2016 Volume 85 Issue 2 Pages 148-153
2016 Volume 85 Issue 2 Pages 148-153
The aim of this work was to study the effect of substrate disinfection and application of plant growth-promoting rhizobacteria (Bacillus subtilis) on the yield, quality and safety of watercress grown in a floating system. Substrate disinfection had a positive effect on plant development because it increased the shoot antioxidant capacity and general plant growth and decreased the colony-forming units of molds. In turn, inoculation with B. subtilis increased the antioxidant capacity but decreased the chlorophyll a, chlorophyll b, and carotenoid contents and did not affect the rest of the parameters measured. In conclusion, our results showed that the effects of substrate disinfection were more pronounced than those obtained by B. subtilis inoculation, suggesting that it would be more convenient to reserve the use of plant growth-promoting rhizobacteria for other conditions, such as abiotic stress.
Changes in lifestyle and eating habits have led to the growing popularity of fresh-cut vegetables. Among them, watercress is considered as a valuable food product in the fresh salads industry, for its supposed high content of health-promoting compounds such as antioxidants and phenolics (Niñirola et al., 2014). Among the different systems available for the cultivation of watercress, the floating systems (FS) are an easy and profitable growing technique. With FS, seeded trays float continuously on a water-bed or nutrient solution (NS) during plant growth (Fontana and Nicola, 2008). One of the main advantages of FS is the possibility of directly influencing the nutritional status of plants by changing the composition of the NS, facilitating the production of vegetables of prime quality and sometimes fulfilling specific dietetic requirements.
In light of the problems associated with the use of synthetic chemicals in agriculture, the use of plant growth-promoting rhizobacteria (PGPR) in agriculture is gaining in importance and acceptance worldwide, and appears to be a trend for the future (Raj et al., 2005). Significant increases in the growth and yield of important agronomic crops in response to inoculation with PGPR have been widely reported (e.g. Barreto-Figuereido et al., 2010, and citations therein). Among strains that show growth-promoting activity, species belonging to the genera Pseudomonas and Bacillus have been the most extensively studied. Recently, Kumar et al. (2011) reviewed the literature on the principal growth promotion mechanisms of Bacillus strains and found that they include the production of growth-stimulating phytohormones, phosphate solubilization and mobilization, siderophore production, antibiosis (including the production of antibiotics, the inhibition of plant ethylene synthesis and the induction of plant systemic resistance to pathogens). It seems very likely that the above-mentioned plant growth promotion effects are a result of the combined action of two or more of these mechanisms. Furthermore, the widely established knowledge of its controlling effect against pests and diseases would explain the commercial exploitation of different strains of Bacillus subtilis as biocontrol agents (Kumar et al., 2011).
PGPR and their formulations are commonly applied as a seed treatment, soil amendment or root dip in bacterial suspension before transplanting (Podile and Kishore, 2006). In addition, PGPR can be successfully incorporated into soilless media in vegetable transplant production systems (Yan et al., 2003). The use of PGPR in soilless culture systems (SCS) is increasing as it is considered to induce resistance of plants to biotic and abiotic stresses and to increase plant growth and yield (Gül et al., 2008). Some negative effects of PGPR application on the growth of raspberry ‘Heritage’ have been found, which could be related to a high level of auxin production or some other unknown secondary metabolites (Orhan et al., 2006). However, very little attention has been paid to the effects of PGPR application on the quality at harvest of baby leaf vegetables (BLV) produced in SCS. Improving quality at harvest is associated with beneficial changes in the postharvest maintenance of product quality, which is why microorganisms beneficial to the rhizosphere could be considered as a preharvest biotic factor that affects fruit and vegetable quality (Olalde-Portugal and Mena-Violante, 2008). Some soil microflora factors can affect the success of PGPR inoculation. In particular, soil disinfection can unbalance microbial communities in the soil (Garbaye, 1991) with rather unpredictable results for bacterial inoculation. To our knowledge, little has been published on whether PGPR affect the inherent quality of fresh vegetables, particularly in the case of BLV. In addition, no research has been carried out to investigate the eventual effects of PGPR on the safety parameters usually considered at harvest, such as microbial spoilage. The aim of this work was thus to study the effect of substrate disinfection and Bacillus subtilis application on the yield, quality and safety of watercress grown in an FS, called floating growing system (FGS).
The experiment was conducted in the Experimental Centre of DISAFA (44°53'11.67''N; 7°41'7.00''E - 231 m a.s.l.) in Tetti Frati, Carmagnola (TO), Italy, in a greenhouse. Maximum, minimum, and mean temperatures during the growing season were 43.0°C, 17.0°C, and 29.1°C, respectively. The plant material used was a commercial cultivar of watercress (Nasturtium officinale R. Br.) ‘Large Leaf’ (Tozer Seeds Co., Cobham, UK). The experiment consisted of growing plants in 60-cell styrofoam trays (0.51 m × 0.30 m, with cells of 0.044 m upper and 0.025 m lower diameters, respectively) containing a specific peat-based horticultural medium (Neuhaus Huminsubstrat N17; Klasmann-Deilmann® GmbH, Geeste, Groß-Hesepe, Germany) floating in a NS. The sown trays were placed in a plastic greenhouse until seed germination. Four days after sowing, the trays were moved into a lab-scale pilot plant equipped with 3 benches, each one split into 4 separated flotation beds (2.50 m × 1.40 m; 0.15 m depth) and filled with 200 L of a 40/60 N-NO3−/N-NH4+ NS composed of (all in mM·L−1): 12 N, 2 P, 6 K, 2 Mg, and 2.5 Ca (Fontana and Nicola, 2008; Nicola et al., 2004, 2007). Then, Lysodin® Multimix formulation of microelements (Intrachem Production S.r.l., Grassobbio, Bergamo, Italy) was added to the NS at a dose of 0.30 g·L−1. The pH and the electrical conductivity of the NS were monitored weekly and kept close to ca. 5.5 and 2.0 dS·m−1, respectively. The NS was aerated by a compressor connected to a dripline tube positioned in each flotation bed, to maintain levels of dissolved oxygen close to ca. 5 ppm throughout the growing cycle. The final plant density was 300 plants per tray (ca. 1961 plants·m−2). Harvesting took place after 24 days of cultivation.
The experiment followed a randomized complete block design (RCBD) with three replicates per treatment. Each treatment was placed in a flotation bed containing 12 trays.
Bacterial strain and inoculationTwo factors were considered, disinfection of the substrate and inoculation with B. subtilis. Substrate disinfection was carried out in a flow steam at 100°C for 45 min. A total of 50% of the substrate used in the assay was disinfected. For bacterial inoculation (BI), the commercial product Larminar® (1012 CFU·g−1 of B. subtilis strain AP-01; Agrimor, Agricultura Moderna S.A., Madrid, Spain) was used. Inoculation was performed twice: for the first inoculation, one day before sowing, 50% of the disinfected substrate (DS) and 50% of non-disinfected substrate (NDS) were inoculated with Larminar® at a dose of 0.5 kg·m−3. All of the seeds used were disinfected in 20% NaClO (w/v) and rinsed three times with sterile deionized water. A total of 50% of the disinfected seeds were inoculated by immersion for 1 h in a B. subtilis suspension at a concentration of 108 CFU·mL−1 in 0.9% of NaCl solution (w/v) obtained from Larminar® in Plate Count Agar (PCA) (Fluka Analytical, Sigma-Aldrich S.r.l., Milan, Italy). In the case of non-inoculated (NBI) seeds, they were kept for 1 h in 0.9% NaCl. For the second inoculation, eleven days after sowing, a re-inoculation was performed by placing the inoculated trays (substrate and seeds) on a solution containing 0.167% Larminar®/water (w/v).
Biometrical measurements and phytochemical analysesWhole plants were harvested and divided into aerial and root parts. The biometrical measurements recorded were: fresh and dry weights of the shoots in 30 plants per treatment and per block, shoot height (SH), leaf number (LN) per plant, leaf area (LA) using ImageJ 1.47v picture analyzer developed at the National Institutes of Health (Bethesda, Maryland, USA), leaf color (LC) using a CR10 colorimeter (Konica-Minolta Sensing Inc., Osaka, Japan), relative chlorophyll content (RCC) using a chlorophyllmeter (Minolta SPAD-502; Konica-Minolta Sensing Inc.), and fresh and dry weights of roots in 12 plants per treatment and per block. These measurements enabled the following parameters to be calculated: specific leaf area (SLA), dry matter of shoot (SDM) and roots (RDM), and hue angle (H*) and chroma (C*), as described by Niñirola et al. (2014). Phytochemical analyses were conducted on the shoots. Ascorbic acid and dehydroascorbic acid were determined as described by Zapata and Dufour (1992) from 10 g of frozen tissue per sample. Antioxidant capacity (AC) was performed following the procedures of Benzie and Strain (1996) using the ferric reducing ability of plasma (FRAP) assay as a measure of AC from 2 g of frozen tissue per sample. Total phenolics (TP) were determined using the Folin-Ciocalteu procedure based on the method of Singleton and Rossi (1965) from 2 g of frozen tissue per sample. Chlorophyll a (Chl a), chlorophyll b (Chl b), and carotenoids (Car) were determined according to the Lichtenthaler and Wellburn (1983) method from 1 g of frozen tissue per sample. Browning potential (BP) and soluble o-quinone (So-Q) were determined based on the method of Couture et al. (1993) and Loaiza-Velarde and Saltveit (2001) from 5 g of frozen tissue per sample. Peroxidase (POD), polyphenol oxidase (PPO), and phenylalanine ammonia lyase (PAL) activities were determined from 0.5 g of frozen tissue per sample. POD activity was determined as described by Nickel and Cunningham (1969) by the increase in absorbance, PPO activity was determined as described by Degl’Innocenti et al. (2005), and PAL activity was determined as described by Campos et al. (2004) and Degl’Innocenti et al. (2005). Nitrate (NO3−) was determined on the shoots using a refractometric kit (Merck Reflectoquant RQflex2©; Merck KGaA, Darmstadt, Germany), following the manufacturer’s instructions from 10 g of frozen tissue per sample.
Microbiological analysisTotal bacterial count (TBC) was determined by PCA, while the mold and yeast counts (MC and YC, respectively) were determined using Yeast Extract Glucose Chloramphenicol Agar (Fluka Analytical). For each sample, 25 g of fresh tissue from the aerial part was used. The TBC was performed after incubation at 30°C for 48 h, while MC and YC were performed after incubation at 30°C for 5 d.
Statistical analysisData were analyzed using Statgraphics Plus. Analysis of variance was performed considering the factorial design substrate disinfection (DS and NDS) × bacterial inoculation (BI and NBI) in RCBD. When interactions were significant, they were included in the ANOVA, and the least significant difference (LSD) test was performed to separate means.
The growth and development of watercress were considered adequate for all of the treatments, although plants grown in BI had some minor post-emergence problems due to damping-off (data not shown). A significant interaction between the disinfection and inoculation was found for SH (Table 1). SH in the DS × NBI treatment was significantly higher than in NDS × NBI (Fig. 1a). In addition, the disinfection also affected LN, RCC, SLA, and C* (Table 1). Plants growing in DS conditions had higher LN, RCC, and SLA and lower C* than those grown in NDS (Table 1), producing plants with grayish green leaves. The higher growth obtained in DS conditions agrees with the findings of Saubidet et al. (2002), who recorded higher growth for wheat plants grown in pots filled with disinfected substrate than in non-disinfected substrate. This was probably due to the release of nutrients such as N and P in the disinfected substrate after natural reinfection, which would increase yields in the short term (Paul and Clark, 1989). As regards B. subtilis inoculation, no significant differences were found for any of the measured parameters with respect to the non-inoculated treatment. Similar results were obtained for B. subtilis by Corrêa et al. (2010) and Balanza et al. (2012) in a hydroponic lettuce crop. Nevertheless, some studies have found a positive effect of PGPR application on shoot growth in a hydroponic culture (Urashima and Hori, 2003), particularly when plants were grown under stress conditions, since PGPR application could have a function on abiotic stress relief (Liu et al., 2010).

Influence at harvest of disinfected substrate (DS) and non-disinfected substrate (NDS), and bacterial inoculation (BI) and no bacterial inoculation (NBI) on the growth parameters [shoot height (SH), leaf number (LN) per plant, relative chlorophyll content (RCC), yield, specific leaf area (SLA), dry matter from shoot (SDM) and roots (RDM), hue angle (H*), chroma (C*) and nitrate content (NO3−)] of watercress cultivated in a floating growing system (FGS).

Influence at harvest of disinfected substrate (DS) and non-disinfected substrate (NDS), and bacterial inoculation (BI) and no bacterial inoculation (NBI) on shoot height (SH) (a) and phenylalanine ammonia lyase (PAL) (b) in watercress cultivated in a floating growing system (FGS). Values are the mean of three replicates and vertical lines are the least significant difference (LSD) intervals at P ≤ 0.05.
Disinfection of the substrate affected the shoot nitrate content, which significantly increased by 80.5% (Table 1). This result agrees with that observed by Saubidet et al. (2002) in wheat, in which the NO3− concentration in wheat tops was higher in plants grown in disinfected soil than in those grown in non-disinfected soil. There were no differences in the ion contents between BI and NBI (Table 1). Our results also agree with those obtained in a lettuce crop by Balanza et al. (2012), who did not find any difference in nitrate content between non-inoculated plants and plants inoculated with B. subtilis. However, gains in nutrition for plants inoculated with rhizobacteria have been demonstrated as a benefit of the presence of this group of microorganisms in the rhizosphere (Barreto-Figuereido et al., 2010).
Antioxidants and pigmentsThe AC, TP, Chl a, Chl b, and Car of plants growing in DS conditions increased by 36.4, 19.3, 20.5, 18.7, and 23.5% (Table 2), respectively, with respect to NDS plants. As regards inoculation, BI increased AC by 27.8% and decreased Chl a, Chl b, and Car by 20.4, 18.7, and 23.5%, respectively, compared with NBI (Table 2). Not many studies have focused on the effect of PGPR on the accumulation of antioxidants and pigments, while most of those that have been reported looked at the use of PGPR strains to alleviate the effect of abiotic stress. Heidari and Golpayegani (2012) demonstrated that the application of rhizobacteria improved the antioxidant and photosynthetic pigments of basil plants under water stress and Han and Lee (2005) showed that PGPR increased the chlorophyll content and decreased enzyme activity in plants under salinity stress.
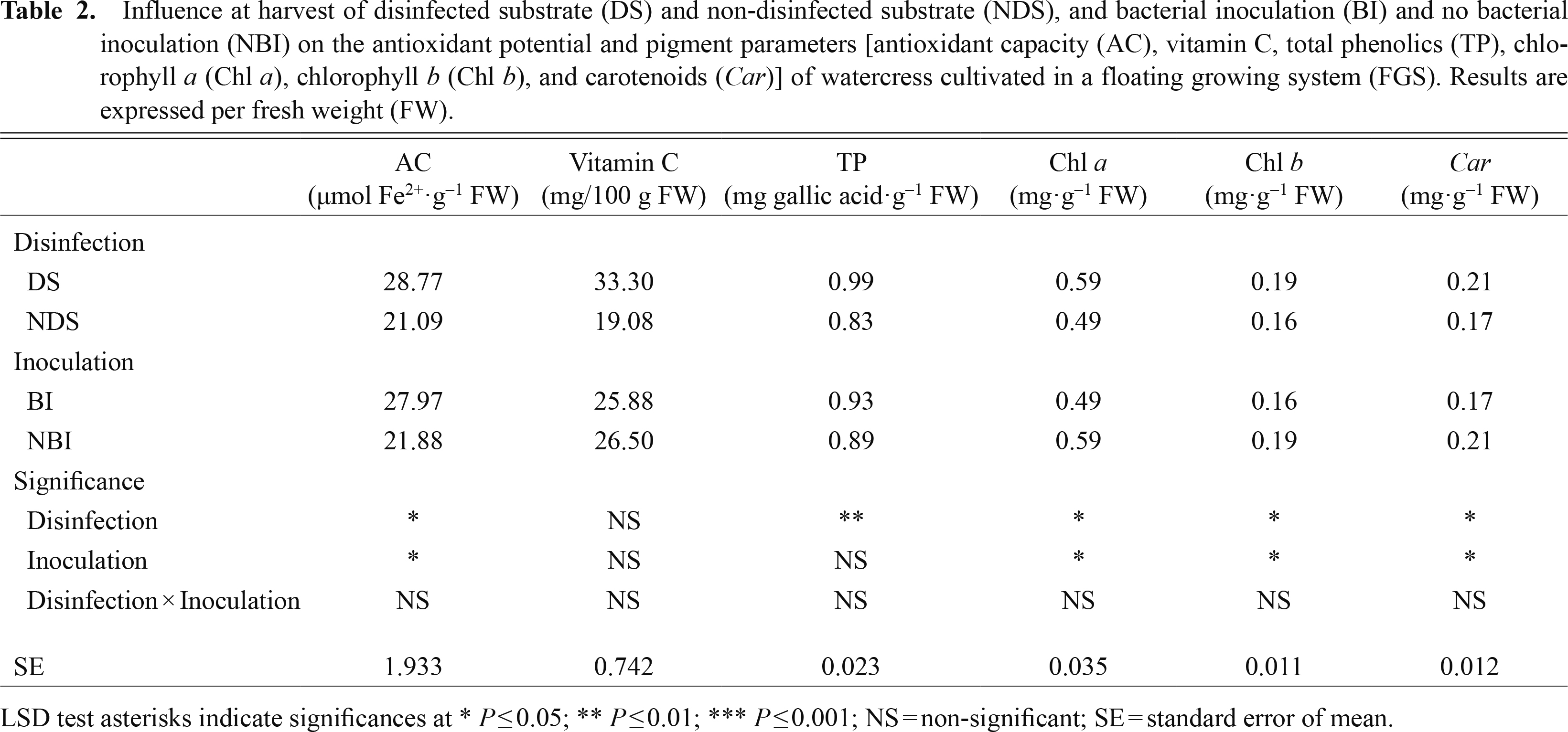
Influence at harvest of disinfected substrate (DS) and non-disinfected substrate (NDS), and bacterial inoculation (BI) and no bacterial inoculation (NBI) on the antioxidant potential and pigment parameters [antioxidant capacity (AC), vitamin C, total phenolics (TP), chlorophyll a (Chl a), chlorophyll b (Chl b), and carotenoids (Car)] of watercress cultivated in a floating growing system (FGS). Results are expressed per fresh weight (FW).
The measured parameters are related to the enzymatic activity produced by cuts or injury to plant tissues. Neither disinfection nor inoculation affected the reaction of watercress to the damage and cuts that occurred during harvest and no significant differences were observed for these factors in any parameter separately. The significant interaction of two factors was only observed in the PAL activity, when antagonistic behavior was evident (Table 3), the effect of BI depending on whether the substrate had been disinfected or not (Fig. 1b). In DS conditions, NBI plants showed significantly higher PAL activity than BI plants, whereas in NDS conditions, BI plants had significantly higher values for the activity of the enzyme than NBI plants (Fig. 1b). Vivekananthan et al. (2006) suggested that the preharvest application of biocontrol agents may help overcome pre- and postharvest infection by increasing levels of defense-related enzymes and phenolic substances.

Influence at harvest of disinfected substrate (DS) and non-disinfected substrate (NDS), and bacterial inoculation (BI) and no bacterial inoculation (NBI) on the enzymatic browning parameters and on the microbial growth [browning potential (BP), soluble o-quinone (So-Q), peroxidase (POD), polyphenol oxidase (PPO), phenylalanine ammonia lyase (PAL), total bacterial count (TBC), mold count (MC), and yeast count (YC)] of watercress cultivated in a floating growing system (FGS). Results are expressed per fresh weight (FW).
Disinfection of the substrate only affected mold CFU in watercress at harvest (Table 3). The MC was significantly higher under the NDS factor effect, demonstrating that disinfection provided slight control of the mold population. In addition, there were no differences in the TBC between BI and NBI (Table 3), meaning that inoculation neither of the seeds nor of the substrate would affect the microbiological quality of the final product.
In conclusion, disinfection of the substrate had a positive effect on the development of the watercress because it increased the shoot AC and general plant growth and decreased the CFU of molds. Inoculation with B. subtilis had a less pronounced effect because it increased the AC and decreased the contents of Chl a, Chl b, and Car.