homogenates were centrifuged at 700×g for 3 min. The pellets were washed twice with homogenizing buffer. The pellets were suspended in homogenizing buffer and stored at −80°C until use. The suspension (2.5 mg protein/mL as determined by the Bradford dye binding method) was used for 5α-reductase assay. 5α-Reductase activities were analyzed as previously described.41) The reaction were initiated with S. muticum extract (0.1, 1, 10, 100 µg/mL), the solvent fraction of S. muticum extract (0.1, 1, 10 µg/mL) or apo-9′-fucoxanthinone (0.01, 0.1, 1, 10 µM) containing reaction buffer [40 mM potassium phosphate buffer (pH 6.6), 1 mM DTT, 2 mM reduced nicotinamide adenine dinucleotide phosphate, and 120 nCi [1,2,6,7-3H] testosterone]. An independent set of the reaction (n=3) was examined and finasteride (2 nM) was used as a positive control. The mixture was incubated at 37°C for 60 min, and then the reaction was stopped by adding 1 mL of EtOAc. The samples were centrifuged at 1000×g for 5 min. The supernatant was carefully aspirated, dried on the heating plate. The residues were dissolved in 50 µL of EtOAc containing 500 µg/mL of testosterone and 500 µg/mL of DHT, and applied to a silica gel 60 F254 TLC plate. The plate was developed in a 1 : 1 mixture of solution of EtOAc–cyclohexane, and the plate was air dried. The TLC spot of testosterone (T) was visualized under UV light (254 nm). The plate was soaked in 10% H2SO4 solution to confirm the spot of DHT before heating the plate. The plate was clipped off to get area containing androgen, the strips were soaked in 5 mL of ULTIMA GOLD™ Cocktail. Radioactivity was measured by a liquid scintillation counter (Packard Bioscience, Meriden, CT, U.S.A.). The activity of 5α-reductase was expressed as a ratio calculated with the equation: [DHT/(T+DHT)]×100.
Assay for the Proliferation of DPCsRat vibrissa immortalized DPCs42) were donated by the Skin Research Institute, Amore Pacific Corporation R & D Center, Korea. The DPCs were cultured in DMEM supplemented with 10% FBS and penicillin/streptomycin (100 unit/mL, 100 µg/mL, respectively) at 37°C in a humidified atmosphere under 5% CO2. The proliferation of DPCs was evaluated by measuring the metabolic activity using a MTT assay.43) Briefly, DPCs (1.0×104 cells/mL) were seeded into 96-well plates, cultured for 24 h under 1% serum conditions, and treated with vehicle (DMSO diluted 1 : 500 in DMEM containing 1% FBS), or with the S. muticum extract (0.1, 1, 10 µg/mL), the solvent fraction of S. muticum extract (0.1, 1, 10, 40 µg/mL) or apo-9′-fucoxanthinone (0.001, 0.01, 0.1, 1 µM) for 4 d. After incubation, 0.1 mg (50 µL of a 2 mg/mL solution) of MTT was added to each well, and the cells were incubated at 37°C for 4 h. The plates were centrifuged at 350×g for 5 min and the media was carefully aspirated. DMSO 200 µL was added to each well to dissolve the formazan crystals and the absorbance of the plate at 540 nm was read immediately on a microplate reader (Versamax; Molecular Devices, Sunnyvale, CA, U.S.A.). All experiments were performed in triplicate and the mean absorbance values were calculated. The results were expressed as the percentage of vehicle treated groups. Minoxidil was used as a positive control.
Hair Growth Activity in VivoAnagen was induced on the back skin of C57BL/6 mice that were in the telogen phase of the cycle by depilation, as described previously.44) The anagen phase was induced in the back skin of the 7-week-old female C57BL/6 mice by shaving, which led to synchronized development of anagen hair follicles. From the following day (day 1), 0.2 mL of S. muticum EtOAc fraction in 50% ethanol was topically applied every day for 41 d. MINOXYL™ was used as a positive control. The back skin of the mice was then observed and photographed at 1, 14, 20, 33 and 41 d after shaving. For quantitative assessment, dotmatrix planimetry was performed.45)
Western Blot AnalysisThe DPCs (1.0×105 cells/mL in 100 mm dishes) were pre-incubated for 24 h under 1% serum conditions, and the cells were treated with apo-9′-fucoxanthinone (1 µM) and minoxidil (10 µM) as a positive control for 72 h. The cells were washed twice with ice-cold PBS. The cells were lysed in lysis buffer [50 mM Tris–HCl (pH 7.5), 150 mM NaCl, 2 mM EDTA, 1 mM EGTA, 1 mM NaVO3, 10 mM NaF, 1 mM DTT, 1 mM PMSF, 25 µg/mL aprotinin, 25 µg/mL leupeptin and 1% NP-40] to obtain whole cell protein and kept on ice for 30 min. The cell lysates were centrifuged at 21000×g at 4°C for 15 min. Supernatants were stored at −20°C until analysis. On the other hand, to obtain nuclear and cytoplasmic fractions, cells were lysed with NE-PER nuclear and cytoplasmic extraction reagents according to the manufacturer’s protocol. Fractions were stored at −70°C until analysis. Protein concentration was determined by the Bradford method.46) Equal amounts of protein were separated on 8–12% sodium dodecyl sulfate polyacrylamide gel electrophoresis gels. The proteins were then transferred onto PVDF membranes. The membrane was blocked for 1 h with 5% nonfat dried milk in Tween-20-TBS (T-TBS) (50 mM Tris, pH 7.6, 150 mM NaCl, 0.1% Tween-20), each membrane was incubated with specific primary antibodies against phospho(ser552)-β-catenin (1 : 1000), phospho(ser675)-β-catenin (1 : 1000), β-catenin (1 : 1000), phospho(ser9)-GSK3β (1 : 1000), GSK3β (1 : 1000), Smad2/3 (1 : 1000), α-tubulin (1 : 500), Lamin B1 (1 : 3000), VEGF-R2 (1 : 500) and β-actin (1 : 5000) at 4°C overnight. The membrane was incubated with a secondary horseradish peroxidase antibody (1 : 5000) at room temperature for 1 h. The membrane was exposed on X-ray film, and protein bands were detected using West-zol™ Plus. Band intensities were quantified with NIH Image software (http://rsb.info.nih.gov/ij/).
Statistical AnalysisAll results were expressed as the mean±standard deviation (S.D.) of at least three independent experiments. Student’s t-test was used to determine the statistical significance (p-value <0.05) of differences between values for the various experimental and control groups. SigmaStat (Systat Software Inc., San Jose, CA, U.S.A.) was used for statistical analysis.
In order to examine whether S. muticum extract could promote hair growth, we investigated the effect of the extract using organ culture of rat vibrissa follicles. Culturing rat vibrissa follicles is known to be an effective method to examine the possibility of hair growth, and had been tested in several studies.34,39,47) When rat vibrissa follicles were treated with various concentrations of S. muticum extract for 21 d, the hair-fiber length of vibrissa follicles treated with S. muticum extract (0.1, 1 µg/mL) increased significantly compared with the vehicle-treated control (134.4, 133.0%, respectively: Fig. 1). Furthermore, 0.1 and 1 µg/mL S. muticum extract affected a greater increase than minoxidil 10 µM, the positive control, whereas 10 µg/mL of S. muticum extract did not affect the hair-fiber length compared with the vehicle-treated control (Fig. 2).
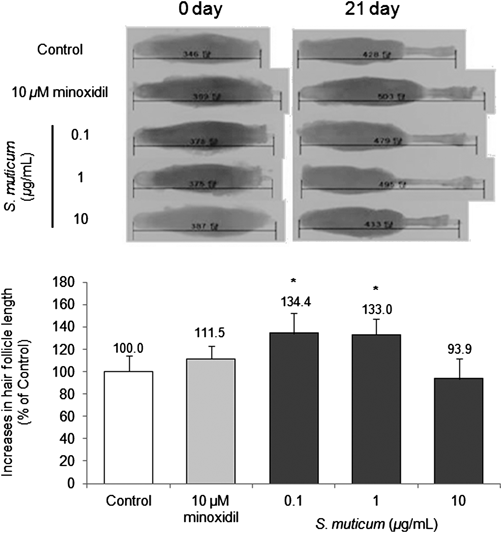
Vibrissa follicles were treated with vehicle (DMSO), S. muticum extract (0.1, 1, 10 µg/mL) for 21 d. Minoxidil was used as a positive control. All experiments were performed in triplicate. The difference in the length of vibrissa follicles in the vehicle-treated control group on day 21 was taken to be 100%. Data are presented as a percentage of the length of the sample-treated follicles based on the mean length of the control follicles±S.D. * p<0.05 vs. control.
The inhibition of 5α-reductase contributes to the improvement of hair loss in androgenetic alopecia patients.9) To determine whether the hair growth effect of S. muticum extract could be mediated by the inhibition of 5α-reductase, we examined the 5α-reductase activity with crude enzymes from rat prostate. As shown in Fig. 3A, S. muticum extract inhibited 5α-reductase activities by 12.4, 18.8, 15.8 and 14.6% at the concentrations of 0.1, 1, 10 and 100 µg/mL. Additionally, several solvent fractions of the S. muticum extract markedly inhibited 5α-reductase activities by 44.8–82.6% (Fig. 3B). Finasteride, a positive control, inhibited 5α-reductase activity by above 85.9% at 2 nM concentration. The results suggest that S. muticum extract and its solvent fractions could have the potential for the treatment of androgenetic alopecia via 5α-reductase inhibition.

(A) S. muticum extract-treated group (B) several solvent fractions-treated group. Assay of 5α-reductase inhibition was performed using a crude extract of rat prostate as described in Materials and Methods. The conversion rate of testosterone (T) to dihydrotestosterone (DHT) was calculated with the equation: [DHT/(T+DHT)]×100. Inhibition rate (%) was expressed as a percentage of reduced conversion rate compared to the control. The inhibition activity of the control group was regarded as 0% (not shown). Treatment with finasteride served as a positive control. Data are presented as the mean±S.D. of three independent experiments. * p<0.05, ** p<0.01, *** p<0.001 vs. control.
Previous reports have shown that the size of the DPCs correlated well with hair growth, and the number of DPCs increased in the growing phase of the hair cycle.16,48) To determine whether S. muticum extract induced a hair growing effect by mediating DPC proliferation, we examined the proliferation of DPCs using the MTT assay. The S. muticum extract increased the proliferation of DPCs by 135.7% at the concentration of 10 µg/mL compared with the vehicle-treated control (Fig. 4A). Moreover, the proliferation of DPCs was increased by treatment with solvent fractions of S. muticum extract (Fig. 4B). Among the solvent fractions, 40 µg/mL of the EtOAc fraction and 1 µg/mL of the hexane fraction significantly increased the proliferation of DPCs by 135.6 and 125.7%, respectively. However, the hexane fraction (10, 40 µg/mL) decreased the proliferation of DPCs (Fig. 4B). Minoxidil, a positive control, enhanced the proliferation of DPCs by 113.4% at the concentration of 10 µM compared with the vehicle-treated control. These results suggest that S. muticum extract and its EtOAc fraction could have hair growth effects via the proliferation of DPCs.

DPCs (1.0×104 cells/mL) were plated in 96 well plates. DPCs were treated with various concentrations of S. muticum extract (A) and its solvent fractions (B), as indicated for 4 d. Cell proliferation was measured by MTT assay. All experiments were performed in triplicate. Minoxidil (10 µM) was used as a positive control. Data are presented as the mean±S.D. * p<0.05, ** p<0.01, *** p<0.001 vs. control.
To explore whether hair cycle progression was induced by S. muticum, we used C57BL/6 mice, since the dorsal hair is known to have a time-synchronized hair growth cycle.44) As the hair cycle progressed, the pink (telogen) color of the skin turns to black (anagen) in C57BL/6 mice. Among S. muticum extract and its solvent fractions, the EtOAc fraction appeared to be the most effective treatment option on the DPCs proliferation and 5α-reductase inhibition. Especially, considering the DPC proliferation effect, the EtOAc fraction of S. muticum extract was used at 1, 10, and 40 µg/mL in the in vivo experiment. On the 33rd day, the treatment group that received the 40 µg/mL EtOAc fraction of S. muticum extract had gray/black skin, while the control group was visibly less pigmented (Fig. 5A). When the area with black skin was analyzed with dotmatrix planimetry, the area in the treatment group that received the EtOAc fraction of S. muticum extract was significantly larger than that of the control group on the 33rd day after depilation (Fig. 5B). The group treated with MINOXYL™, a positive control, exhibited gray skin starting from 14 d after depilation. These results indicate that the EtOAc fraction of S. muticum extract could effectively promote telogen-to-anagen transition in the in vivo model.

After shaving, the back skins of C57BL/6 mice were treated with the EtOAc fraction of S. muticum, vehicle and 5% minoxidil (MINOXYL™) every day for 41 d. (A) The back skins were photographed at 1, 14, 20, 33 and 41 d after depilation; (B) On day 33, to perform a quantitative comparison of anagen induction, dotmatrix planimetry was performed. A transparent foil was placed on the photo of a mouse to mark the areas that were in different stages (pink=telogen; anagen=black). Afterward, a dotmatrix (sheet with a uniform defined dot pattern) was placed under the marked foil to calculate the percentages of the regions of interest by counting the dots. The percentage of anagen induction was calculated with the equation [(black skin/total skin)×100]. Data are presented as the mean±S.D. (n=6). * p<0.05, *** p<0.001 vs. vehicle-treated control.
To identify the active components in the EtOAc fraction of S. muticum extract responsible for promoting hair growth, apo-9′-fucoxanthinone, a known bio-active component of S. muticum extract, was examined. First, to evaluate whether apo-9′-fucoxanthinone could inhibit 5α-reductase activity, we examined the effect of apo-9′-fucoxanthinone using a crude 5α-reductase from rat prostate. Apo-9′-fucoxanthinone inhibited 5α-reductase activity by 19.3, 14.5, 26.7 and 20.9% at concentrations of 0.01, 0.1, 1 and 10 µM, respectively (Fig. 6A). However, there were no differences in the apo-9′-fucoxanthinone-treated groups, because they showed a slightly higher standard deviation (Fig. 6A). The results suggest that apo-9′-fucoxanthinone could have the potential for the treatment of androgenetic alopecia via 5α-reductase inhibition. On the other hand, we also examined whether apo-9′-fucoxanthinone can promote the proliferation of DPCs. Apo-9′-fucoxanthinone increased the proliferation of DPCs by 121.7% at the concentration of 1 µM compared with the vehicle-treated control (Fig. 6B). The results suggest that apo-9′-fucoxanthinone might have hair growth effects via the proliferation of DPCs.

(A) Assay of 5α-reductase inhibition was performed using a crude extract of rat prostate as described in Materials and Methods. The conversion rate of testosterone (T) to dihydrotestosterone (DHT) was calculated by the equation [DHT/(T+DHT)]×100. Inhibition rate (%) was expressed as a percentage of reduced conversion rate compared to the control. The inhibition activity of the control group was regarded as 0% (not shown). Treatment with finasteride served as a positive control; (B) DPCs (1.0×104 cells/mL) were plated in 96 well plates. DPCs were treated with various concentration of apo-9′-fucoxanthinone, as indicated for 4 d. Cell proliferation was measured by MTT assay. Minoxidil (10 µM) was used as a positive control. Data are presented as the mean±S.D. of three independent experiments. * p<0.05, ** p<0.01, *** p<0.001 vs. control.
The Wnt/β-catenin pathway is known to be one of the regulators of cell proliferation.13,22) To confirm the hair growth promoting effect of apo-9′-fucoxanthinone in relation to the Wnt/β-catenin pathway, we examined the change in the level of phospho-GSK-3β, GSK-3β, phospho(ser552)-β-catenin and phospho(ser675)-β-catenin following apo-9′-fucoxanthinone treatment for 72 h in DPCs. Treatment with apo-9′-fucoxanthinone resulted in an increase in the level of phospho(ser9)-GSK-3β and phospho(ser552)-β-catenin compared to control cells (Fig. 7). Similar results were observed after minoxidil (10 µM) treatment, where the level of phospho(ser9)-GSK-3β was higher than earlier (data not shown). These results suggest that apo-9′-fucoxanthinone could increase the proliferation of DPCs through regulation of the Wnt/β-catenin pathway.

DPCs (1.0×105 cells/mL in 100 mm dishes) were pre-incubated for 24 h under 1% serum conditions, the cells were treated with apo-9′-fucoxanthinone (1 µM) and minoxidil (10 µM) as a positive control for 72 h. (A) Whole cell lysates from DPCs were analyzed by immunoblotting to determine the levels of phospho-GSK3β, GSK3β, phospho(ser552)-β-catenin, phospho(ser675) β-catenin and β-catenin. (B) Quantification of protein levels by immunoblotting in whole cell lysates from DPCs after treatment. The data are presented as the mean±S.D. from three independent experiments. * p<0.05 vs. control.
The level of VEGF-R2 increased in response to VEGF treatment and has been implicated in the proliferation of DPCs and ORS.27,28) We thus investigated whether apo-9′-fucoxanthinone could affect the level of VEGF-R2 in DPCs. When DPCs were treated with apo-9′-fucoxanthinone for 72 h, apo-9′-fucoxanthinone significantly increased the level of VEGF-R2 (Fig. 8). After treatment with minoxidil (10 µM), the level of VEGF-R2 also increased (Fig. 8). These results suggest that apo-9′-fucoxanthinone can increase the proliferation of DPCs through a VEGF-R2 pathway.
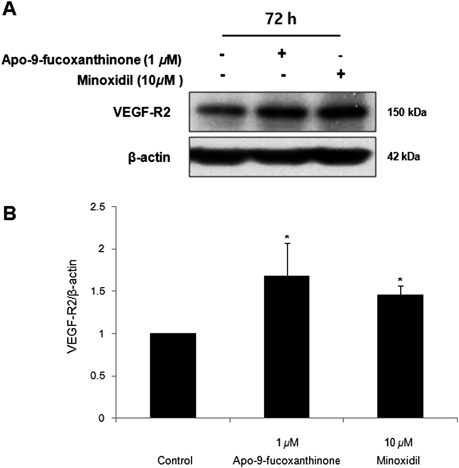
DPCs (1.0×105 cells/mL in 100 mm dishes) were pre-incubated for 24 h under 1% serum conditions, the cells were treated with apo-9′-fucoxanthinone (1 µM) and minoxidil (10 µM) as a positive control for 72 h. (A) Whole cell lysates from DPCs were analyzed by immunoblotting to determine the level of VEGF-R2. (B) Quantification of protein levels by immunoblotting in whole cell lysates from DPCs after treatment. The data are presented as the mean±S.D. from three independent experiments. * p<0.05 vs. control.
TGF-β has been implicated in the regulation of catagen transition and cell proliferation.32,33,49) Binding of TGF-β to receptors can increase the nuclear translocation of Smad2/3, a mediator of the TGF-β pathway.50) To explore the TGF-β pathway related to apo-9′-fucoxanthinone-induced proliferation of DPCs, we evaluated the nuclear transition of Smad2/3 using an immunoblotting assay. As shown in Fig. 9, apo-9′-fucoxanthinone did not affect the level of nuclear and/or cytoplasmic Smad2/3. These results indicate that apo-9′-fucoxanthinone promotes the proliferation of DPCs through a TGF-β-independent pathway.

DPCs (1.0×105 cells/mL in 100 mm dishes) were pre-incubated for 24 h under 1% serum conditions, the cells were treated with apo-9′-fucoxanthinone (1 µM) and minoxidil (10 µM) as a positive control for 72 h. (A) Nuclear and cytoplasmic fractions were prepared as described in Materials and Methods and the level of Smad2/3 was examined by immunoblotting. (B) Quantification of protein levels by immunoblotting in nuclear and cycoplasmic fractions from DPCs after treatment. The data are presented as the mean±S.D. from three independent experiments. * p<0.05 vs. control.
The present study describes the hair-growth promoting effects of S. muticum extract and apo-9′-fucoxanthinone, a principal component of S. muticum extract, via the proliferation of DPCs and by 5α-reductase inhibition. To the best of our knowledge, this study is the first to demonstrate that apo-9′-fucoxanthinone increases DPC proliferation via the activation of Wnt/β-catenin signaling and the VEGF-R2 pathway.
To identify new hair-growing agents, cultures of hair follicles from several species such as human,11,32) rat,39) sheep51) and mouse40,52) were developed. The hair follicle culture method has several advantages in hair research, including the measure of hair-fiber elongation and maintenance of normal architecture.39,40) In this study, we isolated rat vibrissa follicles from 23-d old rats and found that the S. muticum extract significantly promoted hair-fiber length in cultured rat vibrissa follicles. By the way, we couldn’t find the dose-dependency of the S. muticum extract on the elongation of hair-fiber length. We can consider 3 possible reasons as follows; 1) The S. muticum extract contains lots of compounds including apo-9′-fucoxanthinone; 2) In hair follicle, there are various types of cells such as bulge stem cells, outer root epithelial cells, inner root epithelial cells, melanocytes and keratinocytes, including DPCs. The effect of S. muticum extract on hair fiber elongation includes that on DPCs and those on the other cells of hair follicle as well as interactions between various cells; 3) In assay for hair fiber elongation, whenever the culture medium was changed every 3 d, S. muticum extract was also added. For 21 d, S. muticum extract and its metabolite can be accumulated in hair follicle.
Androgenetic alopecia is known as the most common cause of hair loss.14) 5α-Reductase converts testosterone to dihydrotestosterone, which is more potent than testosterone and can trigger the progression of androgenetic alopecia.9,14) In this study, the inhibition of 5α-reductase activity was observed in groups treated with S. muticum extract, solvent fractions of S. muticum extract and apo-9′-fucoxanthinone. The results indicate that S. muticum extract and apo-9′-fucoxanthinone could have the potential for the treatment of androgenetic alopecia via 5α-reductase inhibition. By the way, in comparison, the effect of the EtOAc fraction of S. muticum extract on the 5α-reductase inhibition was much greater than that of apo-9′-fucoxanthinone. To explain the effect of the EtOAc fraction of S. muticum extract on the 5α-reductase inhibition, we need to find the other components except apo-9′-fucoxanthinone from the EtOAc fraction of S. muticum extract.
DPCs reside at the base of the hair follicle and are known as regulators of hair growth.7,16) Mesenchymal DPCs-epithelial cell interactions continue throughout an organism’s lifetime and regulate hair growth as well as the hair cycle.7,16,44) In fact, many studies have shown that natural products induce hair growth through DPC proliferation.34,53,54) S. muticum extract, solvent fractions of S. muticum extract and apo-9′-fucoxanthinone significantly promoted the proliferation of DPCs in this study. These results indicate that S. muticum extract and apo-9′-fucoxanthinone might have hair-growth promoting effects via the proliferation of DPCs.
Among S. muticum extract and its solvent fractions, the EtOAc fraction appeared to be the most effective treatment option in terms of DPCs proliferation and 5α-reductase inhibition, indicating that the EtOAc fraction of S. muticum extract could have some potential in an in vivo experimental model. In subsequent experiments to examine the in vivo effect, the EtOAc fraction of S. muticum extract was used and the induction of the anagen phase was investigated. The skin color of C57BL/6 mice changed from pink to black following treatment with the EtOAc fraction of S. muticum extract in a dose-dependent manner.
To elucidate the action mechanism of apo-9′-fucoxanthinone, the main component of S. muticum extract, on the induction of hair growth, its regulation on the Wnt/β-catenin signaling, the VEGF-R2 pathway and the TGF-β pathway was investigated in DPCs. The Wnt/β-catenin pathway plays an important role in the regulation of the hair cycle, hair growth and cell proliferation.13,19,21,22) Several natural products are reported to increase hair growth through the activation of Wnt/β-catenin proteins in hair follicles and DPCs.53,54) Previous study showed that minoxidil could also induce the hair growth through the activation of β-catenin pathway.13) In the study, when DPCs were treated with apo-9′-fucoxanthinone, the activation of Wnt/β-catenin signaling proteins such as phospho(ser9)-GSK-3β and phospho(ser552)-β-catenin were observed. These findings suggest that activation of the Wnt/β-catenin pathway by apo-9′-fucoxanthinone may increase hair growth via DPC proliferation. VEGF-R2 is expressed by different cells, including endothelial cells,55) keratinocytes,56) cancer cells26) and hair follicle cells.27,28,57) VEGF has been known to increase the cell proliferation of DPCs and ORS, which was attenuated by VEGF-R2 neutralizing antibody. These reports suggest that activation of the VEGF-R2 pathway is required for the proliferation of DPCs. In this study, apo-9′-fucoxanthinone and minoxidil increased the proliferation of DPCs and the level of VEGF-R2, which means that the activation of the VEGF-R2 pathway is involved in apo-9′-fucoxanthinone-induced proliferation of DPCs. TGF-β is known as an important factor in the regulation of cell proliferation, hair cycle and hair growth.33,49) It has been reported that the repression of TGF-β delays the phase transition from angen to catagen,32,34) which suggests that inhibition of TGF-β signaling may increase hair growth. However, apo-9′-fucoxanthinone had no effect on the nuclear translocation of Smad2/3. This result indicates that apo-9′-fucoxanthinone-mediated proliferation of DPCs could be associated with a TGF-β-independent pathway.
In conclusion, we demonstrated that extracts of the edible S. muticum could promote hair growth in vitro and in vivo through proliferation of DPCs and inhibition of 5α-reductase activity. Moreover, apo-9′-fucoxanthinone, a principal component of S. muticum, induced the proliferation of DPCs followed by the activation of the Wnt/β-catenin pathway and the VEGF-R2 pathway, as well as 5α-reductase inhibition. Our results indicate that apo-9′-fucoxanthinone can show both beneficial characteristics of minoxidil and finateride in the treatment of alopecia. The results suggest that S. muticum extract and apo-9′-fucoxanthinone might have potentials for the development of hair growing agents.
This work was supported by the National Research Foundation of Korea Grant funded by the Korean Government (MEST) (NRF-C1ABA001-2011-0021037).
The authors declare no conflict of interest.