Abstract
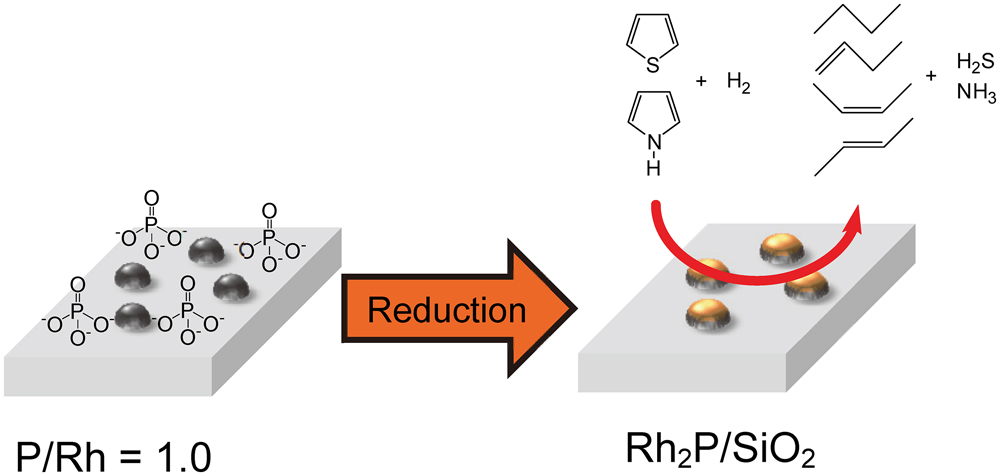
Metal phosphide has been widely investigated as a hydrotreating catalyst. The preparation and performance of noble metal phosphide catalyst was examined to develop new phosphide hydrotreating catalysts. The supports affect reducibility of phosphate as a P precursor. Since phosphate does not strongly interact with SiO2 and TiO2 supports, Rh2P was easily formed on these supports. Furthermore, formation of Rh2P enhanced the hydrodesulfurization (HDS) activity of supported Rh–P catalyst. The type of noble metal (NM) and P/NM ratio also strongly affect formation of noble metal phosphide and HDS activity. Excess P facilitates formation of noble metal phosphides at lower reduction temperature. In contrast, excess P causes the aggregation of noble metal phosphide and formation of phosphorus rich noble metal phosphide. Rh–1.5P/SiO2 catalyst had high and stable activity for HDS reaction. Furthermore, this catalyst showed significantly higher hydrodenitrogenation (HDN) activity than sulfided NiMoP/Al2O3 catalyst. Therefore, Rh2P has great potential as a new hydrotreating catalyst.