2018 年 64 巻 1 号 p. 1-8
2018 年 64 巻 1 号 p. 1-8
Heparin-induced thrombocytopenia (HIT) is an immune complication of heparin therapy caused by antibodies to complexes of platelet factor 4 (PF4) and heparin. These pathogenic antibodies against PF4/heparin bind and activate cellular FcγRIIa on platelets to induce a hypercoagulable state culminating in thrombosis. Recent studies indicate several conditions, including joint surgery, induce spontaneous HIT, which can occur without exposure to heparin. To determine the real-world evidences concerning the incidences of venous thromboembolism (VTE) after total joint arthroplasty for rheumatic disease, we conducted a multicenter cohort study (J-PSVT) designed to document the VTE and seroconversion rates of anti-PF4/heparin antibody in 34 Japanese National hospital organization (NHO) hospitals. J-PSVT indicated that prophylaxis with fondaparinux, not enoxaparin, reduces the risk of deep vein thrombosis in patients undergoing arthroplasty. Multivariate analysis revealed that dynamic mechanical thromboprophylaxis (intermittent plantar device) was an independent risk factor for seroconversion of anti-PF4/heparin antibodies, which was also confirmed by propensity-score matching. Seroconversion rates of anti-PF4/heparin antibodies were significantly reduced in rheumatoid arthritis (RA) patients compared with osteoarthritis (OA) patients, which may link with the findings that IgG fractions isolated from RA patients not OA patients contained PF4. Our study indicated that a unique profile of anti-PF4/heparin antibodies is induced by arthroplasty for rheumatic diseases.
Heparin-induced thrombocytopenia (HIT) is an immune complication of heparin therapy caused by antibodies reacting to complexes of platelet factor 4 (PF4) and heparin1). These antibodies bind and activate cellular Fc-γRIIa on platelets resulting in life-threating thrombosis1). However, recent investigations indicate that some patients develop HIT without heparin exposure2). Although this syndrome has been called “spontaneous HIT”, it seems to require preceding inflammation, tissue damage or infection2). Presumably, PF4 is bound to immunogenic complexes through binding to polyanionic bacterial surfaces or to chondroitin-sulfate released during infections or orthopedic surgery3). It has been reported that some patients receiving arthroplasty had positive tests for anti-PF4/heparin antibodies4). This review will summarize current clinical and laboratory features of PF4/heparin immunogenicity induced by arthroplasty during rheumatic diseases.
HIT is caused by antibodies that recognize complexes of PF4 and heparin. As shown in Figure 1, PF4 is released from α granules of platelet and binds to polyanions, such as heparin or those found on the surface of bacteria, and undergoes a conformational change5). This results in immunogenic PF4-heparin or PF4-bacteria complexes. After activation, B lymphocytes generate anti-PF4/heparin IgG6). These antibodies can bind to PF4-heparin complexes, forming immune complexes. The Fc domains of the IgG molecules anti-PF4/heparin antibodies bind to the platelet surface receptor, FcγRIIa, resulting in platelet activation and aggregation (Figure 1). This intravascular platelet consumption causes a decrease in platelet count and the release of platelet-derived microparticles that accelerate thrombosis. The clinical features of HIT are the occurrence of thrombocytopenia. Thrombocytopenia can manifest either as an absolute drop in platelet count or a relative decline of 30% to 50% from basal platelet counts. Thrombocytopenia in HIT is often moderate (50-70×109/L) and is typically not associated with bleeding complications7). Thrombosis is the most severe complication of HIT and can affect any vascular bed (venous thrombosis). A diagnosis of HIT cannot be made without laboratory evidence of anti-PF4/heparin antibodies8). Anti-PF4/heparin antibodies can be detected by enzyme immunoassays with high sensitivity. However, clinically evident HIT developed only a minority (2-15%) of heparin-treated patients who had anti-PF4/heparin antibodies. Diagnostic accuracy for HIT can be improved with the use of an anti-PF4/heparin enzyme immunoassay and functional test. In particular, a platelet activation assay (serotonin-release assay) is much more specific for clinically relevant antibody detection9).

Pathogenesis of Heparin-Induced Thrombocytopenia
Platelet factor 4 (PF4) that is released from platelet α-granules binds to polyanions such as heparin or polyanions on the surface of bacteria and undergoes a conformational change. This results in immunogenic PF4-polyanion (heparin) or PF4-polyanion (bacteria) complexes. After activation, B lymphocytes generate anti-PF4-polyanion IgG. The Fc parts of the IgG bind to platelet Fcγ RIIa receptors, resulting in Fcγ-receptor clustering and consequent strong platelet activation and aggregation.
Modified form of figure in the review article by Greinacher A, et al.1).
Venous thromboembolism (VTE) consists of deep vein thrombosis (DVT) and pulmonary emboli (PE), remains a problem in patients receiving total knee arthroplasty (TKA) and total hip arthroplasty (THA)10). Torigoshi et al. reported high seroconversion rates of anti-PF4/heparin antibodies in patients receiving TKA and THA11). Furthermore, they demonstrated that seroconversion of anti-PF4/heparin antibodies was associated with an increased risk of DVT in patients receiving TKA and THA12). More recently, in patients undergoing TKA or THA, thromboprophylaxis with low-molecular weight heparin (LWMH) or factor Xa inhibitors has been recommended to prevent VTE13,14). Meta-analysis also demonstrated that fondaparinux, a Xa inhibitor, showed some benefit by reducing VTE to a greater degree than LMWH in patients receiving arthroplasty15). It was also reported that anti-PF4/heparin antibodies were generated at similar frequencies in patients treated with fondaparinux and LMWH16). Despite the need for evaluating the effectiveness of these thromboprophylactic drugs and the postoperative seroconversion rates of anti-PF4/heparin antibodies in patients with rheumatic disease receiving arthroplasty, few clinical data are available.
The Japanese study of prevention and actual situation of VTE after total arthroplasty (J-PSVT) is a nationwide multicenter cohort study to obtain clinical data concerning the effectiveness of prophylactic treatment of VTE after arthroplasty in 34 Japanese National Hospital Organization (NHO) hospitals17). The major objective of this study was to determine the practical patterns of VTE prophylaxis and their outcomes including VTE and seroconversion rates of anti-PF4/heparin antibodies in Japanese patients undergoing total joint replacement (TKA or THA). Overall, 1,294 patients receiving TKA and 868 patients receiving THA were enrolled. The incidence rates of sonographically-diagnosed DVT up to postoperative day 10 (POP10) was 24.3% in patients receiving TKA, and 12.6% in patients receiving THA17). DVT rates varied among patients receiving different patterns of pharmacologic thromboprophylaxis17). The overall rates of DVT up to POD10 in patients undergoing TKA were 16.7% with fondaparinux, 26.5% with enoxaparin, 33.3% with UFH, 26.7% with other medications, and 26.6% with no medication (Table 1). Multivariate analysis showed that risk factors for postoperative VTE included older age (greater than 75 years), female sex, spinal anesthesia18), and dynamic mechanical thromboprophylaxis (DMT; intermittent plantar compression device;foot pump). Conversely, prophylaxis with fondaparinux, an Xa inhibitor, not LMWH reduced the incidence of VTE, determined by multivariate analysis (Table 2). These data suggested that the use of mechanical devices including foot pump, either alone or in combination with chemical thromboprophylaxis did not reduce the rate of VTE. Sakai et al. conducted a randomized clinical trial to compare the effectiveness of foot pump use for reducing the incidence of DVT after TKA in patients under edoxaban thromboprophylaxis19). As shown in Figure 2, the incidence of all DVTs in patients using a foot pump was 31.0% (18/58) compared with 17.7% (11/62) in patients not using a foot pump. Although there was no significant difference in the incidence of DVT between the groups, the incidence of DVT was increased in patients using a foot pump. These data indicated that foot pump use did not affect the incidence of DVT under edoxaban thromboprophylaxis in patients undergoing TKA.

Incidences of primary effectiveness outcomes in patients receiving TKA
DVT; deep vein thrombosis, UFH; unfractionated heparin, TKA; total knee arthroplasty, PE; pulmonary embolism
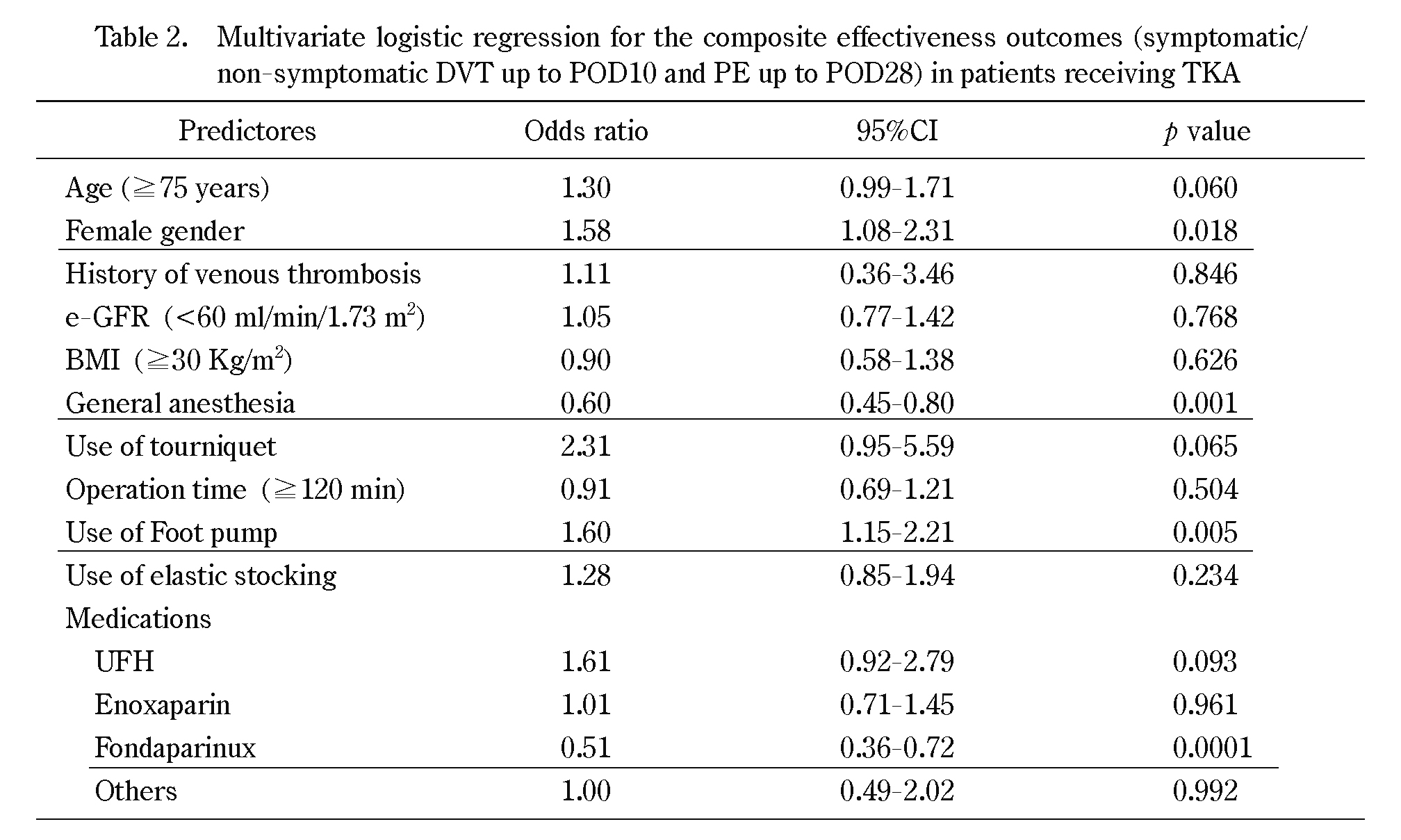
Multivariate logistic regression for the composite effectiveness outcomes (symptomatic/non-symptomatic DVT up to POD10 and PE up to POD28) in patients receiving TKA
e-GFR; estimated glomerular filtrating ratio, BMI; body mass index, 95%CI; 95%confidence interval, DVT; deep vein thrombosis, PE; pulmonary embolism, UFH; unfractionated heparin

Incidence of Postoperative VTE
We compared the incidence of postoperative total DVTs between the patients with and without use of the foot pump. Although there was no significant difference in the incidence of DVTs between the 2 groups (P = 0.089), the incidence of postoperative DVTs were higher in patients with the use of the foot pump (31.0%) compared to those without use of foot pump (17.7%).
Modified form of figure in the article by Sakai T, et al.19).
Recent investigations have demonstrated that non-pharmacological factors, such as operations, influence the seroconversion rates of anti-PF4/heparin antibodies. Nucleic acids and hyper sulfated chondroitin released by orthopedic surgery can induce anti-PF4/heparin antibodies20). We investigated the seroconversion rates of anti-PF4/heparin antibodies in patients treated with dynamic mechanical thromboprophylaxis (DMT;foot pump) plus anticoagulants using the J-PSVT database21). Multivariate analysis revealed that higher seroconversion rates were significantly associated with DMT (Table 3). We further explored the effects of DMT against the seroconversion rates of anti-PF4/heparin antibodies in patients receiving TKA, under various pharmacological thromboprophylaxis (UFH, LMVH fondaparinux, etc). As shown in Figure 3, the proportion of patients with high titers of anti-PF4/heparin antibody (OD values >1.4 units) was higher among patients treated with DMT than untreated controls under any pharmacological thromboprophylaxis regimen21). These findings suggest that DMT plays a crucial role in the postoperative immune responses against PF4 in patients undergoing arthroplasty. The mechanisms by which DMT enhances the postoperative seroconversion of anti-PF4/heparin antibodies had not be determined. A recent study of anti-PF4/heparin immunization after cardiac surgery suggested that perioperative inflammation affects the immune response22). Following arthroplasty and its associated tissue damage, DMT-induced inflammatory stimuli may modify the immune responses against PF4.

Multivariate logistic regression analysis of anti-PF4/heparin antibody seroconversion
Multivariate logistic regression was performed to identify risk factors independently associated with anti-PF4/heparin antibody seroconversion after controlling simultaneously for potential confounders. Variables, which were selected by univariate logistic regression analysis with a p value <0.2 using the chi-square test or Fisher’s exact test, were included into a multivariate logistic regression model with stepwise forward selection method with forced entry of the variables; gender, surgical type, and each pharmacological prophylaxis, which have identified as risk factors for anti-PF4/heparin antibody formation in the previous studies. A two-tailed p value <0.05 was considered significant.
PF4, platelet factor 4; CI, confidence interval; TKA, total knee arthroplasty; DMT, dynamic mechanical thromboprophylaxis; foot pump, intermittent plantar compression device; IPCD, intermittent pneumatic compression device

Seroconversion rates and proportion of patients who tested strongly positive (ELISA values ≥1.4 OD units) were calculated for patients receiving UFH, LMWH, fondaparinux, or only mechanical thromboprophylaxis after TKA. Each group was divided into 2 subgroups based on the use of DMT (intermittent plantar compression device [foot pump] or IPCD). The seroconversion rates and proportion of patients who tested strongly positive with ≥1.4 OD units were compared between the patients treated with or without DMT in each group using the χ2 test.
Modified form of figure in the article by Bito S, et al.21).
VTE is considered more likely to develop in patients with rheumatoid arthritis (RA) than osteoarthritis (OA), as inflammatory mediators in RA are associated with prothrombotic factors and RA patients are at higher risk of developing atherosclerosis23). However, few studies have compared the incidence of VTE after TKA in patients with RA and OA. Using the J-PSVT database, we compared the incidence of VTE following TKA between patients with RA and OA24). The study cohort was composed of 1084 OA patients and 204 patients with RA. PE occurred in 2 patients with OA and in none with RA. The incidence of VTE (PE+DVT) was 24.3% and 24.0% in patients with OA and RA, respectively. No significant difference in the incidence of VTE was noted between patients with RA and OA24). These findings contradict our hypothesis that thrombotic complications would be higher in patients with RA than in patients with OA. Using these patients, we compared the seroconversion rates of anti-PF4/heparin antibodies after TKA25). The postoperative seroconversion rates of anti-PF4/heparin antibodies were lower in RA patients (10.4%) than in OA patients (21.8%). We further evaluated the seroconversion rates between patients with RA and OA under various thromboprophylaxis regimes (Figure 4). Similar trends of the seroconversion rates being higher in OA patients than RA patients, were observed in patients receiving UFH, LMWH, fondaparinux, and no thromboprophylaxis agents25). We examined the circulating IgG bound to PF4 using sera from RA patients and OA patients. Serum IgG fractions isolated from healthy subjects, and patients with RA and OA were subjected anti-PF4 immunoblot analysis25). As shown in Figure 5, IgG fractions isolated from healthy individuals or OA patients did not contain PF4. Whereas PF4 bands were evident in the IgG fractions isolated from RA patients. These results indicated that circulating IgG was bound to PF4 in the majority of RA patients, suggesting preexisting immune responses against PF4 during the chromic immune-mediated inflammatory processes of RA. These responses may interfere with anti-PF4/heparin B cell differentiation through immune-tolerance mechanisms. These findings suggest that the autoimmune-like features of anti-PF4/heparin antibody production26) cannot be applied to the postoperative induction of this antibody in RA.

Anti-PF4/heparin seroconversion rates and proportion of patients who tested strongly positive in patients who underwent TKA. Seroconversion rate and the proportion of patients who tested positive (ELISA values ≥ 0.4 optical density (OD) units) were calculated for patients receiving unfractionated heparin (UFH), low-molecular-weight heparin (LMWH), fondaparinux, or only mechanical thromboprophylaxis (no anticoagulant) after TKA. Each group was divided into two subgroups based on the primary diseases (RA or OA). The seroconversion rates were compared between the RA patients or OA patients in each group using the chi-square test.
Modified form of figure in the article by Izumi M, et al.25).

Anti-PF4 immunoblot analysis using the heparin-binding fractions isolated from RA or OA patient sera. Heparin-binding fractions isolated from RA and OA patient sera using heparin-Sepharose-4B beads were subjected to anti-PF4 immunoblot analysis. PF4 bands (MW=7,800) were detected in the heparin-binding fractions from RA patients or OA patients under reducing conditions.
Modified form of figure in the article by Izumi M, et al.25).
We conducted a multicenter cohort study to assess VTE risk and seroconversion rates of anti-PF4/heparin antibodies in patients undergoing arthroplasty of the knee or hip. In this study, we found factors, associated with increased risk for VTE and seroconversion of anti-PF4/heparin antibodies in patients receiving TKA or THA. Chemoprophylaxis with a Xa inhibitor, fondaparinux, was found to be a VTE protective factor. Conversely dynamic mechanical thromboprophylaxis (DMT) and spinal anesthesia were found to be risk factors for VTE. DMT increased the postoperative seroconversion rates of anti-PF4/heparin antibodies, whereas reduced seroconversion rates of anti-PF4/heparin antibodies were observed in RA patients. Our data provide a unique profile of anti-PF4/heparin antibodies induced by arthroplasty without exposure to heparin. These paradoxical serological features of anti-PF4/heparin antibodies should be carefully considered for preventing thromboembolic events during perioperative periods in patients receiving arthroplasty.