Article ID: CJ-18-0337
Article ID: CJ-18-0337
Background: Due to concern about bleeding complications, a maintenance dose of prasugrel 2.5 mg may be used in elderly or low-body-weight patients in Japan. There is little information, however, on the efficacy and safety of a 2.5-mg maintenance dose of prasugrel.
Methods and Results: In this single-center, prospective, open-label, cross-over study, a total of 44 elderly (≥75 years old) or low body-weight (<50 kg) Japanese patients >1 month after percutaneous coronary intervention who were treated with aspirin 81–100 mg and clopidogrel 75 mg were randomized to either prasugrel 2.5 mg or 3.75 mg instead of clopidogrel for 14 days, with a cross-over directly to the alternate treatment for another 14 days. Platelet inhibition was assessed with the VerifyNow assay (Accumetrics, San Diego, CA, USA) at 3 time points: baseline; day 14; and day 28. P2Y12 reaction units (PRU) ≤95 was defined as low on-treatment platelet reactivity (LPR), and PRU ≥262 as high on-treatment platelet reactivity (HPR). The prevalence of LPR was 2.2% in patients treated with clopidogrel, 2.2% in those with prasugrel 2.5 mg, and 22.7% in those with prasugrel 3.75 mg (P<0.001). Clopidogrel resulted in the higher prevalence of HPR compared with 2.5-mg and 3.75-mg prasugrel (40.9% vs. 18.2% vs. 6.8%, P<0.001).
Conclusions: Prasugrel 2.5 mg may be more appropriate in elderly or lower-body-weight Japanese patients.
Dual antiplatelet therapy (DAPT) with aspirin and a P2Y12 inhibitor plays an important role in preventing thrombotic complications in patients undergoing percutaneous coronary intervention (PCI).1–3 With the advent of new P2Y12 inhibitors such as prasugrel or ticagrelor and the improvement of stent technology, thrombotic events including stent thrombosis tend to be decreased. In recent years, however, much attention has been focused on bleeding complications.4–6 Randomized studies in the USA and Europe have noted a higher incidence of bleeding complications with prasugrel compared with clopidogrel.7 Considering the higher average age, lower body weight, and increased bleeding risk with other thrombotic agents in Japanese patients compared with Western patients, the maintenance dose of prasugrel in Japanese patients was determined to be approximately one-third that of Western patients (3.75 mg vs. 10 mg). Furthermore, the lower maintenance dose of prasugrel (2.5 mg) may be used in elderly patients or those with a low body weight. There is little information, however on the efficacy and safety of a 2.5-mg maintenance dose of prasugrel. Japanese Phase III trials (The PRASugrel compared with clopidogrel For Japanese patIenTs with Acute Coronary Syndrome undergoing PCI [PRASFIT-ACS] study and the PRASugrel compared with clopidogrel For Japanese patIenTs with CAD undergoing Elective PCI [PRASFIT-Elective) study) have shown optimal efficacy and safety of prasugrel at loading and maintenance dose 20 mg and 3.75 mg.8,9 Those studies, however, did not use a 2.5-mg maintenance dose in elderly patients or in those with low body weight. Thus, the present study evaluated the efficacy and safety of a 2.5-mg maintenance dose of prasugrel in elderly or low-body-weight Japanese patients.
This study was a single-center, prospective, open-label, cross-over study to evaluate the efficacy and safety of a 2.5-mg maintenance dose of prasugrel by measuring platelet inhibition compared with 3.75-mg prasugrel in elderly or low-body-weight patients >1 month after PCI who were treated with aspirin 81–100 mg and clopidogrel 75 mg. Elderly was defined as ≥75 years old, and low body weight as <50 kg. Patients were excluded in the presence of any of the following: (1) contraindications to prasugrel; (2) other anti-thrombotic agents except aspirin; (3) severe liver dysfunction (Child-Pugh class C); (4) hemodialysis; (5) platelet count ≤10×104 µL, and (6) history of stroke. A flow chart of the study is shown in Figure 1.
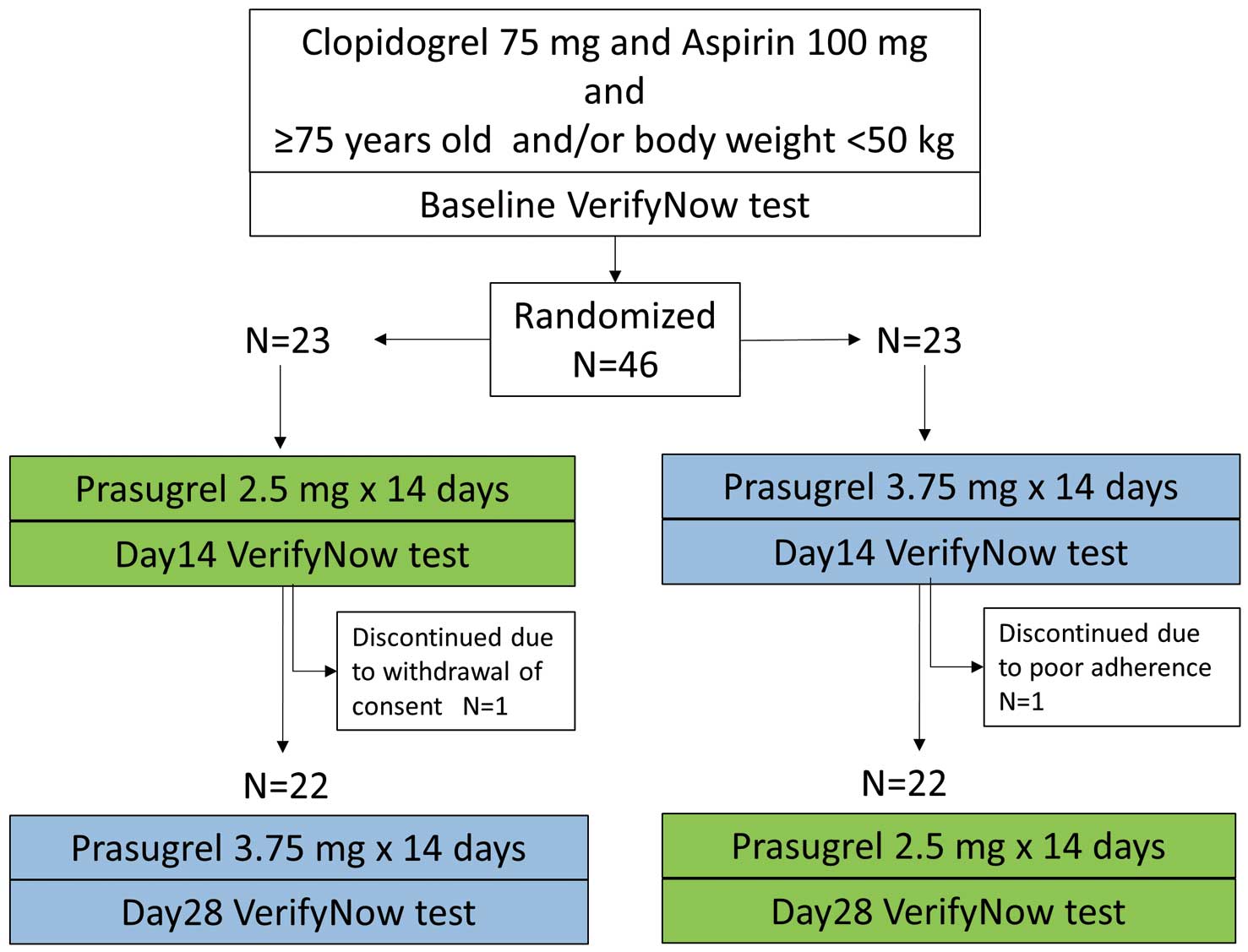
Flow chart of subject selection.
Patient were randomized to either prasugrel 2.5 mg or 3.75 mg instead of clopidogrel for 14 days, with a cross-over directly to the alternate treatment for another 14 days. There was no wash-out period. Blood samples for platelet function analysis were collected at 3 time points: baseline (clopidogrel 75 mg); day 14 (prasugrel 2.5 mg or 3.75 mg); and day 28 (prasugrel 3.75 mg or 2.5 mg). Platelet inhibition was assessed with the VerifyNow assay (Accumetrics, San Diego, CA, USA). It measures adenosine diphosphate-induced platelet function that is reported as P2Y12 reaction units (PRU). It was performed according to standard protocols described in detail elsewhere.10,11 Taking into account the racial difference and based on previous studies,4–6,12 PRU ≤95 was defined as low on-treatment platelet reactivity (LPR), PRU ≥262 as high on-treatment platelet reactivity (HPR), and 95<PRU<262 as optimal treatment reactivity (OPR).
The primary end-point was comparison of the prevalence of LPR between the 2.5-mg and 3.75-mg maintenance doses of prasugrel. Additional end-points were mean PRU and the prevalence of HPR and LPR between clopidogrel, 2.5 mg-prasugrel, and 3.75-mg prasugrel. The bleeding events were assessed using Bleeding Academic Research Consortium (BARC) criteria.13
The protocol was approved by the institutional review boards at Chiba University Hospital, and the study was conducted in accordance with regulatory standards and ethics guidelines for clinical studies according to the Declaration of Helsinki. All patients provided written informed consent. The present study was registered at the University Hospital Medical Information Network Clinical Trials Registry (number: UMIN 0000 19424) in Japan.
Statistical AnalysisThere are no published data on the difference in platelet reactivity assessed using VerifyNow in the same patients given 2.5- and 3.75-mg prasugrel. Thus, we used the data of a Japanese Phase II trial of prasugrel, in which patients received clopidogrel, 2.5- or 3.75-mg prasugrel.14 Based on that trial, the rate of LPR was estimated as 5.6% on 2.5-mg prasugrel treatment and 20.8% on 3.75-mg prasugrel. We estimated that 70 patients were required for a power of 80% and a 2-sided α level of 0.05, assuming a dropout rate of 5%. For baseline characteristics, continuous variables are presented as mean±SD, and analyzed using Student’s t-test or Mann-Whitney U-test. Categorical variables are presented as count and percentage, and compared using the chi-squared test or Fisher exact test. Comparison of proportion, including primary end-point, was performed using McNemar test. Post-hoc analysis with a general linear model was performed to evaluate PRU difference between groups. Analysis of the primary and secondary outcomes was done in the full analysis set. All analyses were performed with SAS version 9.4 (SAS Institute, Cary, NC, USA). Statistical significance was defined as 2-sided P<0.05.
EthicsThis study complied with the Declaration of Helsinki in regard to investigation in humans and was approved by the institutional ethics committees at Chiba University Hospital. There was no industry involvement in the design, conduct, financial support, or analysis of this study.
Between December 2016 and February 2017, a total of 46 post-PCI patients ≥75 years of age and/or body weight <50 kg were enrolled. All patients were randomly assigned to the 2 groups: 2.5-mg prasugrel followed by 3.75-mg prasugrel; and 3.75-mg prasugrel followed by 2.5-mg prasugrel. Two patients dropped out because of poor adherence and withdrawal of consent, leaving 44 patients available in the final analysis. There was no significant difference in baseline characteristics between the 2 groups (Table 1). The elderly; low-body-weight; and elderly–low-body-weight groups consisted of 39, 8, and 3 patients, respectively. Because of the end of the initially determined inclusion period, the present study was terminated before the planned number of patients were enrolled.
| Variable | All (n=44) |
Prasugrel | P-value | |
|---|---|---|---|---|
| 2.5 mg→3.75 mg (n=22) |
3.75 mg→2.5 mg (n=22) |
|||
| Age (years) | 78.6±4.3 | 78.2±4.2 | 79.0±4.5 | 0.53 |
| Men | 30 (68.2) | 15 (68.2) | 15 (68.2) | >0.99 |
| Body weight (kg) | 61.1±10.6 | 60.5±8.6 | 61.8±12.4 | 0.67 |
| BMI (kg/m2) | 24.3±3.5 | 24.1±2.7 | 24.5±4.2 | 0.73 |
| Hypertension | 33 (75.0) | 16 (72.7) | 15 (77.3) | >0.99 |
| Diabetes mellitus | 24 (54.5) | 11 (50.0) | 13 (59.1) | 0.76 |
| Dyslipidemia | 35 (79.5) | 17 (77.3) | 18 (81.8) | >0.99 |
| Current smoker | 3 (7.2) | 2 (9.1) | 1 (4.5) | >0.99 |
| Clinical presentation for PCI | ||||
| ACS | 25 (56.8) | 9 (40.9) | 16 (72.7) | 0.32 |
| Non-ACS | 19 (43.2) | 13 (59.1) | 6 (27.3) | 0.07 |
| DES use | 44 (100) | 22 (100) | 22 (100) | >0.99 |
| Medication | ||||
| Aspirin | 44 (100) | 22 (100) | 22 (100) | >0.99 |
| Statin | 42 (95.5) | 22 (100) | 20 (90.9) | 0.49 |
| ACEi/ARB | 27 (61.4) | 12 (54.5) | 15 (68.2) | 0.54 |
| Calcium channel blockers | 28 (63.6) | 14 (63.6) | 14 (63.6) | >0.99 |
| β-blockers | 23 (52.3) | 12 (54.5) | 11 (50.0) | >0.99 |
| Proton pump inhibitors | 36 (81.8) | 17 (77.3) | 19 (86.4) | 0.70 |
| Laboratory data | ||||
| Hematocrit (%) | 37.8±3.7 | 38.8±3.1 | 36.9±4.0 | 0.08 |
| Platelet count (×104/μL) | 23.5±20.7 | 26.8±28.4 | 20.2±6.8 | 0.30 |
| Creatinine clearance (mL/min) | 53.4±13.0 | 55.7±13.1 | 51.0±12.8 | 0.23 |
| Creatinine clearance <60 mL/min | 33 (75.0) | 16 (72.7) | 17 (77.3) | >0.99 |
| Platelet reactivity at baseline | 240.3±65.8 | 245.3±57.4 | 235.4±74.3 | 0.93 |
| CYP2C19 genotype | ||||
| Extensive metabolizer | 18 (40.9) | 10 (45.4) | 8 (36.3) | 0.76 |
| Intermediate metabolizer | 20 (45.5) | 8 (36.3) | 12 (54.5) | 0.36 |
| Poor metabolizer | 6 (13.6) | 4 (18.2) | 2 (9.1) | 0.66 |
Data given as n (%) or mean±SD. ACEi, angiotensin-converting enzyme inhibitor; ACS, acute coronary syndrome; ARB, angiotensin II receptor blocker; BMI, body mass index; DES, drug-eluting stent; PCI, percutaneous coronary intervention.
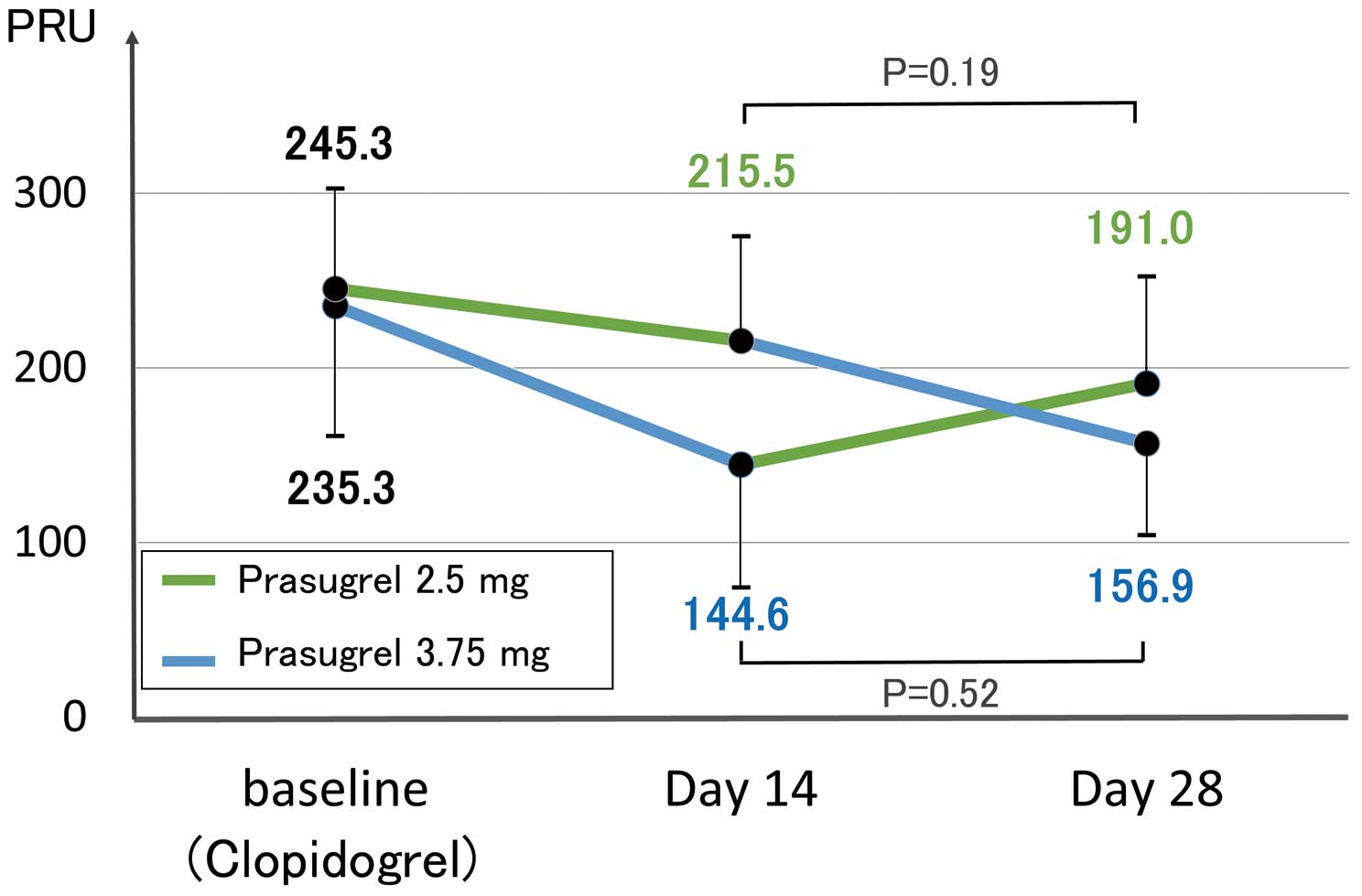
Figure 2 shows the PRU of the pre-crossover and post-cross-over measurements. No period or carryover effect was found. Mean PRU was lower in patients treated with prasugrel 3.75 mg than in those with clopidogrel or prasugrel 2.5 mg (Figure 3). Individual PRU are also presented in Figure 3. Moreover, the prevalence of LPR, HPR and OPR obtained from Figure 3 is summarized in Table 2. The primary end-point, that is, prevalence of LPR, was observed less frequently for 2.5-mg prasugrel than for 3.75-mg prasugrel. Clopidogrel resulted in the higher prevalence of HPR compared with 2.5- and 3.75-mg prasugrel. The prevalence of OPR was significantly higher with 2.5-mg prasugrel compared with clopidogrel.
Platelet inhibition during the pre-cross-over and post-cross-over periods. PRU, platelet reactivity unit.

Individual and mean±SD platelet reactivity units (PRU) on treatment. HPR, high on-treatment reactivity; LPR, low on-treatment reactivity.
| Clopidogrel | Prasugrel 2.5 mg |
Prasugrel 3.75 mg |
P-value | |||
|---|---|---|---|---|---|---|
| Clopidogrel vs. Prasugrel 2.5 mg |
Prasugrel 2.5 mg vs. Prasugrel 3.75 mg |
Clopidogrel vs. Prasugrel 3.75 mg |
||||
| LPR | 1 (2.3) | 1 (2.3) | 10 (22.7) | >0.99 | 0.004 | 0.012 |
| HPR | 18 (40.9) | 19 (43.1) | 7 (15.9) | 0.021 | 0.063 | <0.001 |
| OPR | 25 (56.8) | 35 (76.1) | 31 (70.5) | 0.031 | 0.424 | 0.286 |
Data given as n (%). HPR, high on-treatment platelet reactivity (PRU ≥262); LPR, low on-treatment platelet reactivity (PRU ≤95); OPR, optimal on-treatment platelet reactivity (95<PRU<262); PRU, platelet reactivity units.
There was no stent thrombosis or major adverse cardiac event. No major bleeding (BARC class ≥2) occurred during the study period. BARC class 1 bleeding complications, most of which were nosebleed or subcutaneous bleeding, were observed in 11 (25.0%) and 17 patients (38.6%) with 2.5- and 3.75-mg prasugrel, respectively (P=0.083).
The present study has shown that (1) the prevalence of LPR on 2.5-mg prasugrel was significantly lower than 3.75-mg prasugrel; (2) the prevalence of HPR on 2.5- and 3.75-mg prasugrel was substantially lower than on clopidogrel; and (3) the prevalence of OPR was significantly higher with 2.5-mg prasugrel compared with clopidogrel. To prevent stent thrombosis, patients undergoing PCI must take DAPT. Furthermore, it also reduces the risk of myocardial infarction in non-culprit lesions. DAPT, however, increases the risk of bleeding complications associated with an increased risk of morbidity and mortality.4–6,15–17 Thus, balancing the long-term risks of ischemic and bleeding complications with DAPT in patients undergoing PCI is important. Based on the Japanese Phase II trial, prasugrel loading and maintenance dose in Japanese patients were determined as 20 mg and 3.75 mg, respectively.14 Although the PRASFIT-ACS study and PRASFIT-Elective study showed the usefulness of these doses of prasugrel in Japanese patients,8,9 it may be better to prescribe a lower dose of prasugrel in patients with a high risk of bleeding complications.
Prasugrel achieves greater platelet inhibition with less variability between patients than does clopidogrel. Bleeding complications with prasugrel may be a concern in elderly and low-body-weight patients. The Japanese Phase II trial compared outcomes between 3.75- and 5-mg prasugrel.14 In the present study, elderly (≥75 years old) and low-body-weight (≤50 kg) patients received 2.5- (n=37) and 3.75-mg prasugrel (n=37). Lower mean PRU was noted with 3.75-mg prasugrel compared with 2.5-mg prasugrel (178.2±80.76 vs. 240.1±54.75, P<0.01). In contrast to the present study, the Japanese phase II trial was not a cross-over study, which is statistically efficient and which reduces the influence of confounding covariates. Furthermore, the Japanese Phase II trial did not describe the prevalence of LPR.14
A collaborative analysis of 17 studies with 20,839 patients noted LPR <95 PRU and HPR >208 PRU for VerifyNow. The risk ratio (RR) of stent thrombosis was 2.73 (95% CI: 2.03–3.69) in patients with HPR. In contrast, patients with LPR had a higher RR of bleeding complications (1.74; 95% CI: 1.47–2.06), without any further benefit in stent thrombosis (RR, 1.06; 95% CI: 0.68–1.65). East Asian patients, including Japanese patients, have a higher incidence of bleeding complications with antithrombotic therapy.18,19 More careful attention should be paid to LPR with P2Y12 inhibitors. With regard to this, 2.5-mg prasugrel may be more appropriate in Japanese patients. In this study, there were no cases of major bleeding either with 2.5- or 3.75-mg prasugrel, whereas there was a trend toward more frequent minor bleeding with 3.75-mg prasugrel despite the short observation period. Even non-critical bleeding may be associated with drug discontinuation, which results in a risk of stent thrombosis and ischemic complications. In contrast, whether the difference in platelet activity assessed using VerifyNow has clinical significance in terms of lower thrombotic events in Japanese population is unknown. A previous study in Western patients noted the usefulness of assessment of platelet reactivity to predict long-term outcomes after drug-eluting stent implantation.20 Furthermore, the PRASFIT-ACS study also noted an optimal PRU cut-off of 262 for preventing major adverse cardiac events in Japanese patients undergoing PCI.12 The incidence of major adverse cardiac events was 8.8% in patients with PRU ≤262 and 15.5% in those with PRU >262 (OR, 0.51; 95% CI: 0.29–0.90).12 Further studies with large Japanese patient populations, however, are required to strengthen the evidence of the present study.
Study LimitationsThere are several limitations in the present study. First, this study was based on a relatively small sample and ended early, although it did not reach the initially planned number of patients. There was a significant difference, however, between 2.5- and 3.75-mg prasugrel in terms of the primary end-point, which was the prevalence of LPR. Second, this was a pharmacodynamic study and it had a short observation period. Further studies with a larger number of patients and longer follow-up period are required to confirm the clinical impact of higher LPR on clinical outcome. Third, there was no clopidogrel arm in this study. All patients were randomized to either prasugrel 2.5 mg or 3.75 mg instead of clopidogrel for 14 days, with a cross-over directly to the alternate treatment without a wash-out period. Therefore, the present data may not completely prove the safety and efficacy of prasugrel 2.5 mg in comparison with clopidogrel 75 mg. Fourth, randomization was performed >1 month after PCI regardless of clinical presentation, such as acute coronary syndrome (ACS). Therefore, the results might not apply to patients with ACS at the time of revascularization. Fifth, it is unclear whether the definition of LPR (≤95 PRU) is appropriate in Japanese patients. East Asian subjects, however, have more bleeding complications compared with Western patients. Thus, utilizing this definition, it may be possible to confirm a higher risk of 3.75-mg prasugrel for bleeding complications in elderly and low-body-weight Japanese patients.
LPR is more frequent in patients treated with 3.75-mg prasugrel compared with 2.5-mg prasugrel in elderly or lower body-weight Japanese patients. Prasugrel 2.5 mg may be more appropriate in those patients.
The authors wish to acknowledge Dr. Kengo Nagashima, Department of Global Clinical Research, Graduate School of Medicine, Chiba University, for his support in statistical analysis.
Y.K. received research grant from Daiichi Sankyo (Tokyo, Japan) and Sanofi (Paris, France). The other authors declare no conflicts of interest.