Sinus of Valsalva aneurysm (SVA) is a congenital or acquired enlargement of the aortic root area between the aortic valve annulus and sinotubular ridge that is present in approximately 0.09% of the general population.1
In addition, ruptured SVA (RSVA) is rare, comprising only 0.3–3.56% of all congenital heart disease, and usually occurs in adolescence to early adulthood.2,3
It is a thin-walled outpouching of the sinus of Valsalva and may be tubular or saccular in shape. The right sinus of Valsalva is most commonly involved (76.8%), followed by the non-coronary sinus, and, least commonly, the left coronary sinus (3%).2
The right sinus of Valsalva usually ruptures into the right heart chambers.4
Rupture into the left ventricle (LV), however, is extremely rare. SVA frequently presents as an incidental finding on cardiac imaging. Echocardiogram is the standard imaging technique for diagnosis of SVA. We report an unusual case of right SVA involving rupture into the LV. Accurate diagnosis before surgery was difficult.
A 53-year-old man with no medical history presented to the local doctor with a 1-week history of worsening dyspnea at rest (New York Heart Association class III–IV). On, admission, blood pressure was 118/52 mmHg, pulse rate was 78/min, body temperature was 36.7℃ and respiratory rate was 18/min. A systolic ejection murmur, grade IV/VI, and diastolic murmur, grade III/VI, was heard at the second and third intercostal spaces along the left sternal border. The rest of the physical examination was normal. Plain chest X-ray showed cardiac dilatation. Electrocardiography indicated normal sinus rhythm. Besides elevation of B-type natriuretic peptide (506 pg/mL), laboratory findings were unremarkable. On transthoracic echocardiography (TTE), LV ejection fraction was 62% with normal wall motion. In addition, TTE showed a ruptured aneurysm of the right coronary cusp or right SVA, and non-ruptured aneurysm around the right coronary cusp protruding into the LV outflow tract with severe aortic regurgitation. Color Doppler echocardiography showed turbulent flow through the ruptured aneurysm (Figure 1, Upper panels). Transesophageal echocardiography (Figure 1, Lower panels) and 64-slice multidetector computed tomography also showed the same thing. Before surgery, it was difficult to determine whether this was right aortic valve aneurysm or RSVA. After control of heart failure, the patient underwent surgery including patch closure of rupture aneurysm, aortic valve replacement, mitral annuloplasty and tricuspid annuloplasty. During surgery the aneurysms were seen to originate not from the right coronary cusp but from the sinus of Valsalva. In this case, the right SVA was pouch shaped (length, 1.6 cm) and it had protruded and ruptured into the LV (Figure 2). In addition, there was no ventricular septal defect. The postoperative course was uneventful. On follow-up echocardiography at 1 year after surgery, both the LV and the prosthetic aortic valve function were normal.
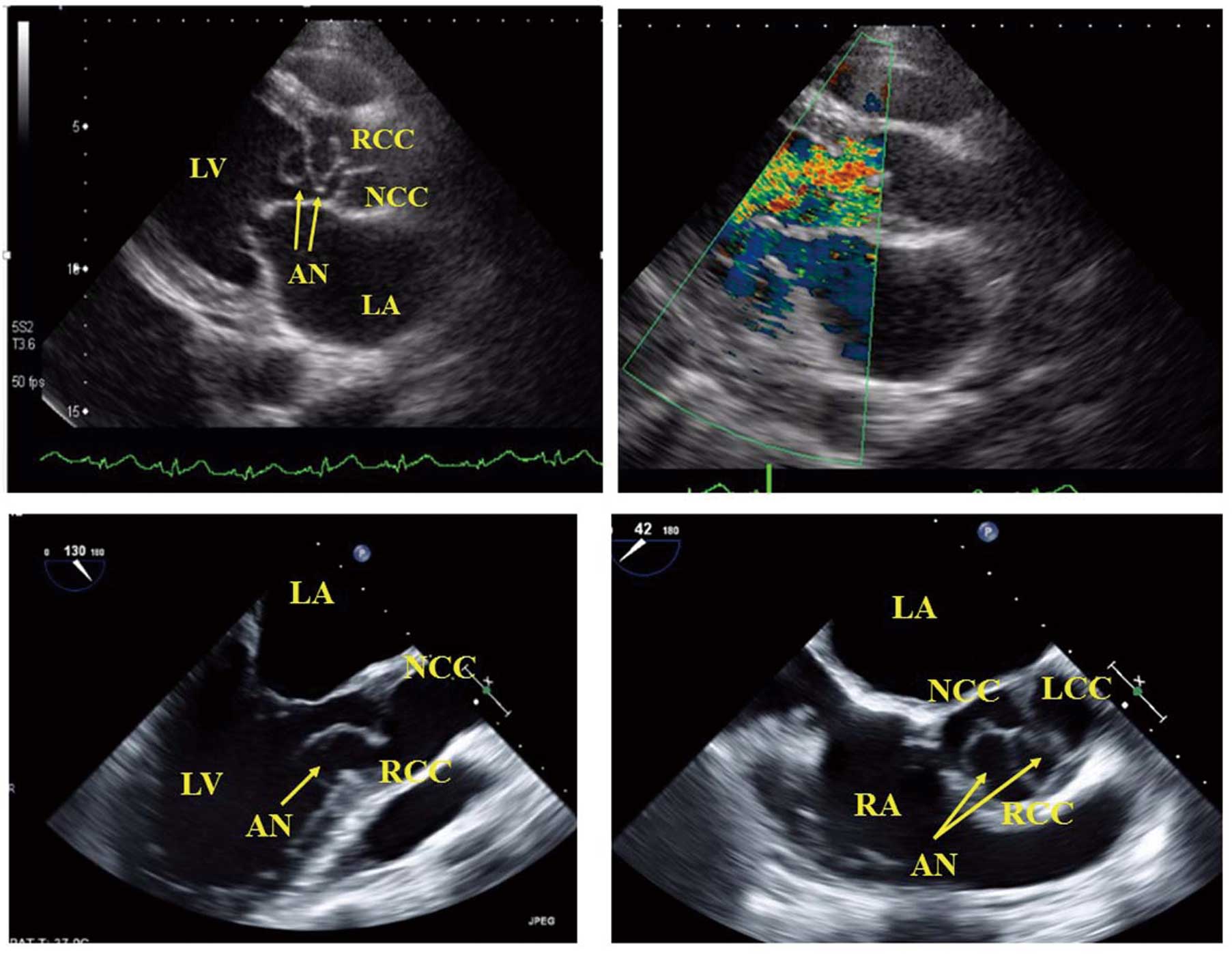
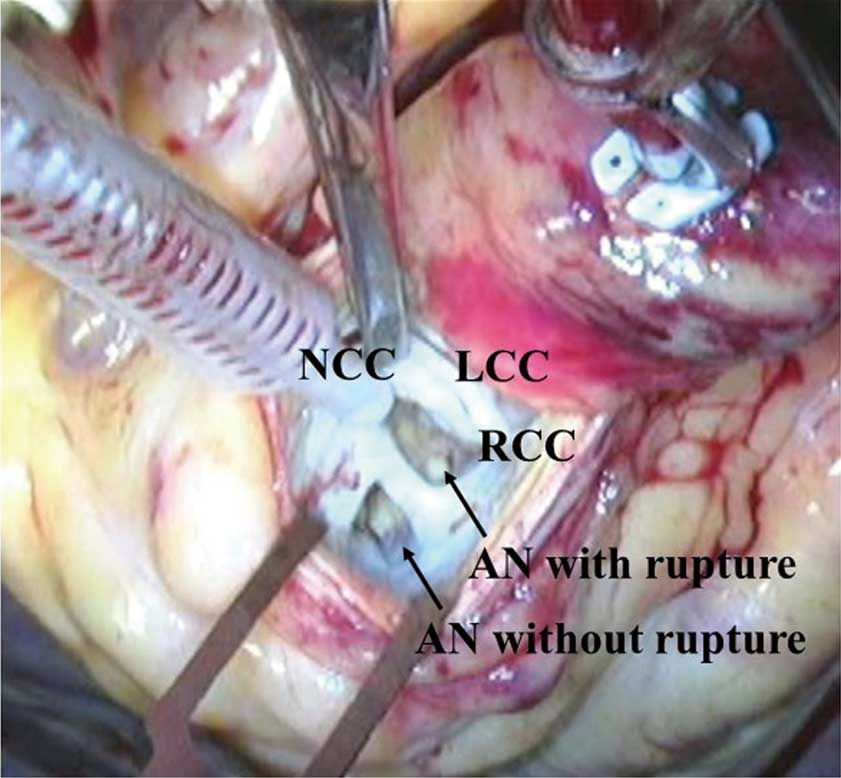
SVA is a rare cardiac malformation that may be congenital or acquired. In this case, RSVA was difficult to diagnose before surgery. First, it was thought to be a possible rupture of the right aortic valve aneurysm. Aortic aneurysm is also rare and is usually correlated with (asymptomatic) endocarditis.5,6
We also considered the possibility of RSVA, because RSVA usually originates from the right SVA, but right SVA almost always ruptures into the right heart chambers.7
We were unable to visualize the connection between the LV and right SVA on imaging. In addition, the patient had no evidence of connective tissue disorder such as Marfan or Ehlers-Danlos syndrome, and had no other cardiac abnormalities. SVA frequently presents with ventricular septal defect (30–60%), aortic insufficiency (20–30%), bicuspid aortic valve (10%) and coronary anomalies.8
Therefore, it was difficult to determine whether the aneurysm originated at the right coronary cusp or was an SVA. As a result, in this case, the rupture site was communicating with the aortic root and LV like a “wind sock”.
RSVA is rare but is usually diagnosed on echocardiography. In the present case, however, right SVA ruptured into the LV, making accurate preoperative diagnosis extremely difficult. We should keep in mind that right SVA can, albeit very rarely, rupture into the LV.
References
- 1.
Feldman DN, Roman MJ. Aneurysms of the sinuses of Valsalva. Cardiology 2006; 106: 73–81.
- 2.
Chu SH, Hung CR, How SS, Chang H, Wang SS, Tsai CH, et al. Ruptured aneurysms of the sinus of Valsalva in oriental patients. J Thorac Cardiovasc Surg 1990; 99: 288–298.
- 3.
Edwards JE, Burchell HB. The pathological anatomy of deficiencies between the aortic root and the heart, including aortic sinus aneurysms. Thorax 1957; 12: 125–139.
- 4.
Kim HR, Kim SJ, Lim KH, Kim JM, Lee JH, Kim YG, et al. A case of perimembranous ventricular septal defect associated with sinus of Valsalva aneurysm mimicking membranous septal aneurysm. J Cardiovasc Ultrasound 2015; 23: 113–117.
- 5.
Morimoto K, Matsuzaki M, Anno Y, Tanimoto Y, Kobayashi Y, Takashiba K, et al. Echocardiographic assessment of aneurysm of the aortic valve caused by infective endocarditis: A case report. Jpn Circ J 1989; 53: 819–824.
- 6.
Qian H, Liu Y, Li Y, Zhang E. Aortic valve aneurysm involved in interventricular septum. Anatol J Cardiol 2016; 16: E2–E3.
- 7.
Kucukoglu S, Ural E, Mutlu H, Ural D, Sonmez B, Uner S. Ruptured aneurysm of the sinus of Valsalva into the left ventricle: A case report and review of the literature. J Am Soc Echocardiogr 1997; 10: 862–865.
- 8.
Liu YL, Liu AJ, Ling F, Wang D, Zhu YB, Wang Q, et al. Risk factors for preoperative and postoperative progression of aortic regurgitation in congenital ruptured sinus of Valsalva aneurysm. Ann Thorac Surg 2011; 91: 542–548.



