2013 Volume 38 Issue 1 Pages 1-9
2013 Volume 38 Issue 1 Pages 1-9
Neonicotinoids ushered in a new era of crop protection, and several molecules headed up a billion-dollar industry. As one of the largest agricultural and most populous countries, China plays an essential role in the development of neonicotinoids. This review delves into the production, application and innovation of neonicotinoids throughout China.
China is a huge traditional agricultural country with a total of 1219 billion square meters under pressure annually by insect pests, weeds and plant pathogenic fungi. There are 1648 types of agricultural pests in China, including 724 plant diseases, 838 insects (mites), 64 weeds, and 22 rodents.1) As a developing country, China has rapidly expanded its use of crop and non-crop pesticides, particularly over the past decade; pesticide output has nearly doubled along with the varieties used. China is now the largest producer and exporter of pesticides in the world. It produces about 500 varieties of pesticides as well as the largest quantities. Production reached 2.65 million tons in 2011. Its export and import volumes reached 796,000 and 53,000 tones, respectively, in 2011. Its export and import turnover reached to 2.91 billion $ in 2011. In addition, China is also an enormous pesticide consumer, with sustainable tillage exceeding 150 million hectares annually and annual demand exceeding 319,400 tones.2–4)
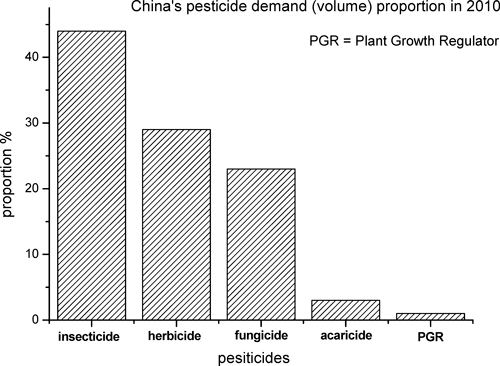
Insecticides account for 44% of all pesticides used across the China. Organophosphate and carbamate insecticides are gradually being removed from the domestic market, and, with this development, the demand for highly efficient and low-toxicity neonicotinoids has been increasing. Neonicotinoids, represented by imidacloprid, are the most commonly used insecticide, with an annual expenditure of $1.9 billion, accounting for 24% of the total insecticide market.5) Eight neonicotinoids, acetamiprid, clothianidin, dinotefuran, imidacloprid, nitenpyram, thiacloprid, thiamethoxam and sulfoxaflor, have been commercialized and used worldwide (Fig. 2). As a major producer, China plays an extremely important role as a worldwide neonicotinoid supplier. The survival of many large pesticide companies depend significant on neonicotinoids, in particular, imidacloprid. Meanwhile, in combating pests, particularly in the rice field, neonicotinoids play an irreplaceable role. Thus, studies on neonicotinoids are attracting attention in China today. Under financial support of some major projects such as “973” or “863” program, research related to neonicotinoids is underway in some universities or institutes. These researches mainly focus on R&D of novel neonicotinoids, MoA, and toxicology. The perspective presented here provides an overall view of neonicotinoids in China, including the production, application, and innovation of this extremely effective pesticide.
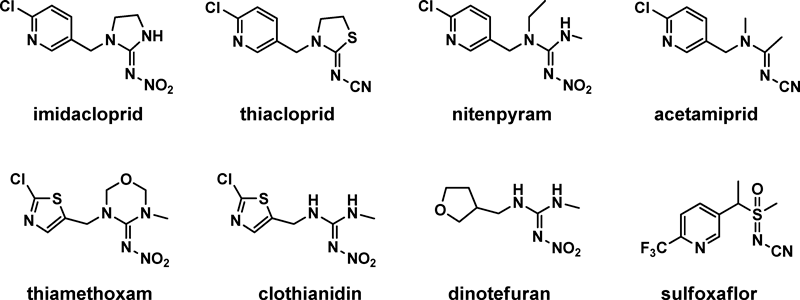
Presently, six types of neonicotinoids are registered for production on the domestic market. Whereas imidacloprid, acetamiprid, nitenpyram, and thiacloprid are developed by companies outside China, paichongding and imidaclothiz are newly developed species with independent intellectual property rights. Among all neonicotinoids, imidacloprid is the most common, produced in the largest quantities by the most manufacturers. Currently, there are 36 domestic manufacturers of imidacloprid (52 Active Ingredients (AI) manufacturers). China is the largest producing and exporting country of imidacloprid, and its annual output and export are 14,000 and 8000 tons, respectively. The second largest neonicotinoid in terms of output is acetamiprid with 34 registered manufacturers and an annual output of 8000 tons AI. There are eight registered manufacturers for nitenpyram, with an output of 100 tons, and 4 registered manufacturers for thiacloprid, with an output of less than 1000 tons.
1.1. Related policies for neonicotinoids in ChinaLimits on production are being established by the Chinese Ministry of Industry and Information Technology (MIIT) because the current production rates for imidacloprid and acetamiprid are considered to be excessive. The existing factories will be regulated, and those that fail to meet the standards set for waste solids, water and gases will be shut down.
Therefore, the China Crop Protection Industry Association (CCPIA) launched and established an Imidacloprid Products Collaborative Group and proposed to MIIT a set of guidelines, entitled “The Permission for Imidacloprid AI Production.” The document contains information on manufacturing locations, technology, equipment, production scale, resource consumption, comprehensive use, environmental protection, safety, sanitation, quality insurance, raw material quality, management, supervision, and supplementary data.
The implementation of these guidelines was expected to control redundant construction, reduce the number of factories, and scale the current producers. The guidelines should be appropriate for not only imidacloprid but also other neonicotinoid insecticides.
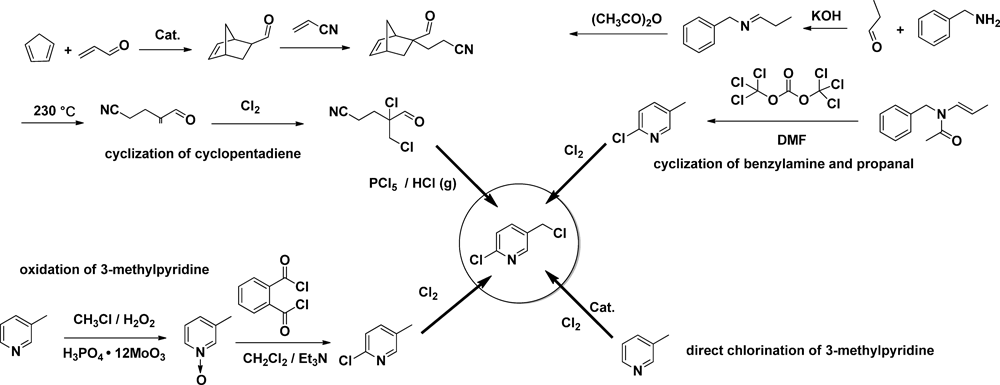
1.2. Preparative technology and processes for major neonicotinoid insecticidesUp to now, the major neonicotinoid insecticides are imidacloprid and acetamiprid, while 2-chloro-5-chloromethylpyridine is the common intermediate except for imidaclothiz. Nearly all of the preparation technologies are based on this particular intermediate, which affects the final price. Before proceeding, it is important to clarify how the 2-chloro-5-chloromethylpyridine has been produced.
There are four methods for producing 2-chloro-5-chloromethylpyridine: 1) cyclization of cyclopentadiene; 2) oxidation of 3-methylpyridine; 3) cyclization of benzylamine and propanal; and 4) direct chlorination of 3-methylpyridine. The first method is the least expensive, and most manufactures use this method. However, the severe environmental pollution associated with this method, which produces large amounts of solid waste, remains a challenge. At present, with the increase of starting material 3-methylpyridine and the use of byproducts for other pesticides, the method based on 3-methylpyridine is gradually becoming dominant.6)
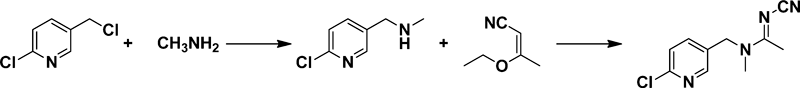
1.2.1. Synthesis of imidaclopridIn 1992, imidacloprid AI obtained a temporary pesticide registration from the Chinese Ministry of Agriculture, which permitted the production and sale of imidacloprid on the domestic market. Two years later, Jiangsu Wuxian Pesticide Company produced the first batch of imidacloprid and sold it under the trademark “Yibianjing.” However, the price of imidacloprid AI was continuously high at that time, reaching 0.2 million $ per ton. Afterward, with the discoveries of novel synthetic routes, improvement of the production process, and increasing number of manufactures, the imidacloprid price gradually fell, approaching 0.02 $ million per ton. Although nearly two decades have passed since imidacloprid was first used in China in 1992, the competitive production has resulted in enormous profits to the manufacturers and Chinese government.
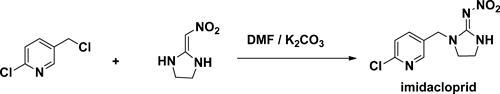
Imidacloprid was prepared by condensing 2-chloro-5-chloromethylpyridine and 2-(nitroimino)imidazolidine in the base.
To phase transfer catalysis liquid was added 2-chloro-5-chloromethylpyridine and methylamine aqueous solution (40%), the obtained product N-methyl-2-chloro-5-methylaminopyridine was then reacted with ethanol and ethyl ethyl N-cyanoacetimidate. After completion, the mixture was cooled and filtered to afford the crystalized acetamiprid.
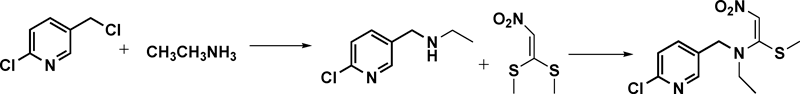
1,1-Dimethylthio-2-nitroethene in hot ethanol was added to a solution of N-ethyl-6-chloro-3-pyridinemethylamine in anhydrous ethanol, which was obtained by the reaction of 2-chloro-5-chloromethylpyridine and ethylamine in ethanol. N-[(6-Chloro-3-pyridyl)methyl]-N-ethyl-1-methylthio-2-nitroethylene amine was obtained after workup.
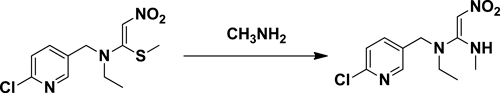
The above intermediate in hot ethanol was added to a solution of 40% methylamine in ethanol. Nitenpyram was afforded after workup.
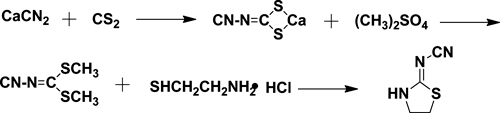
2-Chloro-5-chloromethylpyridine was added to a mixture of 2-cyanoimino-1,3-thiazolidine, potassium carbonate and n-butyl alcohol. The mixture was stirred, cooled, filtered, washed and dried to get solid thiacloprid. The intermediate 2-cyanoimino-1,3-thiazolidine was obtained by the reaction of 2-aminoethanethiol hydrochloride and 2-dimethyl cyanoimidodithiocarbonate, which was synthesized by calcium cyanamide, carbon disulfide and dimethyl sulfate.
Imidacloprid has the largest sales and application amount among neonicotinoids, accounting for around 65% of the total. As the largest supplier of imidacloprid, China has much superiority in producing imidacloprid, such as mature and efficient technology, stable and sufficient raw material supply, low labor cost. Due to the significant resistant to imidacloprid across the China, the domestic market of imidacloprid has been shrinking in recent years. Nowadays, China produce the largest amount of imidacloprid AI in the world, whose output has reached 13,500 tons in 2010 including export amount of 8000 tons, while the import amount was only tens of tons.
Around 50% of imidacloprid technical producers locate in Jiangsu province. The major imidacloprid manufactures included Jiangsu Kwin Group Co., Ltd. with a production capacity of 5000 tons (AI), Shandong United Pesticide Industry Co., Ltd. with a production capacity of 1000 tons (AI), Nanjing Red Sun Co., Ltd. with a production capacity of 1200 tons (AI), Jiangsu Changlong Chemicals Co., Ltd. with a production capacity of 2500 tons (AI), Jiangsu Changqing Agrochemical Co., Ltd. with a production capacity of 3000 tons (AI), Qingdao Hailier Pharmaceutical Co., Ltd. with a production capacity of 4000 tons (AI) and a 4000 tons AI production units is under construction, Jiangsu Yangnong Chemical Group Co., Ltd. with a production capacity of 500 tons (AI), Anhui Huaxing Chemical Industry Co., Ltd., Jiangsu Fengshan Group Co., Ltd., etc. The total annual production capacity in China was 25,000 tons (AI).
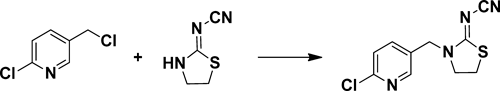
In China, the total acetamiprid productivity was 8000 tons (AI) per year, while nearly 5000 tons were exported and tens of tons were imported.
The major manufacturers for acetamiprid are: Jiangsu Changlong Chemicals Co., Ltd., Anhui Huaxing Chemical Industry Co., Ltd., Jiangsu Huangma Agrochemicals Co., Ltd., Jiangsu Kwin Group Co., Ltd., Jiangsu Fengshan Group Co., Ltd., Jiangsu Changqing Agrochemical Co., Ltd., Shandong United Pesticide Industry Co., Ltd., Qingdao Kaiyuanxiang Chemical Co., Ltd., etc.
1.3.3. ThiaclopridThe major manufacturers for thiacloprid AI are: Tianjing Xingguang pesticide factory, Jiangsu Flag Chemical Co., Ltd., Shandong United Pesticide Industry Co.,Ltd., Jiangsu Liming Chemical Industry Co., Ltd.
1.3.4. NitenpyramThe manufacturers for nitenpyram AI are: Jiangsu Changlong Chemicals Co., Ltd., Qingdao Kaiyuanxiang Chemical Co., Ltd., Shandong United Pesticide Industry Co., Ltd., Hebei Wuqiao Pesticide Co., Ltd., Hubei Xianlong Chemicak Co., Ltd. In the year of 2011, 13 companies companies approved the nitenpyram registration. They are Beijing Yoloo Agrochemical Co., Ltd., Shanxi Biaozheng Crop Science Co., Ltd., Shandong United Agrochemical Industry Co., Ltd., Nanjing Huazhou Pharmaceutical Co., Ltd., Lianyungang Liben Agrochemical Co., Ltd., Jiangxi Sprin Agri-chemicals Co., Ltd., Shanxi Welch Crop Protection Co., Ltd., Lianyungang Liben Agrochemical Co., Ltd., Shanxi Thompson Biotechnology Co., Ltd., Shanxi Sunger Road Bio-Science Co., Ltd., Chengdu Royal Crop Science Co., Ltd., Qingdao Hailir Pesticides and Chemicals Co., Ltd., Chengdu West Aidi Crop Science Co., Ltd.
1.3.5. ClothianidinHunan Bide Biochemical Co. Ltd. and Jiangsu Flag Chemical Co., Ltd. registered clothianidin TC product on October 12th, 2011 and December 13th, 2011, respectively.
Currently, the neonicotinoids insecticides are registered majorly to cure rice plant hopper, aphids, leafhopper, whitefly, thrips, etc. in rice, wheat, cotton, fruit tree, vegetable, orange and flowers. Rice is the largest application scope of imidacloprid. In addition, thiacloprid is used to prevent longicorn beetle in forests and imidacloprid is used to cure louse on pets etc.
Different characters of the AIs cause different formulations. The common ones are EC, WP, ME, EW, SP, SC, SP, WG and EA. Due to the limitation on EC by the government, the current trend is toward water-based EW, WG, SC and high-contented formulations, which are environmental friendly.
2.1. EC (Emulsifible concentrate)EC imidacloprid is in 1%, 2%, 2–5%, 5%, etc.
EC acetamiprid is in 3% and 5%.
Due to the catalogues update, formulation development limitation and the low solubility in common solvents, nitenpyram and thiamethoxam are seldom processed as EC.
2.2. ME (Micro-emulsion)ME imidacloprid is in 5%, 30%, 400 g/L, 45%, etc.
ME acetamiprid is in 3%, 5%, 6%, 10%, etc.
Due to the high water solubility, nitenpyram is seldom processed as ME.
2.3. EW (Emulsion, oilin water)Due to the low solubility in common solvents, imidacloprid and thiamethoxam are usually not processed as EW, while nitenpyram is usually not processed as EW either for its high water solubility.
EW acetamiprid is in 10%.
2.4. WP (Wetable powder)WP imidacloprid is in 2–5%, 5%, 20%, 25%, 30%, 50%, 70%, etc.
WP nitenpyram is in 20%.
2.5. SP (Water soluble powder)SP acetamiprid is in 20%.
2.6. SC (Suspension concentrate)SC imidacloprid is from 5% to 600 g/L, mostly in 35%. It’s developing as 600 g/L AI in these two years.
SC thiacloprid is in 40% and 48%.
SC acetamiprid is in 5% and 15%.
2.7. SL (Soluble concentrate)SL imidacloprid is in 5%, 6%, 10% and 20%.
SL acetamiprid mostly is in 20%.
2.8. WG (Water dispersible granules)WG is the most developed formulations over recent years.
WG imidacloprid is in 65% and 70%.
WG acetamiprid is in 20%, 25%, 26%, 33%, 36%, 40%, 50%, 60%, 70%, etc. The most common ones are 40% and 70% WG.
2.9. OthersBesides above, nitenpyram has 10% AS, thiacloprid has 2% CS and 1% CJ.
2.10. The development of formulationsIn order to meet the requirement for environmental protection and combining with the advent of novel types of formulas, some new formulas with high AI concentration appear in the maket. These include 80% WP, 600 g/L SC and 450 g/L ME imidacloprid; 70–80% WG and 70% WP acetamiprid and 70–80% WG thiacloprid.
After the invention of imidacloprid, research on neonicotinoids increased significantly across China. Most of it was focused on seeking new generations of neonicotinoids. Furthermore, the Chinese government promotes and funds research into neonicotinoids. Currently, a considerable amount of research is being conducted relative to the design and development of novel neonicotinoid molecules and related chemistry. In this section, we report our own results in this field and describe the work of other research groups.
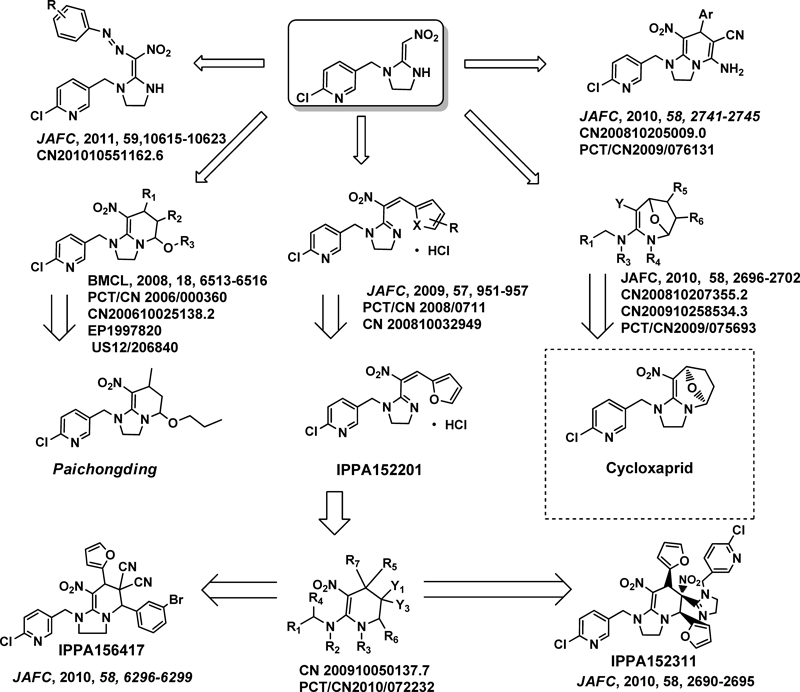
Professor Zhong Li from East China University of Science and Technology (ECUST) is investigating a new type of neonicotinoid called cis-neonicotinoids.7,8) It is well established that trans/cis isomers differ significantly in biological activity, toxicity, and metabolic properties, which emphasizes the value of addressing trans/cis chemistry in neonicotinoid development. The rationale for their molecular design involved the use of fused heterocycles or bulky groups to fix the direction of the nitro or cyano group, forcing them in the same direction as heteroaromatic moiety (cis-configuration). Centering on cis-neonicotinoids, 12 series and more than 600 compounds have been synthesized (Fig. 9). Certain compounds possess worthwhile insecticidal activities, thereby indicating potential. Some compounds have excellent insecticidal activities against homoptera insects and lepidoptera insects, such as cowpea aphids, Nephotettix bipunctatus (Fabricius), brown planthopper (Nilaparvata lugens), armyworm (Pseudaletia separate Walker) and the diamondback moth (Plutella xylostella). Most importantly, some compounds exhibited noteworthy activities against imidacloprid-resistant insects, which might be used in the control of resistant strains. Two cis-neonicotinoids, paichongding and cycloxaprid might soon be available on the Chinese market. Disappointingly, IPPA152201 was water unstable and failed to meet insecticidal requirements. Interestingly, IPPA152201 exhibited excellent activity on Taeniae and Coccidia in either injecting or oral way, which might be forged into a veterinary drug. Further, IPPA152201 could undergo Diels–Alder reactions to form novel neonicotinoids with a tetrahydropyridine fixed cis-configuration, which is structurally related to paichongding. On the basis of this observation, the highly active IPPA152311 and IPPA153417 series were discovered. Studies on modes of action (MOA) showed that these cis-compounds have different MOA from imidacloprid. Paichongding and IPPA152201 are agonists of nAChRs, while Cycloxaprid and IPPA152311 act on nAChRs as antagonists.
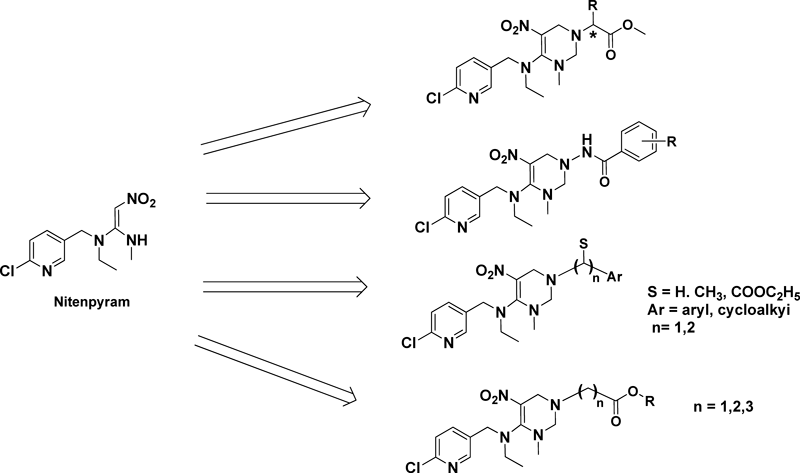
Due to the encouraging results of research on cis-neonicotinoid, Dr. Chuanwen Sun from Shanghai Normal University pursued it. Based on the open-chain neonicotinoid nitenpyram, four series of nitenpyram derivatives with a tetrahydropyrimidine-fixed cis-configuration were prepared by reacting with l-α amino acid methyl ester, substituted benzoyl hydrazine, various substituted amines and amino acid alkyl ester, respectively (Fig. 10).9–11) Most of these compounds were effective against brown planthopper (Nilaparvata lugens). And Chuanwen Sun also assembled the amino acid to the neonicotinoid core structure to generate some 1,5-disubstituted 1,3,5-hexahydrotriazine-2-N-nitroimine analogues, which also bearing good insecticidal potency.12)
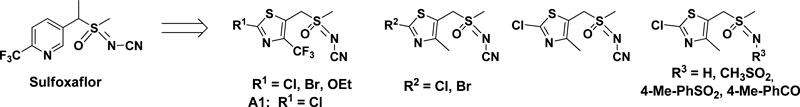
Sulfoxaflor, the newest neonicotinoid discovered by Dow AgroSciences, acts on nAChRs as an agonist.13,14) It exhibits high potency and lacks insecticidal cross-resistance; thus it could be used as an alternative to control sap-feeding insect pests.15) It is expected to be introduced in 2013 in China as long as the registrations are granted. Starting from the core structure of Sulfoxaflor, Professor Jianxin Fang from Nankai University synthesized some thiazolyl analogues by replacing pyridyl in Sulfoxaflor with a thiazolyl moiety. These compounds exhibited some degree of activity against peach aphid (Myzus persicae). Compound A1 (Fig. 11) had the highest activity, which was comparable to Sulfoxaflor among these analogues.16)
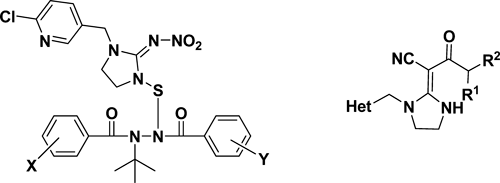
By incorporating imidacloprid and N-tert-butyl-N,N′-diacylhydrazines (insect growth regulators), Professor Qingmin Wang from Nankai University reported a series of novel N-tert-butyl-N′-thio[1-(6-chloro-3-pyridylmethyl)-2-nitroiminoimidazolidine]-N,N′-diacylhydrazines (Fig. 12). All derivatives exhibited insecticidal activities against the oriental armyworm (Mythimna separata) and bean aphid (Aphis fabae Scopoli). These bifunctional molecules have insecticidal activities that are as rapid as those in imidacloprid at higher concentrations and insect growth-regulatory activity similar to that of diacylhydrazines at lower concentrations.17)
Professor Song Yang from Guizhou University explored some novel neonicotinoid analogs bearing both cyano and amide pharmacophore units (Fig. 12). Some of the synthesized compounds were shown to exhibit moderate activity (>50%) against Nilaparvata lugens at 100 mg/L.18)
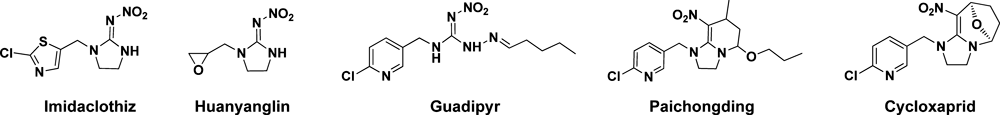
With increasing sales of these effective materials, several neonicotinoids, such as imidacloprid and thiamethoxam, have grown into a billion-dollar industry. Motivated by the profitability of these substances, university and commercial researchers in China are working hard to bring new products to the market. Five novel neonicotinoids have been commercialized, and others are about ready for the market. The five are Imidaclothiz, Guadipyr, Huanyanglin, Paichongding and Cycloxaprid (Fig. 13).
Imidaclothiz was a chlorothiazolyl analogue of imidacloprid developed by Nantong Jiangshan Agrochemical & Chemical Co., Ltd. (Jiangsu Province). It gained full registration from the Chinese Ministry of Agriculture in 2006.19) It is a kind of broad-spectrum effective pesticide against sucking insects. It is safe for crops and is useful on vegetables and fruits.20) However, with regards to toxicity, it is low for fish, moderate for birds, and high for bees and silkworms, and, as a result, attention is required during application.
Guadipyr (PCT/CN2008001919) and Huanyanglin (200810236885.X) are two novel neonicotinoids discovered recently by the China Agricultural University and Wuhan Institute of Technology, respectively. They are under development and might enter the market in two years. They act on nAChRs as an agonist. Field trials show that both of them are effective against sucking insects, such as aphids and rice planthoppers.
Paichongding was jointly developed by Jiangsu Kwin Group Co., Ltd. and ECUST. It obtained a temporary registration as a pesticide in 2009, and its 10% suspension has been sold in the Chinese market. Presently, Jiangsu Kwin is the exclusive producer of this pesticide in China. The R&D and industrial project concerning paichongding is listed as a branch project (Innovation and Creation) in China’s Eleventh Five-Year Plan. Paichongding effectively controls planthoppers and aphids and is particularly effective against resistant strains.
Cycloxaprid was invented by ECUST and licensed by the manufacturer Shanghai Shengnong Pesticides Co., Ltd. (SSPC). On Feb. 22, 2012, ECUST, SSPC, and FMC signed agreements to form a joint venture for a new active ingredient for a neonicotinoid insecticide. Cycloxaprid showed excellent activities against a variety of insects, such as Nephotettix bipunctatus, Laodelphasx cstriatellus, Nilaparvata lugens, Myzus persicae, Frankliniella occidentalis, Bemisia argentifolii, and Aphis craccivora. Except for the outstanding activity against homoptera insects, Cycloxaprid also has high activities against lepidoptera insects, such as Pseudaletia separate Walker and Cnaphalocrocis medinalis Guenee. Most importantly, it exhibited better bioactivity against imidacloprid-resistant insects. From 2009 to 2011, a flurry of field trials regarding 25% Cycloxaprid WP and 70% Cycloxaprid WD were performed in more than 10 provinces in China. Cycloxaprid is superior in pest control. The development of cycloxaprid has been financially supported by the major projects in China, such 973, 863 programs. Moreover, Cycloxaprid has broader market scope thanks to the dual effect of being both a promising agricultural insecticide and a veterinary drug.
Imidaclothiz and paichongding are already on the market, but the sales have been disappointing. Guadipyr, Huanyanglin and Cycloxaprid are under development. Although some of the releases mimic their commercial counterparts, they are quite promising.
Neonicotinoid insecticides are insect-selective nAChRs agonists, and the great abundance of nAChRs within the insect central nervous system (CNS) has led to the quick development and extensive use of neonicotinoid insecticides. Imidacloprid and other neonicotinoid insecticides, like other systemic insecticides, display prolonged persistence which is likely to generate high selection pressure for resistance.21) Resistance to imidacloprid has been reported in a range of species, including Nilaparvata lugens (brown planthopper), a major rice pest in many parts of Asia.22,23) In a laboratory strain of N. lugens with high resistance to imidacloprid, the point mutation Y151S in nAChR α subunits has been identified to contribute to neonicotinoid insecticide resistance.24,25) Radioligand-binding studies, performed with both native nAChR preparations and heterologously expressed recombinant nAChRs in Drosophila S2 cells, demonstrated that the Y151S resistance-associated point mutation is responsible for a dramatically reduced level of specific [3H]imidacloprid binding.24) By expression of recombinant nAChRs in Xenopus oocytes, this Y151S mutation was found to have remarkable effects on all neonicotinoid insecticide potencies but little on Ach.25)
An nAChR point mutation (Y151S) associated with imidacloprid resistance has been previously identified in N. lugens nAChR Nlα1 and Nlα3 subunits; thus, subunit composition studies related to these two subunits were performed. Heterologous expression and co-immunoprecipitation studies demonstrated the co-assembly of Nlα1, Nlα2 and Nlβ1 subunits into one receptor complex, Nlα1/Nlα2/Nlβ1, and of Nlα3, Nlα8 and Nlβ1 into another Nlα3/Nlα8/Nlβ1.26–28) Radioligand-binding studies in N. lugens native nAChRs demonstrated that Nlα1/Nlα2/Nlβ1 constituted the lower-affinity binding site and Nlα3/Nlα8/Nlβ1 constituted the higher-affinity binding site of imidacloprid. In heterologous expression, the Nlα1Y151S mutation showed 1.8-fold effects on imidacloprid sensitivity against Nlα1 and Nlα2 containing receptor Nlα1/Nlα2/rβ2, which was much less than the Nlα3Y151S mutation with 9.2-fold effects on Nlα3 and Nlα8 containing receptor Nlα3/Nlα8/rβ2.27,28) These data indicated that the Nlα3Y151S mutation might be more important for imidacloprid resistance.
All commercial neonicotinoid insecticides possess either an electron-withdrawing nitro (–NO2) or cyano (–CN) group and have been postulated to contribute directly to their selectivity.29) The –NO2 or –CN group in neonicotinoids is in trans-configuration. Recently, some potential neonicotinoids with cis-configuration have been synthesized and shown insecticidal activity in a wide range of insects, including N. lugens.30–32) An interesting finding is that the imidacloprid resistant populations of N. lugens showed little cross-resistance to these potential neonicotinoids with a –NO2 or –CN group in trans-configuration.33,34) Among these potential neonicotinoids, the dicyclic neonicotinoids with cis-configuration discriminated between the high- and low-affinity imidacloprid-binding sites in N. lugens and their potencies were only slightly influenced by the previously identified mutation Y151S in N. lugens.24,34) Another potential neonicotinoids with –NO2 or –CN group in trans-configuration was synthesized by replacing nitromethylene pharmacophore with a nitro-conjugated system, with a representative member IPPA152201.32) IPPA152201 showed a comparable toxicity with imidacloprid against N. lugens in a susceptible strain and had no significant cross-resistance in an imidacloprid resistant strain. The potency of IPPA152201 on hybrid receptors Nlα1/β2 expressed in Xenopus oocytes was also only slightly influenced by the Y151S mutation, relative to that of imidacloprid and other commercial neonicotinoids.33)
China and neonicotinoids create a win–win situation for each other. China has benefited considerably from the excellent control efficacy of neonicotinoid in crop protection and earned significant profits from the neonicotinoid industry. In addition, China has promoted the development of neonicotinoids. China is currently the world leader in neonicotinoid production, consumption, and export. Meanwhile, theoretical research of neonicotinoids has developed rapidly over the past decade, and the elucidation of the resistance mechanism in the brown planthopper and discovery of cis-neonicotinoids are two of the most representative ones.
Because of the resistance of some pests, the domestic neonicotinoid market has been shrinking. A production license for Sulfoxaflor is being sought in China, and it is anticipated that Guadipyr, Huanyanglin and Cycloxaprid, all of them promising, will appear in the domestic market in 2–3 years. Furthermore, the patent of thiamethoxam will expire in July 2013, and the domestic producers will definitely turn to this promising product. Combined with the invasion of the novel chemicals Chlorantraniliprole and Flubendiamide, the structure of neonicotinoids in China will change significantly. There remain some problems in China, such as the overcapacity and lack of innovative and competitive products. During the 12th Five-Year Plan, China will make great efforts to optimize the pesticide industry structure, improve the core competitiveness of Chinese pesticides, and provide more funds for pesticide research. One of the most remarkable changes is that the neonicotinoid producers are expected to undergo great amalgamation and reformation, which would result in the formation of very large companies. Nonetheless, no matter what happens, China will continue to contribute its own effort on the development of neonicotinoid and much more is still to come in this area.
We thank Yibin Zhang from Shanghai Pesticide Research Institute and Congcong Ao from China Crop Protection Industry Association for their kindly providing technical information and insight advice and assistance.
This work was financial supported by National Basic Research Program of China (973 Program, 2010CB126100), National High Technology Research Development Program of China (863 Program, 2011AA10A207) and the National Key Technology R&D Program of China (2011BAE06B01). This work was also partly supported by the Fundamental Research Funds for the Central Universities.