2019 Volume 44 Issue 12 Pages 845-848
2019 Volume 44 Issue 12 Pages 845-848
As the field of utilization of organic-inorganic hybrid molecules expands, the toxicology of these compounds is becoming more important. We have shown previously that there is a strong correlation between cytotoxicity and intracellular accumulation detected as metal content, which is modulated by the substituents, of organic-inorganic hybrid molecules. In this study, we investigated the cytotoxicity of pentavalent organoantimony compounds with three phenyl groups on cultured vascular endothelial cells. The results indicated that the cytotoxicity of pentavalent organoantimony compounds was not correlated with the hydrophobicity and intracellular accumulation of these compounds. Therefore, we suggest that hydrophobicity and intracellular accumulation are not necessarily predictive of cytotoxicity in organic-inorganic hybrid molecules.
Organic-inorganic hybrid molecules, namely organometallic compounds and metal complexes, are used as tools for chemical synthesis. Recently, scientists have begun to use these molecules as tools to analyze biological systems based on a bio-organometallic research strategy (Fujie et al., 2016a, 2017, 2019a; Hara et al., 2017, 2018a). Therefore, the importance of toxicology in organic-inorganic hybrid molecules is increasing (Fujie et al., 2016b). Previously, we have shown the toxicological results of trivalent organoantimony compounds; the cytotoxicity and intracellular accumulation of these molecules can be changed by the intramolecular metal element and organic structure (Kohri et al., 2015; Hara et al., 2018b; Nakamura et al., 2017). As antimony is an element that can take not only a trivalent but also pentavalent form, we investigated the relationships among the hydrophobicity, intracellular accumulation, and cytotoxicity of pentavalent organoantimony compounds with three phenyl groups in cultured vascular endothelial cells.
Bovine aortic endothelial cells were purchased from Cell Applications (San Diego, CA, USA). Tissue culture dishes and plates were from AGC Techno Glass (Shizuoka, Japan). Dulbecco’s modified Eagle’s medium (DMEM) and Ca2+- and Mg2+-free phosphate-buffered saline (CMF-PBS) were obtained from Nissui Pharmaceutical (Tokyo, Japan). Fetal bovine serum (FBS) and a bicinchoninic acid (BCA) protein assay kit were purchased from Thermo Fisher Scientific (Waltham, MA, USA). CytoTox 96 Non-Radioactive Cytotoxicity Assay, a lactate dehydrogenase kit, was obtained from Promega (Madison, WI, USA). May-Grünwald Giemsa stain solutions were purchased from Merck KGaA (Darmstadt, Germany) and other reagents of the highest grade available were obtained from Nacalai Tesque (Kyoto, Japan).
Synthesis of organoantimony compoundsTriphenylantimony (Ph3Sb), triphenylantimony dichloride (Ph3SbCl2), and triphenylantimony diacetate [Ph3Sb(OAc)2] were purchased from Tokyo Chemical Industry. Triphenylantimony difluoride (Ph3SbF2), triphenylantimony dibromide (Ph3SbBr2), and pentaphenylantimony (Ph5Sb) were prepared according to the reported procedure or its modified methods (Kitamura et al., 2017; Fujiwara et al., 1996).
Cell culture and treatmentsVascular endothelial cells were cultured in a humidified atmosphere of 5% CO2 at 37°C in DMEM supplemented with 10% FBS until confluent. They were then transferred into 24-well culture plates and cultured until confluent. The medium was then discarded and the cells were washed with serum-free DMEM. Next, the cells were treated with organoantimony compounds (5, 10, and 25 µM) for 24 hr. After treatment, the cells were used for the following experiments. The structures of organoantimony compounds used in this study and their CLogP value calculated by ChemDraw Ultra 12.0 (CambridgeSoft, Cambridge, MA, USA) are shown in Fig. 1.

The structures of organoantimony compounds and CLogP values of the compounds used in this study.
After a 24-hr incubation, the conditioned media were harvested and aliquots were used for the determination of lactose dehydrogenase (LDH) activity according to the CytoTox 96 Non-Radioactive Cytotoxicity Assay. The cell layers were washed with CMF-PBS and stained with a May-Grünwald Giemsa stain for morphological observation.
Intracellular accumulation of antimonyThe samples were prepared according to our previous report (Hara et al., 2018b). The content of the antimony atom was then analyzed using inductively coupled plasma mass spectrometry (ICP-MS) (Nexion 300S, PerkinElmer, Waltham, MA, USA). The ICP-MS conditions were optimized for a plasma output of 1600 W, a plasma gas flow of 18.0 L/min, and a nebulizer gas flow rate of 0.94 L/min. Another portion of the cell lysate was analyzed for protein content using the BCA protein assay kit to express the content of antimony as nmol/mg protein.
Statistical analysisThe data were analyzed for statistical significance by analysis of variance (ANOVA) and Bonferroni’s multiple t-test, when possible. P values of less than 0.05 were considered statistically significant.
We examined whether pentavalent organoantimony compounds exhibited cytotoxicity or not in vascular endothelial cells. The structures with CLogP values of tested compounds are shown in Fig. 1. Ph3Sb was used as the trivalent organoantimony control. No morphological change was observed in the cells after treatment with Ph3SbF2 and Ph5Sb, as well as Ph3Sb at 25 µM and under (Fig. 2A). However, a collapse occurred in vascular endothelial cell monolayers after treatment with Ph3SbCl2, Ph3SbBr2, and Ph3Sb(OAc)2 at 25 µM. The leakage of LDH from the cells exposed to 25 µM of Ph3SbCl2, Ph3SbBr2, and Ph3Sb(OAc)2 significantly increased (Fig. 2B), which supported the morphological observation that these compounds were toxic to vascular endothelial cells. The cytotoxicity of these compounds was not correlated with their CLogP values, suggesting that the hydrophobicity cannot be necessarily predictive of cytotoxicity in organic-inorganic hybrid molecules.

Cytotoxicity of organoantimony compounds in bovine aortic endothelial cells. [A] Morphological observation of bovine aortic endothelial cells after treatment with organoantimony compounds. [B] The leakage of LDH from the cells into the culture medium. The values are means ± S.E. of four samples. Significantly different from the corresponding control, *P < 0.05,**P < 0.01.
We next investigated whether the cytotoxicity of pentavalent organoantimony compounds with three phenyl groups was related to accumulation within vascular endothelial cells. Overall, the intracellular accumulation of the compounds detected as antimony compounds increased in a concentration-dependent manner. There was no correlation between the antimony accumulation and cytotoxicity in these tested compounds. In addition, the relationship between the CLogP value (Fig. 1) and the antimony accumulation was also not correlated (Fig. 3). These results suggest that organic-inorganic hybrid molecules can accumulate within vascular endothelial cells independently of the hydrophobicity.
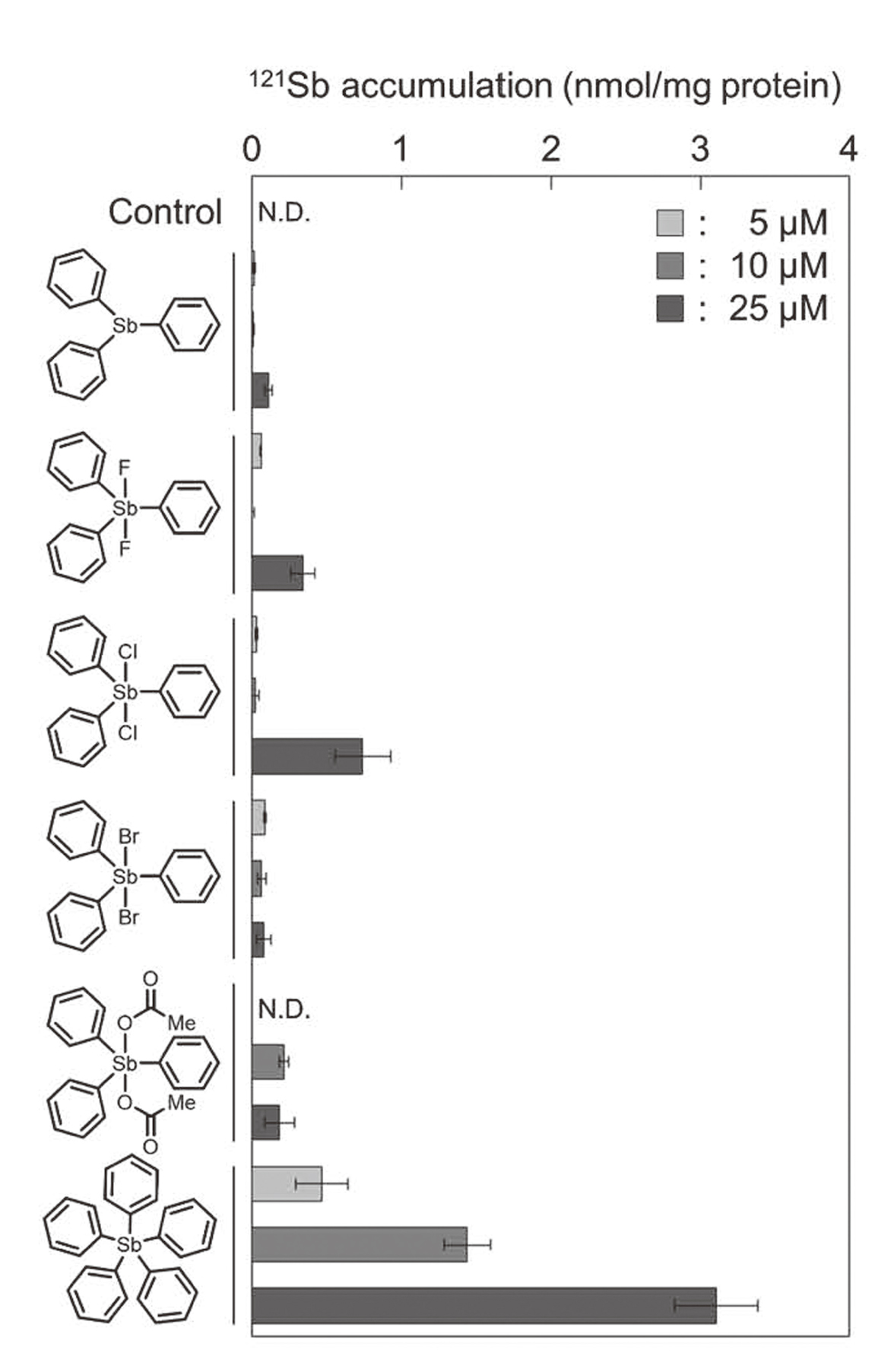
Intracellular accumulation of organoantimony compounds in vascular endothelial cells. Bovine aortic endothelial cells were treated with organoantimony compounds. (N.D. indicates not detected.) The values are means ± S.E. of three samples.
Our research group has previously reported the cytotoxicity of organic-inorganic hybrid molecules in vascular endothelial cells (Murakami et al., 2015; Kohri et al., 2015; Hara et al., 2016, 2018b; Fujie et al., 2019b). These reports indicate that, in general, organic-inorganic hybrid molecules that easily accumulate within the cells exhibit strong cytotoxicity. The present results demonstrated that there are cases in which the cytotoxicity of organic-inorganic hybrid molecules is independent of intracellular accumulation and hydrophobicity, as well as the molecular structure and type of intramolecular metal. Our previous results (Kohri et al., 2015; Murakami et al., 2015; Hara et al., 2016; Nakamura et al., 2017) also show that biological activities, including cytotoxicity, of either the molecular structures or that of the metals that make up the hybrid molecules cannot be used to predict the biological activities of such hybrid molecules. This study revealed a new characteristic of organic-inorganic hybrid molecules—interrelationship among the cytotoxicity, intracellular accumulation, and hydrophobicity—from the viewpoint of toxicology.
This work was supported by JSPS KAKENHI Grant Numbers JP 19K07005 (to S.Y.), JP 18K06638 (to C.Y.), and JP 19K07089 (to T.K.).
Conflict of interestThe authors declare that there is no conflict of interest.