Article ID: CJ-19-0281
Article ID: CJ-19-0281
A 72-year-old man was referred for progressive dyspnea (New York Heart Association class III) and peripheral edema since the previous year. There was no family history of cardiovascular disease. Chest X-ray showed cardiomegaly and bilateral pleural effusion. Electrocardiogram showed sinus bradycardia and low QRS voltage (Figure A). Echocardiogram showed thickened biventricular walls and biatrial enlargement (Figure B,C). Left ventricular (LV) ejection fraction (LVEF) was reduced to 43%, and the LV had a restrictive filling pattern (Figure D). Cardiac magnetic resonance imaging showed diffuse transmural delayed gadolinium enhancement of the LV (Figure E).
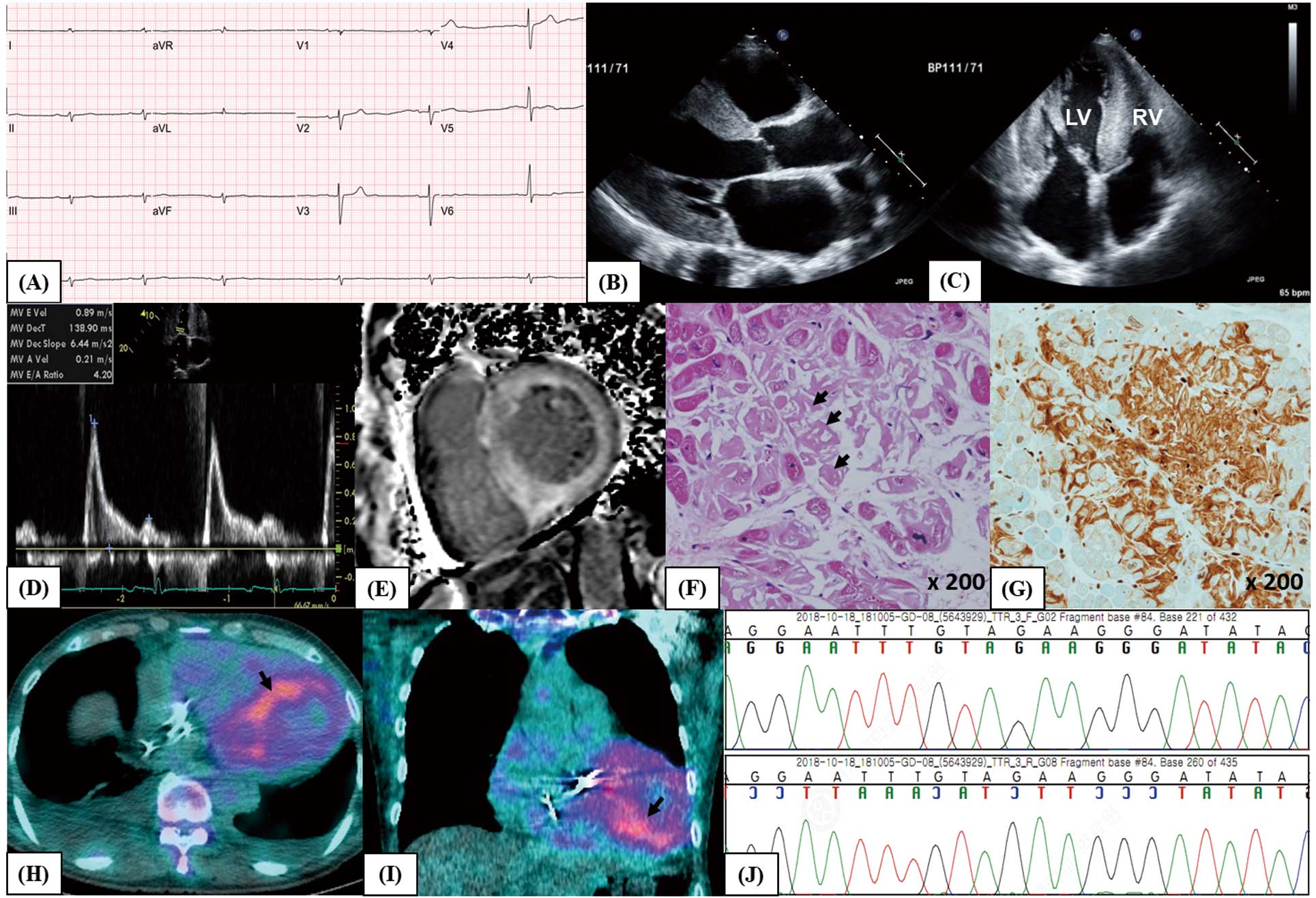
(A) Electrocardiogram showing low QRS voltage in limb leads. (B,C) Echocardiogram showing thickened biventricular walls and valves with biatrial enlargement; (B) parasternal long-axis view; (C) apical 4-chamber view. (D) Trans-mitral left ventricular (LV) inflow showing a restrictive pattern. (E) Cardiac magnetic resonance imaging showing diffuse transmural delayed gadolinium enhancement of the LV. (F,G) Endomyocardial biopsy of the right ventricle (RV) for (F) H&E (arrows, deposition of amorphous material in the myocardial interstitium) and (G) immunohistochemical staining with anti-TTR antibody. (H,I) 99 mTechnetium-pyrophosphate scan showing grade 3 myocardial radiotracer uptake (arrows): (H) axial plane; (I) coronal plane. (J) Direct DNA sequencing of exon 3 in TTR showing the Thr95Ile (c.284C>T) mutation.
Serum free light chain ratio was normal and immunofixation of the serum and urine was negative for monoclonal protein. On cardiac biopsy there was deposition of amorphous material in the interstitium, suggestive of transthyretin-related cardiac amyloidosis (ATTR-CA; Figure F,G). 99 mTechnetium-pyrophosphate scan showed intense radiotracer uptake in the LV myocardium (Figure H,I). Finally, gene sequencing confirmed the diagnosis of sporadic ATTR-CA caused by the Thr95Ile mutation (Figure J),1 the genotype and clinical phenotype of which are described here for the first time.
The LVEF was progressively decreased to 32%. The patient underwent implantable cardioverter defibrillator implantation for primary prevention. He also had paresthesia of both legs, which was diagnosed as sensorimotor polyneuropathy on nerve conduction velocity studies. He had no other neurologic abnormalities. He is currently being treated with tafamidis.2
The authors declare no conflicts of interest.