Article ID: CJ-17-0041
Article ID: CJ-17-0041
Background: Difficulty in detecting and measuring Achilles tendon (AT) xanthomas may be responsible for underdiagnosis of familial hypercholesterolemia (FH). We aimed to determine a cutoff value for AT thickness (AT-T) using ultrasonography to diagnose FH, and to investigate the relationship between AT-T and atherosclerosis.
Methods and Results: Ultrasonographic AT-T and carotid intima-media thickness (IMT) were evaluated in 130 genetically diagnosed FH patients and 155 non-FH patients. The outline and internal properties of the AT could be clearly determined using ultrasonography, and a good correlation in AT-T was observed between ultrasonography and the conventional method of X-ray radiography (r=0.924, P<0.001). Cutoff values for the diagnosis of FH derived from receiver-operating curves were 5.8 mm (sensitivity 71%, specificity 78%) in men, and 5.5 mm (sensitivity 80%, specificity 81%) in women. Importantly, increased AT-T was positively associated with carotid IMT only in the FH group. Additionally, increased AT-T was associated with the presence of coronary artery disease in a logistic regression analysis adjusted for traditional cardiovascular risk factors.
Conclusions: This is the first study to determine a cutoff value for AT-T based on ultrasonography for the diagnosis of FH in Japanese subjects. Clearer detection and easier measurement of AT-T using ultrasonography would encourage clinicians to diagnose FH more actively, and could solve the problem of underdiagnosis of FH.
Familial hypercholesterolemia (FH) is an autosomal dominantly inherited disease characterized by markedly increased serum low-density lipoprotein-cholesterol (LDL-C) levels, formation of xanthomas, including those causing thickening of the Achilles tendon (AT), and early-onset coronary artery disease (CAD),1 FH is reportedly common in patients hospitalized with premature acute coronary syndrome,2 suggesting the importance of early diagnosis and early treatment of this disease. However, the diagnosis rate of FH in Japan is extremely low despite the high frequency of heterozygous FH (1 of 200–500).3 Therefore, improving the diagnosis rate of FH is a clinically important issue to be addressed.
In Japan, AT thickness (AT-T) is an important diagnostic criterion in the guidelines for the management of FH, the intention of which is to achieve better control and prevent CAD.4,5 Soft X-ray radiography has been used for measurement of AT-T, but although the exposure to radiation is quite low, and imaging conditions are described in the guideline, there are some clinicians who hesitate to use X-ray radiography to diagnose FH because of the imaging conditions and radiation exposure. Additionally, the width of the AT is measured through palpation, whereas the thickness is measured using soft X-rays, leading to the possibility of some discrepancy in diagnosis. Furthermore, the border between the skin and the tendon is sometimes unclear in soft X-ray radiography.
Ultrasonography (US) has recently become a widely used imaging modality for the evaluation of AT-T in other countries.6 Therefore, in the present study we focused on US, a noninvasive and simple procedure that allows not only thickness and width to be assessed but also area and internal properties. Several studies of AT xanthoma detection and AT-T measurement using US have been conducted,7–9 but there have been problems with previous studies, including small sample size and the lack of a cutoff value to distinguish FH. A few studies have set cutoff values,10,11 but they are not from Japan. Accordingly, we aimed to set a cutoff value for AT-T measured by US in the diagnosis of FH in Japan in a relatively large-scale cross-sectional study.
We12,13 and other researchers14 have previously reported that AT-T measured by soft X-ray radiography is a risk factor for CAD. Importantly, Tada et al reported that even in FH subjects without mutation, those with clinical signs of FH (tendon xanthomas and/or family history) had 4.6-fold higher odds of developing CAD compared with those without clinical signs after adjusting for traditional risk factors including high LDL-C levels.15 Thus, it is clinically important to test whether AT-T measured by US is also a useful marker for atherosclerosis. Therefore, we additionally investigated the associations between AT-T and the prevalence of CAD as well as carotid intima-media thickness (IMT).
We enrolled a total of 285 subjects (130 FH patients and 155 non-FH patients), consisting of 114 FH patients and 125 non-FH patients visiting the National Cerebral and Cardiovascular Center Hospital (NCVC), and 16 FH patients and 30 non-FH patients visiting the Kenporen Osaka Central Hospital, during the period from October 2013 to August 2016 for both hospitals. Patients aged less than 20 years, untreated patients, initial-visit patients, homozygous FH patients, and patients with a history of AT injury or damage were excluded. In accordance with the guidelines of the Japan Atherosclerosis Society, patients were diagnosed as FH when at least 2 of the following criteria were met, excluding cases of secondary hyperlipidemia: (1) LDL-C before treatment: ≥180 mg/dL; (2) presence of tendon xanthoma (AT-T ≥9 mm on X-ray) or nodular xanthoma on the skin; or (3) family history of FH or premature CAD within 2nd-degree relatives.
This study was performed in conformity with the Helsinki Declaration and approved by the Ethical Committee of the NCVC (Approval No. M25-112). Written informed consent was given by all subjects following explanation of the purpose, method, and risks of the study.
Measurement of Achilles TendonTo perform ultrasonographic AT measurements, ankles were flexed at 90° with the upper abdomen positioned on the bed; the image of the AT was enlarged as much as possible at the horizontal section of the AT, and its thickness (AT-T), width (AT-W), and area (AT-A) were measured (Figure 1A). Measurements were made at the point of the AT with the greatest thickness. The mean of the values for the left and right AT was used as the final measurement. The aspect ratio (T/W) was defined as AT-T divided by AT-W. At the time of obtaining measurements, the AT was examined for abnormalities such as xanthoma or calcification. In order to obtain an overall image of the AT without the ultrasound probe separating from the skin when flexing the ankle, a SONAGERU Ultrasound Gel Pad 10 mm (TAKIRON, Osaka, Japan) was used between the ultrasound probe and the ankle during the examination. The testing equipment and ultrasound probes comprised an α10 device (Aloka, Tokyo, Japan), a high-frequency linear type probe with a central frequency of 10 MHz, and a LOGIQ S8 device (GE Healthcare Japan, Tokyo, Japan), and a high-frequency linear type probe with a central frequency of 9 MHz. Assessments were carried out by 3 ultrasonographists certified by the Japan Society of Ultrasonics in Medicine. In order to confirm inter-rater reliability between the 2 ultrasonographists of the NCVC, who performed measurements for approximately 90% of the subjects, the assessments for 10 subjects by both of them were compared, which produced a correlation coefficient of 0.88, indicating that their assessments were well correlated. Additionally, to investigate intra-examiner reliability, 2 ultrasonographers measured AT-T 10 times in 3 patients each, and the intra-assay variation was from 0.06 to 0.08 for both of them. Measurement of the AT-T using soft X-ray radiography (AT-X) was carried out by the conventional method (Figure 1B).12
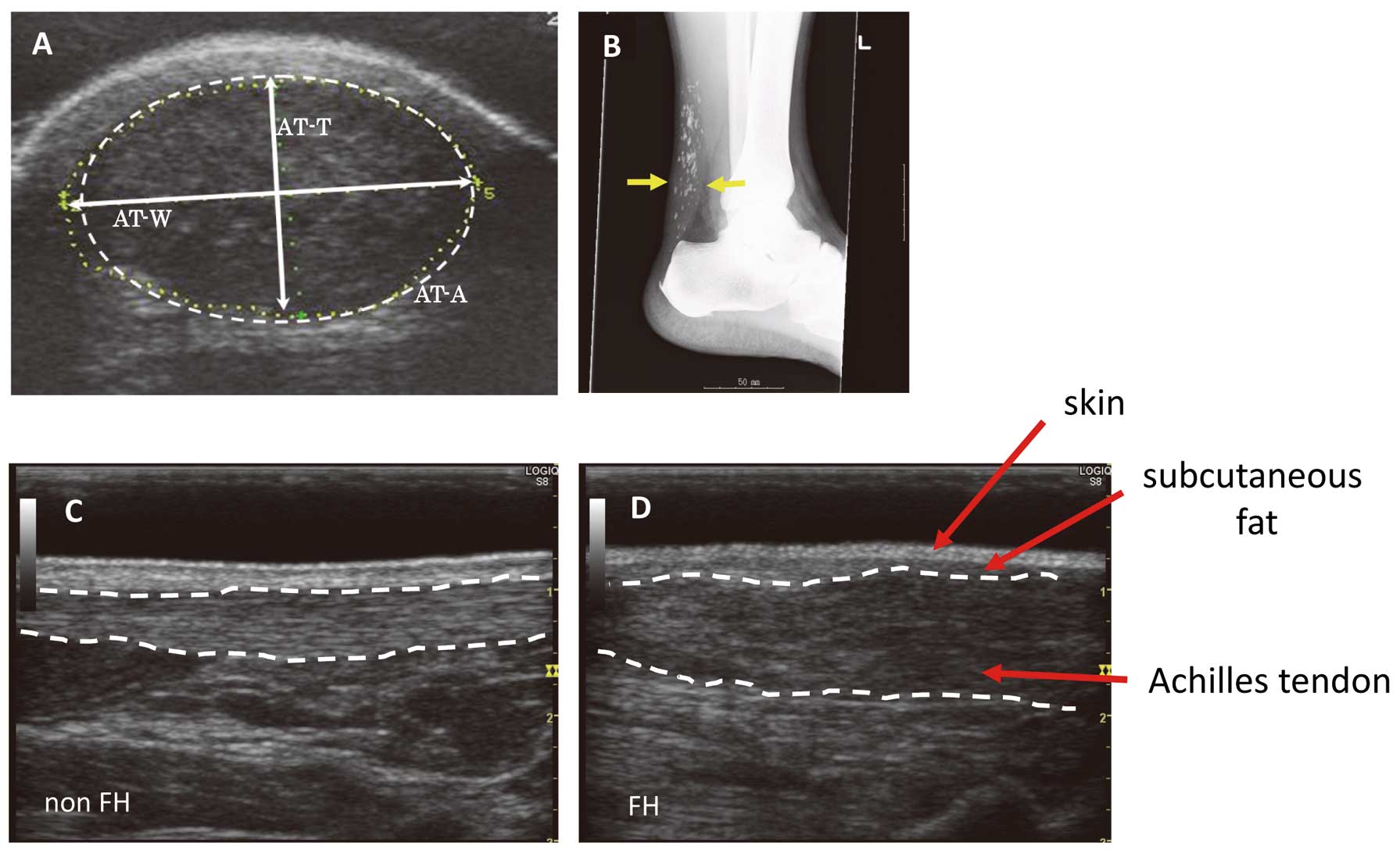
Sonographic and radiographic appearance of the Achilles tendon (AT). (A) 77-year-old man with FH: horizontal sonogram. (B) 59-year-old man with FH: sagittal radiogram. (C) 45-year-old normal male subject: sagittal sonogram. (D) 77-year-old man with FH: sagittal sonogram. AT-T, Achilles tendon thickness; AT-W, Achilles tendon width; AT-A, Achilles tendon area; FH, familial hypercholesterolemia.
At the time of measuring the AT, the IMT was also measured. The location of the maximum IMT (max-C) within the total carotid artery in the supine position was extracted and brought to the center of the screen, and then 3 points, the point itself and 2 points at 1 cm on both sides, were measured, defining the mean value of the 3 points as the mean IMT (mean-C). In the event max-C could not be extracted and brought to the center of the screen, 3 points in total at 1 cm intervals from max-C were measured to calculate the mean-C. The thickest IMT within the bifurcation area to the inner carotid artery was defined as max-BI. The vision depth for observation at the time of measurement was kept to a minimum, and the equipment was zoomed as necessary, for example when the vessel was located deep. The mean of the values for the left and right carotid arteries was used as the measurement result for mean-C, while the greater of the values for the left or right was used for max-C and max-BI. The equipment used for to measure IMT was the same as that for AT.
Clinical and Laboratory CharacteristicsBlood pressure (BP) at rest while sitting was measured manually. Hypertension was defined as use of antihypertensive drugs or BP >140 mmHg systolic or >90 mmHg diastolic, or both, at the time of AT-T ultrasonographic measurement. Concurrent with US, blood was collected from the vein while in a fasting state, measuring the total cholesterol (TC), triglycerides (TG), HDL-cholesterol (HDL-C), and LDL-C using general-purpose equipment (LABOSPECT 008, Hitachi High-Technologies, Tokyo, Japan; Qualligent, Sekisui Medical Co., Ltd., Tokyo, Japan) and hemoglobin A1c (HbA1c) using specialized equipment (HLC 723 G9; Tosoh Corp., Tokyo, Japan), all in the clinical laboratory of the NCVC. Diabetes mellitus was defined according to the Evidence-based Practice Guideline for the Treatment for Diabetes in Japan 2013. Body mass index (BMI) was calculated as the body weight at the time of visit (kg) divided by the square of the height (m). Information on smoking history was obtained in medical interviews with subjects. Those with a current smoking or past smoking habit were defined as subjects with a smoking history. CAD was defined as any of the following: (1) myocardial infarction proven by ECG abnormalities and enzyme changes; (2) diagnosis of angina pectoris with significant stenosis ≥75% on cardio-angiogram; or (3) coronary bypass surgery or percutaneous coronary intervention.
DNA AnalysisDNA analysis was performed as previously reported.16 Primers covering all the exons and exon-intron boundary sequences of proprotein convertase subtilisin/kexin 9 (PCSK9) (12 exons) and LDL receptor (LDLR) (18 exons) were designed.16
Statistical AnalysisSingle regression analysis was used to analyze relationships between AT measurements and individual carotid artery factors, while Pearson’s correlation coefficients were used for the correlation analysis. The Mann-Whitney test was used for comparisons between groups, and the Kruskal-Wallis test was used for comparisons among multiple groups. In the present study, the statistical significance level was set at P<0.05 and SPSS (version16.0, Chicago, IL, USA) was used for the analyses.
The clinical characteristics of the study subjects are shown in Table 1. The age of patients ranged from 20 to 87 years in the FH group and from 20 to 88 years in the non-FH group. There were 101 patients with dyslipidemia, 24 with diabetes, and 30 with both conditions among the non-FH group. Smoking history was noted in 45 patients (35%) in the FH group and 41 patients (26%) in the non-FH group. There were 34 patients (26%) with CAD in the FH group, and 23 patients (15%) in the non-FH group. Age, BMI, TG and HbA1c levels in the non-FH group were significantly higher compared with the FH group, but no significant differences were observed between the groups for lipid parameters. Among the 130 FH patients, 111 (85.4%) had a LDLR-defective or -negative mutation, 10 (7.7%) had a PCSK9 gain-of-function mutation and 9 (6.9%) had both.
| Total | FH | Non-FH | P value (FH vs. non-FH) |
|
|---|---|---|---|---|
| n (persons) | 285 | 130 | 155 | |
| Female, n (%) | 161 (56) | 77 (59) | 84 (54) | 0.388 |
| Age (years) | 57.7±16.4 | 53.2±18.6 | 61.2±13.4 | <0.001 |
| BMI (kg/m2) | 22.8±3.3 | 21.9±2.9 | 23.8±3.6 | <0.001 |
| TC (mg/dL) | 220±58 | 216±60 | 225±57 | 0.423 |
| LDL-C (mg/dL) | 146±54 | 146±55 | 141±48 | 0.219 |
| HDL-C (mg/dL) | 54±17 | 52±16 | 55±18 | 0.095 |
| TG (mg/dL) | 99 (69–149) | 97 (60–118) | 120 (84–176) | 0.001 |
| HbA1c (%) | 6.2±1.1 | 5.8±0.6 | 6.3±1.3 | <0.001 |
| Diabetes mellitus (%) | 74 (26) | 19 (15) | 55 (35) | <0.001 |
| Hypertension (%) | 60 (21) | 28 (22) | 32 (21) | 0.944 |
| Smoking history, n (%) | 86 (30) | 45 (35) | 41 (26) | 0.492 |
| CAD, n (%) | 57 (20) | 34 (26) | 23 (15) | 0.018 |
| Statin treatment (%) | 172 (60) | 108 (83) | 64 (41) | <0.001 |
Values are presented as mean±standard deviation, or median (interquartile range 25–75%) or number. BMI, body mass index; CAD, cardiovascular disease; DBP, diastolic blood pressure; HbA1c, hemoglobin A1c; HDL-C, high-density lipoprotein-cholesterol; LDL-C, low-density lipoprotein-cholesterol; SBP, systolic blood pressure; TC, total cholesterol; TG, triglycerides.
The outline and internal properties of the AT could be clearly determined using US. However, on soft X-ray radiographs, the border with the skin was unclear (Figure 1B). Regarding the internal properties on US, a linear multiple hyper-echoic layer structure, considered to be a fibrous structure, was observed for normal AT-T, but as the AT thickened, this layered structure was no longer present and a non-uniform low-echo image was generally obtained (Figure 1C,D). The appearance of the layered structure along the thickened AT could not be determined using soft X-ray radiography. Although calcification could be extracted using both US and soft X-ray radiography, the latter was superior in this regard because it enabled better understanding of the overall image. Calcification within the AT was observed in 9 patients (7%) in the FH group but in none of the patients (0%) in the non-FH group.
Figure 2 shows the good correlation between US and soft X-ray radiography (r=0.924, P<0.001). The thickness measured by US was relatively lower than that by soft X-ray radiography.

Scatterplots of Achilles tendon thickness (AT-T) using radiography and ultrasonography among FH subjects (n=106). A good correlation was observed between the 2 methods (R=0.924, P<0.001). The thickness measured by ultrasonography was relatively lower than that by soft X-ray radiography. FH, familial hypercholesterolemia; US, ultrasonography.
Table 2 shows sex differences in AT and carotid artery measurements among patients with FH. The AT-T, AT-W, AT-A, mean-C, max-C, and max-BI were significantly higher in male patients. However, no difference between male and female patients was observed for T/W, suggesting that the AT becomes circular in shape in both sexes in a similar manner.
| Male | Female | P value | |
|---|---|---|---|
| AT-T (mm) | 9.2 (5.8–11.70) | 6.2 (5.6–9.7) | <0.001 |
| AT-W (mm) | 17.9 (14.1–20.8) | 13.0 (12.9–17.9) | <0.001 |
| AT-A (cm2) | 1.50 (0.64–1.86) | 1.28 (0.53–1.45) | <0.001 |
| T/W | 0.49 (0.40–0.57) | 0.46 (0.44–0.56) | 0.389 |
| mean-C (mm) | 1.1 (0.7–1.2) | 0.9 (0.6–1.0) | 0.037 |
| max-C (mm) | 1.6 (0.6–2.1) | 1.1 (0.7–1.3) | 0.021 |
| max-BI (mm) | 2.1 (1.3–2.7) | 1.4 (1.2–2.3) | 0.024 |
Values are presented as median (interquartile range 25–75%). AT-A, ultrasonographic Achilles tendon area; AT-T, ultrasonographic Achilles tendon thickness; AT-W, ultrasonographic Achilles tendon width; CCA, common carotid artery; IMT, intima-media thickness; max-BI, maximum IMT in bifurcation and internal carotid artery; max-C, maximum IMT in CCA; mean-C, mean IMT in CCA; T/W, ratio AT-T to AT-W.
We conducted a linear regression analysis with risk factors to clarify the correlations with AT-T in the FH group (Table 3). Age and smoking history were associated with increased AT-T, although no relationships were observed between AT-T and sex, BMI, LDL-C, or TG.
| β | 95% CI | P value | |
|---|---|---|---|
| Age | 0.28 | 0.02 to 0.10 | 0.005 |
| Female | −0.02 | −1.81 to 1.46 | 0.835 |
| BMI | −0.11 | −0.40 to 0.09 | 0.207 |
| LDL-C | −0.02 | −0.01 to 0.01 | 0.833 |
| HDL-C | −0.11 | −0.08 to 0.02 | 0.229 |
| TG | 0.04 | −0.01 to 0.01 | 0.614 |
| Smoking history | 0.27 | 0.62 to 3.63 | 0.006 |
β, standardized regression coefficient; CI, confidence interval. Other abbreviations as in Table 1.
The predictability of FH using each AT factor was analyzed by means of receiver-operating characteristic (ROC) curves (Figure 3). The area under the ROC curve (95% confidence interval [CI]) for AT-T was 0.83 (0.74–0.92) in male patients and 0.90 (0.86–0.95) in female patients. The cutoff values for AT-T in the present analyses were 5.8 mm (sensitivity 71%, specificity 78%) in men, and 5.5 mm (sensitivity 80%, specificity 81%) in women. Thickness thresholds for the diagnosis of FH with specificity of approximately 80% were 6.0 mm (sensitivity 63%, specificity 85%) in men, and 5.5 mm (sensitivity 80%, specificity 81%) in women.
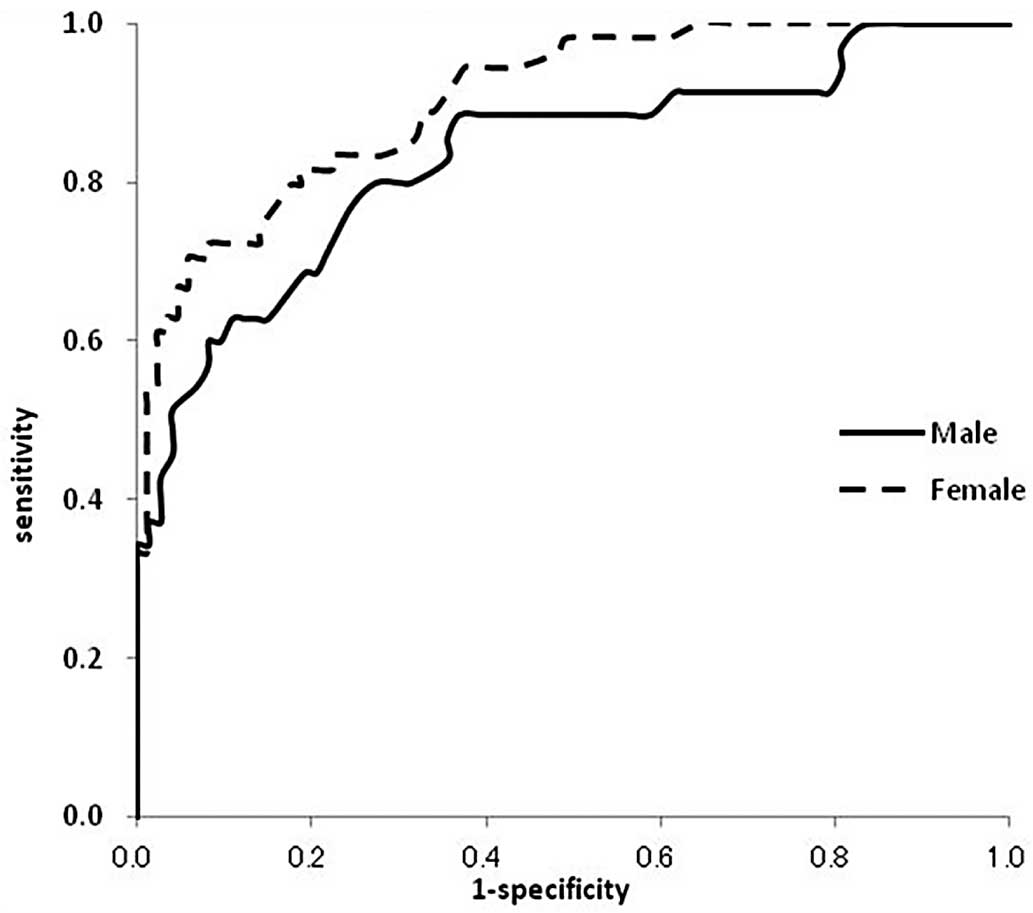
Receiver-operating curves (ROC) for Achilles tendon thickness (AT-T) measured by ultrasonography for the diagnosis of familial hypercholesterolemia. The area under the ROC curve (95% CI) for AT-T was 0.83 (0.74–0.92) in male patients (n=64) and 0.90 (0.86–0.95) in female patients (n=85). The cutoff values for AT-T in the present analyses were 5.8 mm (sensitivity 71%, specificity 78%) in men, and 5.5 mm (sensitivity 80%, specificity 81%) in women.
Scatterplot analyses revealed significant correlations between AT-T and carotid IMT only among the FH patients (Figure 4). Additionally, in a logistic regression analysis adjusted for age, sex (female) and traditional cardiovascular risk factors (smoking history [current and past], presence of hypertension, presence of diabetes mellitus, obesity, LDL-C, HDL-C, and log-triglycerides), increased AT-T was associated with the presence of CAD (odds ratio (OR) per 1-SD increase, 1.33; 95% CI, 1.10–1.60; P=0.004) (Table 4), suggesting that AT-T measured by US is not only a diagnostic criterion but also a useful risk marker for both asymptomatic atherosclerosis and CAD in FH. Although we found that AT-W (OR per 1-SD increase, 1.43; 95% CI, 1.07–1.82; P=0.008) and AT-A (OR per 1-SD increase, 1.72; 95% CI, 1.07–2.78; P=0.026) were also independently associated with increased risk for CAD, we concluded that AT-T was the best marker among the values of AT size.

Correlation between ultrasonographic Achilles tendon thickness (AT-T) and carotid artery intima–media thickness (IMT) among patients with familial hypercholesterolemia (FH) (A–C, n=130) and those without FH (D–F, n=155). AT-T was positively associated with carotid IMT only among patients with FH. mean-C, mean IMT in common carotid artery (CCA); max-C, maximum IMT in CCA; max-BI, maximum IMT in bifurcation and internal carotid artery.
| OR | 95% CI | P value | |
|---|---|---|---|
| AT-T | 1.17 | 1.02–1.36 | 0.030 |
| Age | 1.02 | 0.99–1.05 | 0.170 |
| Female | 0.77 | 0.24–2.50 | 0.665 |
| Smoking history | 2.00 | 0.65–6.17 | 0.228 |
| LDL-C | 1.00 | 0.99–1.01 | 0.445 |
| HDL-C | 0.93 | 0.89–0.97 | <0.001 |
| TG | 1.00 | 0.99–1.02 | 0.263 |
| Hypertension | 2.94 | 0.77–11.3 | 0.115 |
| Obesity | 1.02 | 0.24–4.39 | 0.977 |
| Diabetes mellitus | 2.87 | 0.58–14.2 | 0.197 |
OR, odds ratio. Other abbreviations as in Tables 1–3.
The present study, for the first time, determined a cutoff value for AT-T measured by US to discriminate FH from non-FH individuals among Japanese patients with dyslipidemia. In addition, we found that AT-T measured by US was not only a diagnostic criterion for FH but also an important predictor of CAD in FH patients.
In the FH diagnosis criteria of the Japan Atherosclerosis Society, the same criterion of AT-T ≥9 mm on soft X-ray radiography is used for both men and women. Thus, it is possible that female patients with mild AT thickness may be underdiagnosed, as noted in a previous study.11 In the present study, the median AT-T measured by US was 9.2 mm for male FH patients, and 6.2 mm for female patients (Table 2). As there was no relationship between sex and AT-T in the linear regression analysis, we set cutoff values for both sexes to discriminate FH patients. Using the same criterion for both sexes, the area under the ROC curve (95% CI) for AT-T was 0.86 (0.81–0.91) and the cutoff value for AT-T was 5.8 mm (sensitivity 72%, specificity 82%). Sex-segregated analyses allowed us to set better cutoff values. We also determined AT-T thresholds for the diagnosis of FH with a specificity of approximately 80%, which were a maximum thickness of 6.0 mm for men and a maximum thickness of ≥5.5 mm for women. We propose that male and female patients fulfilling these criteria, respectively, be regarded as having thickening of AT.
In line with other studies,8,10 we found that the AT-T was less on US than on soft X-ray radiography (Figure 2). The outline of the AT could be clearly distinguished by US, but its border with the skin was unclear on soft X-ray radiography, making it difficult to measure the AT alone with the latter. Therefore, we speculate that the discrepancy in AT-T measurements is related to overestimation by soft X-ray radiography. As the AT-T cutoff value for US in Westerners has been reported to be 5.8 mm10 or 4.8–5.3 mm14, the cutoff values in the present study (5.8 mm in men and 5.5 mm in women) are not so different from those for Westerners.
In the present study, there was a relationship between smoking history and AT-T, which is interesting given smoking’s involvement in inflammation and oxidative stress. Oosterveer et al reported that the presence of AT xanthomas in FH patients is associated with genetic variation in LDL oxidation pathways.17 Furthermore, Artieda et al reported that AT xanthoma formation was associated with higher intracellular lipid content, and a stronger inflammatory response of macrophages to oxidized LDL.18 Accordingly, we speculate that the oxidative and inflammatory properties of LDL might play significant roles in the formation of AT xanthomas.
Interestingly, a significant correlation between AT-T and carotid IMT was observed in the FH group, but not in the non-FH group, suggesting that cumulative LDL in vessels rather than other lipids or glucose might be an important common factor for the development of both xanthomas and atherosclerosis. It is also important that increased AT-T was associated with increased risk of CAD, even after adjusting for traditional cardiovascular risk factors, in FH patients. We have shown that cholesterol efflux capacity,13 a metric function of HDL, and a gene variant (PCSK9 V4I variant with LDL receptor)16 are risk factors for developing atherosclerosis. In the present study, we found that AT-T measured by US was useful in risk assessment for asymptomatic arteriosclerosis as well as symptomatic CAD in FH patients.
Study LimitationsFirst, the study had a cross-sectional design, so we could only demonstrate associations and not causal relationships between AT-T and IMT, as well as CAD. In addition, although it seems difficult to achieve decreases in AT-T by medical treatment, it is ethically unacceptable to investigate the effect of an increase in AT-T on CAD and IMT in prospective observation studies without medical treatment. Second, multiple ultrasonographers carried out the examinations across 2 study sites, which may have affected the reliability of the measurement results. However, when we investigated inter-examiner reliability for the AT-T measurements of 10 patients, a high inter-examiner correlation coefficient of approximately 0.9 was obtained. Third, the mean age of the non-FH group was higher than that of the FH group. Although we observed a positive association between age and AT-T in FH patients, this relationship was not found in the non-FH group (r=−0.087, P=0.296). Therefore, we think that we did not overestimate AT-T in the non-FH group. Finally, we did not measure LDL-C levels before lipid-lowering treatment or evaluate the length of time that patients had been untreated. Therefore, pretreatment duration and levels of LDL-C as a predictor for AT-T might have been underestimated. Because AT-T was not associated with serum LDL-C levels in this study, we speculate that the AT-T was not affected by the LDL-C at the time of measuring, but by the severity and duration of high LDL-C levels as in previous studies.19,20 Therefore, our findings suggested that early diagnosis and adequate treatment are important to prevent development of atherosclerosis as well as thickening of the AT.
The present study set an AT-T cutoff value for US to discriminate FH in Japanese people for the first time. In addition, we revealed that AT-T was a useful risk marker for CAD and asymptomatic arteriosclerosis. Because ultrasonography is a safe, low-cost, rapid, and reliable procedure, many clinicians, including those in general practice, have an ultrasound imaging system. Although the issue of standardization of AT measurement by US remains, our findings should encourage clinicians to diagnose FH more actively, which could solve the problem of underdiagnosis of FH.
None.
M.O. received speaker honoraria from Astellas Pharma Inc., Amgen Astellas BioPharma., and Sanofi. M.H.-S. received speaker honoraria from Astellas Pharma Inc., Amgen Astellas BioPharma., and Sanofi.
Intramural Research Fund (25-2-5) for Cardiovascular Diseases of National Cerebral and Cardiovascular Center.