2024 年 49 巻 3 号 p. 168-178
2024 年 49 巻 3 号 p. 168-178
Widespread pesticide use for decades has caused environmental damage, biodiversity loss, serious human and animal health problems, and resistance to insecticides. Innovative strategies are needed to reduce treatment doses in pest management and to overcome insecticide resistance. In the present study, combinations of indoxacarb, an oxadiazine insecticide, with sublethal concentrations of deltamethrin encapsulated in lipid nanocapsules, have been tested on the crop pest Acyrthosiphon pisum. In vivo toxicological tests on A. pisum larvae have shown a synergistic effect of nanoencapsulated deltamethrin with a low dose of indoxacarb. Furthermore, the stability of deltamethrin nanoparticles has been demonstrated in vitro under different mimicking environmental conditions. In parallel, the integrity and stability of lipid nanoparticles in the digestive system of aphid larvae over time have been observed by Förster Resonance Energy Transfer (FRET) imaging. Thus, the deltamethrin nanocapsules/indoxacarb synergistic association is promising for the development of future formulations against pest insects to reduce insecticide doses.
Pests destroy up to 40% of global crop production and cause US $220 billion in trade losses each year according to the United Nations and invasive insects cost a minimum of US $70 billion per year globally.1,2) Among pest insects, the aphids especially pea aphid Acyrthosiphon pisum, a key pest of leguminous and pulse crops worldwide3) can cause serious economic damage and yield losses. This sap-sucking insect has a wide host range including fava bean, lupin, alfalfa, lentil, chickpea, grass pea, and field pea. A. pisum as phloem-feeders can injure crops directly by consuming the sap of plants and indirectly by vectoring over 30 phytopathogenic viruses, including cucumber mosaic virus, pea enation mosaic virus, and bean leafroll virus.4,5) Aphids have a complex life cycle, including a succession of parthenogenetic generations during spring and summer and a single sexual generation in autumn.6,7) This mode of reproduction together with i) very prolific parthenogenetic adult females capable of producing 50 to 100 nymphs, ii) short generation time of aphids as well as iii) their remarkable ability adaptation to environmental conditions contribute to the rapid growth of aphid populations and make the control of these crop pests difficult.3,7,8) Currently, among strategies developed to fight against aphids, chemical insecticides are still primarily used. However, the development of resistance to insecticides must be considered in management strategy for aphid control.3,4,7,9)
Since decades, Integrative Pest Management (IPM) and Insecticide Resistance Management (IRM) programs have been developed to evaluate the proper insect pests control methods both for effectiveness and risk. According to the Food and Agriculture Organization of the United Nations, IPM is defined as “an ecosystem approach to crop production and protection that combines different management strategies and practices to grow healthy crops and minimize the use of pesticides”.10) Many networks like PAN Europe or ENDURE have been created to contribute to IPM strategies implementation.11,12) However, a gap exists between IPM concepts and practices in the fields.13) Indeed, despite i) the increasing development of insecticide resistances, ii) the rising number of restrictions regarding the use of agrochemicals with high health risks, iii) the implementation of laws or national programs to reduce the adverse effects on the environment, the beneficial insects and on the human health, current pest control strategies still rely on the use of insecticides.14–16) These problems reinforce the need to develop more efficient strategies allowing a decrease in the amount of active ingredients in the formulations used for insecticide treatments.
Among innovative strategies to reduce doses of insecticides treatment, it has previously been shown that sublethal concentration of deltamethrin was able to potentiate the effect of indoxacarb, an oxadiazine insecticide, on the cockroach Periplaneta americana.17) Indoxacarb is a pro-insecticide that undergoes bioactivation to the more effective N-decarbomethoxyllated indoxacarb (DCJW) by esterases and amidases in insects.18,19) This insecticide is part of the group 22A in the mode of action classification table of insecticide resistance action committee IRAC20) as the DCJW metabolite is a highly potent voltage-gated sodium channel blocker.21,22) In this strategy, deltamethrin produces an increase of intracellular calcium concentration in isolated insect neurons through the reverse Na/Ca exchanger leading to activation of calcium-dependent intracellular signalling pathways. This activation induces a modification in the voltage-gated sodium channel conformational states, leading to increase its sensitization to DCJW, thus optimizing the efficacy of the treatment. Furthermore, to protect deltamethrin from insect detoxification enzymes like esterases and cytochrome P450 monooxygenases, this molecule was nanoencapsulated in lipid nanocapsules. This was an essential first step to optimize the synergistic association when combined with indoxacarb.17) Because this previous study demonstrated that nanoencapsulated deltamethrin combined with indoxacarb increased cockroaches mortality rate at lower concentrations compared to indoxacarb used alone, we decided to adapt and to test such strategy on aphid, A. pisum. The pro-insecticide indoxacarb has been designated by the EPA to be a “reduced-risk” pesticide and is used against a broad spectrum of insect pests. This insecticide is therefore compatible with IPM programs23–25) and it has been reported to be effective on aphids under laboratory conditions.26,27) Here, toxicological tests on A. pisum larvae have demonstrated the efficacy of the deltamethrin nanocapsules/indoxacarb association. In parallel, Förster Resonance Energy Transfer (FRET) imaging has been used for monitoring in vivo the integrity of lipid nanoparticles, showing a stability and storage in aphid larvae digestive system over time. Furthermore, in vitro experiments have demonstrated a high stability of deltamethrin-nanoparticles against various environmental-mimicking conditions, making this association a promising strategy in pest management.
Deltamethrin (Sigma-Aldrich, Saint-Quentin-Fallavier, France) was diluted in dimethylsulfoxide (DMSO) (Sigma-Aldrich) to obtain a stock solution at 10−2 M and stored at −20°C. The insecticide indoxacarb (Avaunt® 15% Emulsion Concentrate (EC)) was supplied by DuPont™ (Wilmington, Delaware, USA). Piperonyl butoxide (PBO) (Sigma-Aldrich) was prepared in DMSO to obtain a stock solution at 500 mg/mL stored at 4°C. Lipophilic carbocyanine dyes 1,1′-dioctadecyl-3,3,3′,3′-tetramethylindodicarbocyanine 4-chlorobenzenesulfonate (DiD) and 1,1′-dioctadecyl-3,3,3′,3′-tetramethylindocarbocyanine perchlorate (DiI) were supplied by Molecular probes® (Eugene, OR, U.S.A.) and stored at 4°C.
2. InsectsExperiments were performed on the pea aphids, Acyrthosiphon pisum strain LSR1.28) They were generously provided by INRAE-Rennes IGEPP. Unwinged parthenogenetic females were reared on faba bean (Vicia fabae) plants under laboratory conditions at 22°C and a photoperiod of 16 : 8 hr (L : D). Under these conditions, aphids reproduced via viviparous parthenogenesis as clonal females, and newborn larvae became adults after four molts.
3. In vivo toxicological studiesAll the experiments were conducted with an artificial diet bioassay as previously developed.29,30) According to the method, artificial diet was prepared and stored at −20°C for a period up to 6 months. The artificial diet was used to prepare all compound dilutions. Tests were performed using 100–375 larvae (for n=4 to 15) per concentration of insecticide. In brief, adults were put on a feeding apparatus containing 200 µL of artificial diet at day 0. At day 1, 25 first-instar nymphs were placed to 200 µL of freshly prepared diet containing molecules alone or in combination. Concentrations ranging from 10−3 to 10−8 M for indoxacarb and from 10−4 to 10−8 M for deltamethrin were tested. For PBO, a sublethal concentration of 1 mg/mL was used. The mortality was observed after 48 hr (day 3) and 72 hr (day 4). Aphids that were unable to walk were considered dead and were removed.
Corrected mortality percentages were calculated using Henderson Tilton’s formula after 48 hr and 72 hr of insecticide exposure.31) The Model Deviation Ratio (MDR) (observed toxicity/predicted toxicity) was calculated to define the nature of the association.32) MDR values<0.7 mean “antagonistic effect”, results between 0.7 and 1.3 mean “absence of effect” and results>1.3 mean “synergistic effect”.
4. Formulation and characterization of lipid nanocapsules4.1. Preparation of lipid nanocapsules, deltamethrin-loaded lipid nanocapsules and dye-loaded lipid nanocapsulesLipid nanocapsules (LNCs) were performed following a previously developed phase inversion-based method.33) Briefly, 75 mg of Lipoid® 75-3 (Lipoïd GmbH, Ludwigshafen, Germany) and 846 mg of Kolliphor® HS 15 (BASF, Ludwigshafen, Germany) (surfactants), 1028 mg of Labrafac® WL 1349 (oil phase) (Gattefossé S.A., Saint-Priest, France), 89 mg of NaCl (Prolabo, Fontenay-Sous-Bois, France) and 2962 mg of de-ionized water (Milli® Q system, Millipore, Billerica, MA, USA) (aqueous phase) were precisely weighed.34) Under continuous agitation (magnetic stirring), the mix was heated up to 95°C and cooled down to 50°C three times. During the last cooling, 5 mL of deionized cold water (at 4°C) were quickly added at about 75°C corresponding to the transition between the oil-in-water (low temperature) and water-in-oil (high temperature) emulsions, to stop the emulsion process, leading to the suspension of LNCs. For deltamethrin-loaded LNCs (deltamethrin-LNCs) formulation, deltamethrin was first dissolved in Labrafac® WL 1349 at a concentration of 1 mg/g Labrafac® WL 1349 (w/w), before adding other constituents.17) Before dye-loaded LNCs formulation, 4-chlorobenzenesulfonate and perchlorate ions for DiD and DiI, respectively, were replaced by tetraphenylborate (TPB) ion to improve dye solubility in Labrafac® WL 1349 and consequently the dye loading capacity.35) Then, these modified dyes were solubilized in Labrafac® WL 1349 before formulation process to achieve DiI/TPB-loaded LNCs (DiI/TPB at 1% (w/w) in Labrafac® WL 1349) (DiI/TPB-LNCs), DiD/TPB-loaded LNCs (DiD/TPB at 1% (w/w) in Labrafac® WL 1349) (DiD/TPB-LNCs), and both DiI/TPB and DiD/TPB-loaded LNCs (both DiI/TPB and DiD/TPB at 1% (w/w) in Labrafac® WL 1349) (FRET-LNCs). LNCs, deltamethrin-LNCs, DiI/TPB-LNCs, DiD/TPB-LNCs and FRET-loaded LNCs suspensions were stored at 4°C.
4.2. Characterization of lipid nanocapsules and deltamethrin-loaded lipid nanocapsulesLNCs and deltamethrin-LNCs were diluted by factor 60 (v/v) in de-ionized water and size distribution, i.e., hydrodynamic diameter (Z-ave), polydispersity index (PdI) and Zeta potential (ZP) were measured using a Zetasizer® Nano ZS (Malvern Panalytical Ltd., Worcestershire, UK) at 25°C. The instrument is equipped by a Helium–Neon laser, 4 mW, with an output wavelength of 633 nm and a scatter angle fixed at 173°. The correlation functions were fitted using an exponential fit (Cumulant approach) for Z-ave and PdI determinations. Smoluchowski approach was used to evaluate the electrophoretic mobility to calculate ZP. The encapsulation efficiency of deltamethrin in LNCs was evaluated by chromatography, as previously described.17)
4.3. LNC stabilityThe stability of deltamethrin-LNCs was followed over time up to 14 days depending on different settings: temperature (4, 20 and 37°C), pH (4, 6, 8, 10) and brightness (photoperiod of 16 : 8 hr (L : D)) or darkness, UV 365 nm (24 hr). Deltamethrin-LNCs were diluted to 7/10 in de-ionized water and four parameters (Z-ave, PdI, ZP and encapsulation efficiency) were characterized as described above.
5. Statistical analysisData were analyzed with GraphPad Prism version 5.00 (GraphPad software, Inc.) and displayed as mean and S.E.M. (Standard error of the mean). Values are means of at least three independent experiments. Between-group comparisons were assessed by nonparametric Mann–Whitney U-test or Kruskal–Wallis test followed by post-hoc Dunn test for pairwise comparison. A p<0.05 was considered statistically significant.
6. In vivo imaging6.1. Monitoring of LNCs by FRETFRET methodology was used to monitoring the integrity of LNCs in vivo.36) Studies were realized on first instar larvae (aged 0–24 hr). Dye-loaded LNCs were diluted with artificial diet to obtain a concentration of 850 and 85 µg/mL. Ten larvae were placed in diet cages and fed with different tests’ conditions (DiI-LNCs, DiD-LNCs and FRET-LNCs) for 5 or 24 hr. Then tests’ conditions were removed and replaced by artificial diet for 24 hr to 7 days. At the end of each experiment, larvae were frozen at −20°C.
6.2. Fluorescence analyzingFluorescence analyzing was performed at Plateau Technique Mutualisé d’IMAgerie Cellulaire (PTM IMAC) (SFR 4207 Quasav, Angers, France). Larvae having previously ingested dye-loaded LNCs were placed on slide (Hecht Karl™, Sondheim, Germany), and recovered by a coverslip (Thermo FisherScientific Inc., Illkirch, France). Larvae were thus placed under fluorescence microscope (NIKON, Ti-E A1, France), at a CFI Plan Fluor 10X objective (NA 0.30, WD 16.0 mm). Adequate excitation and emission wavelength couples (λex/λem) were used to analyze fluorescence. These couples were 561/570–620 for DiI, 640/663–738 nm for DiD and 561/663–738 for FRET. Data obtained were compared each other with application of specific settings common to all samples (excitation time 10 msec, laser powerful 2%) with the software NIS-Element Arv 5.21.03 (NIKON Corporation, Tokyo, Japan).
To evaluate the potential synergy between deltamethrin and indoxacarb in vivo on A. pisum larvae, we first investigated the effect of each molecule alone on the mortality rate. The toxicological effects of deltamethrin or indoxacarb upon these sucking insects were studied by using an artificial diet system as previously described.29) The average corrected percentages of mortality were plotted against the noncumulative concentrations of indoxacarb or deltamethrin after 48 hr and 72 hr ingestion. The mortality rate after 24 hr ingestion was not investigated as preliminary experiments showed absence of larval mortality after ingestion of indoxacarb. The sigmoid curves corresponded to the fit according to the Hill equation (Table 1). The concentration–response curve established with indoxacarb after 72 hr post-ingestion was steeper than the curve at 48 hr after ingestion. The corresponding slope parameters were 1.93 and 5.82 at 48 hr and 72 hr, respectively (Fig. 1A, Table 1). This variation may reflect the bioactivation of indoxacarb in its active metabolite DCJW. The increase of the Hill slope parameter at 72 hr could therefore be due to a higher quantity of DCJW, which has a better binding affinity to the voltage-gated sodium channel compared to indoxacarb. The maximum corrected percentage of mortality was 100% for 48 hr (n=3) and 72 hr (n=4) after 10−3 M indoxacarb ingestion, respectively. The threshold concentration inducing very low mortality (<10%) was 3×10−6 M at 48 hr and 72 hr (Fig. 1A). The LC50 values estimated for indoxacarb were 1.1×10−5 M and 6.2×10−6 M at 48 hr and 72 hr after ingestion, respectively (Table 1).
| Indoxacarb | Deltamethrin | |||
|---|---|---|---|---|
| 48 hr | 72 hr | 48 hr | 72 hr | |
| LC50 (M) | 1.1 10−5 M | 6.2 10−6 M | 7.8 10−6 M | 6.3 10−6 M |
| Hill slope | 1.93 | 5.82 | 1.50 | 2.67 |
| R2 | 0.95 | 0.96 | 0.91 | 0.88 |

For deltamethrin, the Hill slope was slightly increased for the sigmoid curve obtained after 72 hr ingestion (Hill slope 2.67) compared to the concentration–response curve at 48 hr (Hill slope 1.50) (Fig. 1B, Table 1). The maximum corrected mortality rates were 93±3% (n=6) and 99±1% (n=6) at 48 hr and 72 hr after ingestion, respectively. The LC50 values estimated for deltamethrin were 7.8×10−6 M and 6.3×10−6 M at 48 hr and 72 hr after ingestion, respectively. The 48 hr post-ingestion LC50 of deltamethrin in A. pisum had been previously evaluated to 3.1×10−5 M, which is in the same concentration range compared to our results.37)
This set of experiments allowed to determine the lower concentrations of indoxacarb and deltamethrin to be tested for the synergy study between both compounds. Indeed, in our strategy, deltamethrin was used at concentrations that induce the lowest lethal effect, it must therefore be used at concentrations less than or equal to 10−7 M.
Thus aphid larvae were fed with various concentrations of indoxacarb (10−7 to 10−5 M) alone and associated with deltamethrin (10−7 M). The percentages of corrected mortality after 48 hr and 72 hr ingestion are illustrated in Fig. 2. No significant difference was observed between mortality rates with indoxacarb alone or associated with deltamethrin 10−7 M at 48 hr or 72 hr after ingestion.
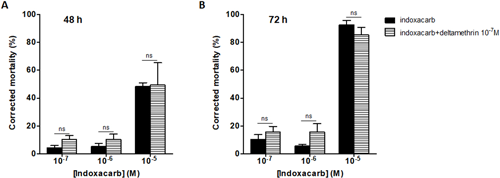
The absence of synergy between the deltamethrin and indoxacarb probably reflects the rapid metabolization of deltamethrin by detoxification enzymes of aphid larvae.38,39) It has been previously shown that encapsulation of deltamethrin in lipid nanocapsules (LNCs) protects deltamethrin from enzymatic degradation allowing the optimization of the synergistic association.17) Since few years, nanotechnology research is raising interest in the agricultural sector as nano-based pesticide formulations have several advantages such as increased efficacy, controlled release or reduced environmental impacts.40–45) Indeed various classes of insecticides have been already nanoformulated like pyrethroids (deltamethrin, β-cypermethrin), benzoylphenyl urea (novaluron) or avermectines (abamectine).41,44) Among these nano-insecticides, various types of nanosystems have been developed such as nanoemulsion, nanospheres, nanomicelles, nanosuspensions or nanocapsules. In the latter the insecticides are encapsulated in a core-shell structural vesicular system usually composed of biodegradable polymers (i.e., chitosan, polyethylene glycol, poly(lactic-co-glycolic acid)) whom slow degradation in the environment increases the chemical stability.17,45,46) Consequently, we first studied the physico-chemical stability of deltamethrin-loaded nanocapsules (deltamethrin-LNCs) under various conditions mimicking environmental factors affecting release. The size distributions of deltamethrin-LNCs were measured in vitro after incubation at different conditions of temperature (4, 20 and 37°C), pH (4, 6, 8, 10) and brightness or darkness, (photoperiod L:D), UV 365 nm (24 hr). All conditions tested were compared to reference values obtained at 20°C, pH 6, in the presence of light at time 0. Regardless of all the parameters tested, encapsulation values remain constant over time, from 24 hr to 14 days, i.e., Z-ave=56–64 nm, PdI=0.05–0.12, ZP=−13–−6 mV and deltamethrin encapsulation rate=92–113% (Table 2). The deltamethrin-LNCs did not change at acidic or basic pH values and were not degraded in different conditions, suggesting that the deltamethrin still remained encapsulated within the LNCs and protected in the external environment before ingestion by the insects.
| a. Particle size (Z-ave) (nm) | ||||||||
|---|---|---|---|---|---|---|---|---|
| Experimental conditions | Time (days) | |||||||
| Temp. | pH | Light | 0 | 1 | 2 | 3 | 7 | 14 |
| 20°C | 6 | + | 56±2 | 59±3 | 59±3 | 58±1 | 59±1 | 59±1 |
| 4 | 57±2 | 59±4 | 58±1 | 59±1 | 59±1 | |||
| 8 | 57±2 | 58±1 | 59±3 | 58±2 | 60±1 | |||
| 10 | 57±1 | 57±1 | 59±2 | 59±2 | 62±5 | |||
| 4°C | 57±1 | 57±1 | 57±1 | 58±1 | 59±1 | |||
| 37°C | 57±1 | 57±1 | 57±1 | 58±1 | 64±11 | |||
| — | 56±2 | 60±5 | 58±1 | 59±6 | 59±3 | |||
| UV | 57±1 | |||||||
| b. Polydispersity index (PdI) | ||||||||
| Experimental conditions | Time (days) | |||||||
| Temp. | pH | Light | 0 | 1 | 2 | 3 | 7 | 14 |
| 20°C | 6 | + | 0.06±0.02 | 0.10±0.07 | 0.10±0.08 | 0.11±0.07 | 0.07±0.03 | 0.07±0.01 |
| 4 | 0.06±0.01 | 0.10±0.11 | 0.06±0.01 | 0.06±0.02 | 0.07±0.01 | |||
| 8 | 0.07±0.04 | 0.08±0.03 | 0.10±0.09 | 0.06±0.01 | 0.08±0.02 | |||
| 10 | 0.06±0.01 | 0.06±0.01 | 0.11±0.09 | 0.09±0.05 | 0.11±0.08 | |||
| 4°C | 0.09±0.04 | 0.06±0.02 | 0.07±0.01 | 0.07±0.01 | 0.07±0.01 | |||
| 37°C | 0.08±0.04 | 0.06±0.01 | 0.08±0.03 | 0.06±0.02 | 0.12±0.13 | |||
| — | 0.06±0.01 | 0.10±0.08 | 0.08±0.04 | 0.09±0.08 | 0.09±0.07 | |||
| UV | 0.05±0.02 | |||||||
| c. Zeta potential (ZP) (mV) | ||||||||
| Experimental conditions | Time (days) | |||||||
| Temp. | pH | Light | 0 | 1 | 2 | 3 | 7 | 14 |
| 20°C | 6 | + | −12±5 | −9±2 | −8±1 | −8±2 | −9±3 | −13±4 |
| 4 | −9±2 | −9±1 | −8±1 | −9±2 | −10±3 | |||
| 8 | −8±1 | −7±1 | −8±2 | −8±2 | −8±2 | |||
| 10 | −10±2 | −9±2 | −8±2 | −10±4 | −8±3 | |||
| 4°C | −8±1 | −7±1 | −7±2 | −7±2 | −6±2 | |||
| 37°C | −7±0 | −9±2 | −8±2 | −7±2 | −10±4 | |||
| — | −9±2 | −8±1 | −9±1 | −8±2 | −10±5 | |||
| UV | −9±1 | |||||||
| d. Deltamethrin encapsulation rate (%) | ||||||||
| Experimental conditions | Time (days) | |||||||
| Temp. | pH | Light | 0 | 1 | 2 | 3 | 7 | 14 |
| 20°C | 6 | + | 105±3 | 106±3 | 104±7 | 109±4 | 107±5 | 104±6 |
| 4 | 103±2 | 101±5 | 102±4 | 104±7 | 100±3 | |||
| 8 | 101±4 | 102±5 | 101±6 | 94±6 | 92±9 | |||
| 10 | 105±1 | 101±4 | 105±1 | 104±3 | 98±10 | |||
| 4°C | 107±2 | 106±5 | 104±8 | 105±4 | 107±2 | |||
| 37°C | 108±4 | 107±4 | 107±3 | 107±4 | 113±5 | |||
| — | 110±2 | 109±4 | 108±3 | 109±4 | 112±4 | |||
| UV | 105±2 | |||||||
Four parameters have been studied: (a) particle size (Z-ave), (b) polydispersity index (PdI), (c) Zeta potential (ZP) and (d) deltamethrin encapsulation rate. n=4, mean±S.D.
We then focused on the in vivo stability and fate of the nanocapsules. To carry out this study, control experiments have been performed to estimate the toxicity of empty LNCs on A. pisum larvae. Aphids were fed with artificial diet containing different concentrations of LNCs (from 8.5 to 8500 µg/mL). The highest tested concentration corresponded to the maximal concentration that could be obtained in the experimental system. At the concentration of 8500 µg/mL, LNCs were toxic for aphid larvae with corrected mortality of 64±4% at 48 hr and 91±7% at 72 hr (Fig. 3). However, no effect on mortality rates was observed for lower LNCs concentrations and in vivo studies were then performed with LNCs at the maximal concentration of 850 µg/mL.

The fate and integrity of LNCs in A. pisum larvae were monitored by FRET (Förster resonance energy transfer) imaging. Co-encapsulation of two fluorescent probes (DiI, FRET donor, and DiD, FRET acceptor) in LNCs (FRET-LNCs) enables to monitor the nanocapsules stability in vivo.35)
Before tracking the LNCs integrity in A. pisum larvae over time, we had to define experimental conditions for which a FRET signal was obtained after FRET-LNCs ingestion. Different ingestion times (5 or 24 hr) and concentrations (850 or 85 µg/mL) of FRET-LNCs were tested. Analysis by fluorescent microscopy in whole insects has shown a stronger FRET signal after 24 hr ingestion of FRET-LNCs at 850 µg/mL compared to shorter time or lower concentration of FRET-LNCs (Supplementary Fig. 1). LNCs integrity was thus tracked in vivo over 8 days after 24 hr ingestion of FRET-LNCs at 850 µg/mL. As shown on Fig. 4, a FRET signal has been observed over 8 days with a maximum intensity around 72–96 hr post-ingestion. This FRET signal was correlated with a weak signal of DiI, the donor molecule, indicating a decrease of the donor fluorescence intensity due to energy transfert to acceptor molecule. This signal was localized in a part of the abdomen of aphid larvae. The gut anatomy of A. pisum is very simple with a stomach followed by an intestine without diverticula.47,48) Based on our fluorescent microscopy observations, the FRET-LNCs seemed to be stocked in the stomach. At 72 hr post-ingestion, a FRET signal was observed in the whole abdomen, reflecting a probable diffusion of FRET-LNCs in A. pisum larvae. The presence of FRET signal indicated an integrity of LNCs over time. One may hypothesized that LNCs were dissociated in stomach, thus releasing the fluorescent probes, but that the latter remained sufficiently close for the transfer of energy. Two control experiments have been performed, aphid larvae have ingested a mix of free fluorescent probes (DiI and DiD) or a mix of DiI-LNCs and DiD-LNCs. No fluorescent signal was observed after 24 hr post-ingestion of free probes, indicating that even these probes were released, they were not stocked in stomach and capable of energy transfer (Supplementary Fig. 2). Control experiment with ingestion of DiI-LNCs/DiD-LNCs provided confirmation that full LNCs are concentrated in stomach after 24 hr post-ingestion, as FRET signal was observed, meaning that these LNCs were close enough to have energy transfer from DiI-LNCs to DiD-LNCs (Supplementary Fig. 2). Our results indicated that LNCs were stable over time in A. pisum. The integrity of LNCs in vivo is an important point for our strategy, as the deltamethrin needs to remain protected from enzyme degradation to allow time for the indoxacarb to be bio-activated in DCJW.

Results on deltamethrin-loaded LNCs stability under various physico-chemical conditions and the fact that the LNCs remain intact in vivo over time in A. pisum indicated that deltamethrin-LNCs could be an added value in the crop protection.
Thereafter, the impact of the encapsulation of deltamethrin on A. pisum toxicity was investigated. The mortality rates of two concentrations of encapsulated deltamethrin (10−8 M and 10−7 M) were compared to mortality rates obtained with free deltamethrin (Fig. 5). For both concentrations of deltamethrin (10−7 M and 10−8 M), no significant difference was observed between encapsulated deltamethrin and non-encapsulated deltamethrin.

To study the efficacy of the association deltamethrin-LNCs/indoxacarb, aphids were fed with deltamethrin-LNCs at 10−8 M or 10−7 M of deltamethrin associated with various concentrations of indoxacarb (from 10−7 to 10−5 M). As no synergistic effect has been observed with associations of indoxacarb with deltamethrin-LNCs 10−8 M (data not shown), only comparative mortality rates with deltamethrin-LNCs at 10−7 M estimated at 48 hr and 72 hr are illustrated in Fig. 6. At 48 hr, significant differences were observed between corrected mortality of indoxacarb alone and deltamethrin-LNCs 10−7 M/indoxacarb for three concentrations of indoxacarb. Indeed, the mortality rate increased from 3±2 to 34±5% at 3×10−6 M (U(6,6)=0.0, p=0.0048), from 20±5 to 50±12% at 5×10−6 M (U(6,4)=2.0, p=0.0381) and from 49±3 to 87±6% at 10−5 M (U(7,12)=6.0, p=0.0027) (Fig. 6A). At 72 hr, significant differences in mortality rates were observed for concentrations of indoxacarb from 10−6 to 10−5 M. At this time, the corrected mortality increased from 6±1 to 37±7% at 10−6 M (U(13,16)=0.0, p=0.004), from 11±5 to 94±3% at 3×10−6 M (U(5,6)=0.0, p=0.0080), from 28±5 to 93±4% at 5×10−6 M (U(5,4)=0.0, p=0.0159) and from 92±3 to 99±1% at 10−5 M (U(8,12)=27.0, p=0.0377) (Fig. 6B). The model deviation ratio (MDR) was calculated to define the nature of these associations (Table 3).32,49,50) At 48 hr, the MDR values were higher than 1.3 for three deltamethrin-LNCs/indoxacarb combinations meaning a synergistic effect. Indeed, the MDR values were 3.0, 1.8 and 1.4 for the deltamethrin-LNCs 10−7 M associated with indoxacarb at 3×10−6 M, 5×10−6 M and 10−5 M, respectively. At 72 hr, synergistic effect was observed with deltamethrin-LNCs 10−7 M associated with indoxacarb at 10−6 M, 3×10−6 M and 5×10−6 M (MDR values of 2.2, 2.9 and 2.8, respectively). The best synergistic effect was obtained with deltamethrin-LNCs at 10−7 M combined with indoxacarb at 3×10−6 M.

| MDR | ||
|---|---|---|
| 48 hr | 72 hr | |
| Deltamethrin-LNCs 10−7 M/indoxacarb 10−7 M | na | na |
| Deltamethrin-LNCs 10−7 M/indoxacarb 10−6 M | na | 2.2 |
| Deltamethrin-LNCs 10−7 M/indoxacarb 3.10−6 M | 3.0 | 2.9 |
| Deltamethrin-LNCs 10−7 M/indoxacarb 5.10−6 M | 1.8 | 2.8 |
| Deltamethrin-LNCs 10−7 M/indoxacarb 10−5 M | 1.4 | 0.9 |
This synergy was not observed when deltamethrin was not encapsulated (Fig. 2). To ensure that potentialization of indoxacarb activity was due to encapsulated deltamethrin, control experiment was performed with the association indoxacarb 3×10−6 M and LNCs alone (85 µg/mL). The results indicated a higher mortality at 48 hr (20±4%) (Z(6.9)=2.574, p=0.0604) and at 72 hr (44±2%) (Z(5.9)=2.296, p=0.1300) compared to indoxacarb alone (3±2% and 11±5% at 48 hr and 72 hr, respectively). One may hypothesize that this slight synergistic effect is due to LNCs which act as synergistic agent or to an interaction between indoxacarb and LNC which potentiates indoxacarb activity in insect cells (i.e., increased local concentration of the insecticide). However, the potentialization of indoxacarb by LNCs alone did not reach the synergistic effect obtained with deltamethrin-LNCs at 10−7 M combined with indoxacarb at 3×10−6 M (94±3%) indicating that the synergy is due to deltamethrin encapsulated in LNCs. Indeed, indoxacarb needs to be metabolized to DCJW by esterase/amidase enzymes. This metabolite is a potent voltage-gated sodium channel blocker.22) As previously shown in Periplaneta americana,17) the deltamethrin may modulate voltage-gated sodium channels in aphids and increase intracellular sodium concentration, which could activate the Na/Ca exchanger in reverse mode. The voltage-gated sodium channels may thus be more sensitive to lower concentration of DCJW due to the elevation of intracellular Ca2+ concentration. However, an esterase-induced enzymatic degradation could occur on deltamethrin.17) So nanoencapsulation of deltamethrin could protect its in vivo degradation allowing sufficient time to bioactivate indoxacarb in DCJW. This synergistic effect may be due to deltamethrin protection but also to its sustained release. Indeed, a synergistic effect was observed since 48 hr post ingestion while FRET experiments showed LNCs integrity and storage in stomach of A. pisum larvae until 8 days (Fig. 4). This combination of effects: protection against degradation enzymes, effective delivery or sustained release would allow a synergistic effect with the indoxacarb and reduction of insecticide concentrations, which corresponds to national guidelines.10) Encapsulated deltamethrin could be associated with different other insecticides acting on voltage-gated sodium channel (i.e., pyrethroids). The encapsulation strategy could improve the efficacy of synergistic agent associated with various insecticides.51–53)
5. Comparative studies of the toxicity between deltamethrin-LNCs/indoxacarb and deltamethrin/indoxacarb associated with PBOThe toxicity of the deltamethrin-LNCs 10−7 M/indoxacarb 3×10−6 M association was then compared to the toxicity of deltamethrin 10−7 M/indoxacarb 3×10−6 M associated with the current used synergist piperonyl butoxide (PBO). This experiment enabled to evaluate the in vivo protection of deltamethrin from enzymes degradation by nanocapsules versus PBO. Indeed PBO is known to inhibit the activity of cytochrome P450 enzymes involved in deltamethrin metabolism and thus to be an effective synergist with deltamethrin against resistant insects.54–58) PBO has been used at 1 mg/mL, a non-lethal concentration after 48 and 72 hr ingestion (Supplementary Fig. 3). As expected, at 48 hr, the mortality rates were significantly different between deltamethrin/indoxacarb association (7±5%) and deltamethrin-LNCs/indoxacarb (34±5% with U(4,5)=3.00, p=0.0341) or deltamethrin/indoxacarb/PBO (32±7% with U(4,6)=2.00, p=0.0124) but no significant difference was obtained between mortality rates of deltamethrin-LNCs/indoxacarb and deltamethrin/indoxacarb/PBO (Fig. 7). At 72 hr, the mortality rates were significantly different between deltamethrin/indoxacarb and deltamethrin-LNCs/indoxacarb (94±3% with U(4,5)=0.00, p=0.0050) or deltamethrin/indoxacarb/PBO (55±3% with U(4,6)=0.00, p=0.0080). Theses increased mortality rates are due to the protection of deltamethrin from detoxification enzymes by LNCs or PBO. However, a significant difference was observed between mortality rates of deltamethrin-LNCs/indoxacarb and deltamethrin/indoxacarb/PBO (U(5,6)=0.00, p=0.0043). Our data indicated that encapsulation of deltamethrin allows a better protection from detoxification enzymes and could be an effective alternative to PBO to optimize insecticides efficacy and reduce their doses. Furthermore, the deltamethrin-LNCs/indoxacarb synergistic association represents a promising strategy against resistant pest populations which overproduce detoxification enzymes.

In summary, this study demonstrated that deltamethrin-LNCs could be used with indoxacarb to obtain an efficient synergistic association against A. pisum larvae. Nanoencapsulation represents an alternative to PBO and an interesting strategy to reduce doses of insecticide in pest management. This formulation has shown high stability in vitro against variation of pH, temperature or light, reflecting potential for a successful applicability in field. Nevertheless, complementary approved analyses should be performed in the future to use such nanopesticides in agriculture. Indeed the high stability of this type of formulation could induce negative effects regarding non-target organisms and a long persistence in environment. This issue will have to be checked in the final development of environmentally friendly nanopesticides.
This work was supported by University of Angers for the project ACR 2019 SynPuce. Authors thank Aurélia Rolland and Fabienne Simoneau from the “Plateau Technique Mutualisé Imagerie Cellulaire” (IMAC, SFR QUASAV, Angers, France) for supporting microscopy experiments. Authors also thank Véronique Jean, Julie-Anne Hugel, Eléonore Moreau, Jérôme Bejaud, Marie-Anne Jourdain, Emilie Boulier, Salomée Rousier for their technical assistance.
The authors declare that there is no competing interest.
The online version of this article contains supplementary materials, which are available at https://www.jstage.jst.go.jp/browse/jpestics/.