2022 Volume 71 Issue 1 Pages 1-12
2022 Volume 71 Issue 1 Pages 1-12
The uncontrolled growth of blood vessels is a major pathological factor in human eye diseases that can result in blindness. This effect is termed ocular neovascularization and is seen in diabetic retinopathy, age-related macular degeneration, glaucoma and retinopathy of prematurity. Current treatments for these diseases include laser photocoagulation, topical injection of corticosteroids, intravitreal injection of anti-vascular endothelial growth factor (anti-VEGF) agents and vitreoretinal surgery. Although strategies to inhibit VEGF have proved to be dramatically successful in some clinical studies, there remains the possibility of significant adverse effects regarding the blockade of crucial physiological roles of VEGF and the invasive nature of the treatments. Moreover, it is evident that other pro-angiogenic factors also play important roles in the development of these diseases, as seen in cases in which anti-VEGF therapies have failed. Therefore, new types of effective treatments are required. In this review, we discuss a promising strategy for the treatment of ocular neovascular diseases, i.e., the inhibition of hypoxia-inducible factor (HIF), a master regulator of angiogenesis. We also summarize promising recently investigated HIF inhibitors as treatments for ocular diseases. This review will facilitate more comprehensive approaches to understanding the protective aspects of HIF inhibition in the prevention of ocular diseases.
Ocular ischemic neovascular diseases currently constitute the most common causes of moderate and severe visual loss.1 These diseases can be broadly divided into two categories: retinal neovascular diseases and subretinal neovascular diseases.1 Retinal neovascular diseases involve neovascularization from retinal vessels, whereas subretinal neovascular diseases are associated with subretinal neovascularization that occurs in the avascular outer retina and subretinal space. Retinal neovascular diseases generally include diabetic retinopathy2 and retinopathy of prematurity,3 and subretinal neovascular diseases include age-related macular degeneration.4
To date, treatments for these diseases consist of laser photocoagulation, topical injection of corticosteroids, intravitreal injection of anti-vascular endothelial growth factor (anti-VEGF) agents and vitreoretinal surgery.5 Although VEGF inhibition strategies have proven to be effective in some clinical studies, it is evident that other pro-angiogenic factors play an important role in these diseases, as reported in relation to cases in which anti-VEGF therapies have failed.6,7 Briefly, one report demonstrated that SDF-1/CXCR4 may contribute to the involvement of bone marrow-derived cells that work with VEGF to promote the development of neovascularization in the eye in cases of both retinal and subretinal neovascularization.8 Another report indicated that paracrine collaboration of IGF-1 and VEGF may contribute to the development of choroidal neovascularization.9 Angiopoietins and their interactions with VEGF or other angiogenic factors also have been postulated to regulate pathological vascular development, maintenance and permeability in the eye.10,11,12 In addition to angiogenic factors, several reports have suggested that increased monocyte chemoattractant-1 (MCP-1/CCL2) levels in the eye may enhance diabetic retinopathy13; indeed, upregulation of CCL2 has been consistently reported in the vitreous fluid of patients with diabetic retinopathy and ischemic retinopathies.13,14,15,16,17,18 Finally, the long-term use of anti-VEGF therapies has the possibility of significant adverse effects regarding blockade of crucial physiological roles of VEGF in the eye.19,20 Therefore, new types of effective treatments are desirable.
Hypoxia-inducible factor (HIF) is a master regulator of hypoxia-responsive genes that act against hypoxia.21 Hypoxia-responsive genes are involved in angiogenesis, erythropoiesis, energy metabolism and inflammation.22,23 Therefore, HIF can induce pathological pro-angiogenic gene expressions (including that of VEGF-A) under retinal hypoxia, ultimately leading to the development of ocular ischemic neovascular diseases.24,25 Herein, we describe the inhibition of HIF as a promising therapeutic strategy for ocular diseases and summarize promising recently investigated HIF inhibitors as treatments for ocular ischemic neovascular diseases.
Many organisms use oxygen to produce ATP as an energy source to sustain themselves. The majority of all living things, including humans, cannot maintain their metabolism without oxygen. Organisms are sometimes exposed to transient or chronic hypoxia resulting from environmental factors or diseases. As a response to such factors and diseases, under hypoxic conditions, HIF exerts an important role in adaptive regulation of energy metabolism by triggering the switch from mitochondrial oxidative phosphorylation to anaerobic glycolysis.26
HIF is a basic helix–loop–helix DNA binding protein of the Per-ARNT-Sim family (bHLH-PAS).27 HIF was discovered by Semenza et al. in 1992 as a hypoxia-dependent inducer of erythropoietin in a liver cancer cell line (Hep3B).28 In 1995, HIF-1 was reported to be a heterodimer of HIF-1α and HIF-1β/ARNT (an aryl hydrocarbon receptor nuclear translocator).23,29,30,31 Subsequently, HIF-2α/EPAS2 and HIF-3α were identified.28,32 Among the HIF-α subunits, HIF-1α and HIF-2α have an N-terminal trans-activation domain (N-TAD) and a C-terminal transactivation domain (C-TAD) on the C-terminal side, which is involved in transcriptional activation (Fig. 1). In contrast, HIF-3α does not possess C-TAD (Fig. 1). Multiple variants of HIF-3α have been characterized.33,34 In mouse variants, neonatal embryonic PAS and inhibitory PAS domain (IPAS) proteins have been identified.35 Moreover, there is a large number of human variants of HIF-3α.35 HIF-3α has relatively weak transcriptional activity because it does not include C-TAD. IPAS behaves as a dominant negative form of HIF.34,36,37 Each HIF-α subunit contains an oxygen-dependent degradation domain that regulates the stability of HIF-α.33,34 In contrast to HIF-α family members, HIF-1β is stably expressed in cells and allows HIF-α family members to bind to DNA and stimulate transcription of their target genes.27

Schematic representation of human HIF structures. HIFs are heterodimers (i.e., made up of two subunits): one α (HIF-1α, HIF-2α or HIF-3α) and one β (HIF-β). Each subunit possesses different functional domains implicated in DNA binding [basic helix–loop–helix domain–Per-ARNT-Sim motif (bHLH-PAS)], dimerization (bHLH-PAS), protein stability [oxygen-dependent degradation domain (ODD) and N-terminal transactivation domain (N-TAD)], cofactor interactions [N-TAD and C-terminal transactivation domain (C-TAD)], and transcriptional activity (N-TAD and C-TAD).
Under normoxia, HIF-α is hydroxylated by prolyl hydroxylases (PHD) (Fig. 2). Prolylhydroxylated HIF-α is ubiquitinated by E3 ubiquitin ligase complex (composed of pVHL, Elongin B/C, Cul2 and Rbx1), resulting in the rapid proteasome-dependent degradation of HIF-α (Fig. 2). In contrast, under hypoxic conditions, the activity of PHD is reduced and HIF-α is stabilized (Fig. 2). HIF-α then translocates into the nucleus to form heterodimers with HIF-1β that then bind to the hypoxia-response element, which is located upstream of HIF target genes (Fig. 2). Furthermore, the C-TAD region is important for optimal transcriptional activity of HIF. However, HIF can be induced to a lesser extent without binding to the CREB-binding protein (CBP)/p300. Therefore, recruitment of CBP/p300 enhances HIF-mediated transcriptional activity (Fig. 1 and Fig. 2). Despite the constant biosynthesis of HIF-α, under normal oxygen conditions, the transcriptional activity of HIF is kept very low by the mechanism, i.e., the oxygen-dependent hydroxylation of asparagine by the asparaginyl hydroxylase factor inhibiting HIF-1 (FIH-1) inhibits the interaction of HIF with CBP/p300.38
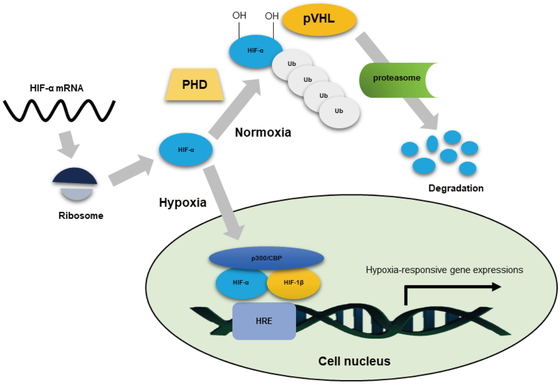
Oxygen-dependent regulation of HIF-α. Physiologically, HIF-α mRNA is translated to HIF-α protein through ribosome protein synthesis. Under normoxia, the product of O2-dependent hydroxylation of HIF-α by prolyl hydroxylases (PHD) is marked for binding by von Hippel–Lindau protein (pVHL), which is the recognition component of ubiquitin (Ub). Ubiquitylated HIF-α is degraded by the proteasome. As a result, hypoxia-responsive gene expressions are not activated. Under hypoxic conditions, the activity of PHD decreases and pVHL cannot bind to HIF-α, resulting in a decreased rate of degradation of HIF-α protein. Non-degraded HIF-α protein can enter the cell nucleus, and p300 and CREB binding protein (CBP) can bind to HIF-α with HIF-1β, which finally allows transcriptional activation of HIF target genes. HRE, hypoxia responsive enhancer elements.
HIF-mediated cellular responses to low levels of oxygen fall into two categories: short- and long-term adaptation. For short-term adaptation, the oxygen supply can be rapidly increased by HIF activation through upregulation of the vasodilatory enzyme iNOS (inducible nitric oxide synthase).39 The enzymatic product of iNOS, nitric oxide, induces a fast increase in blood flow via the relaxation of vascular smooth muscle cells.40 Oxygen homeostasis can also be modulated by an increase in the glycolysis machinery through induction of glycolytic enzymes such as glucose transporter-1 (GLUT1)/SLC2A141,42 and suppression of mitochondrial respiration through induction of pyruvate dehydrogenase kinase-1 (PDK1).43,44 GLUT1 and PDK1 are well known HIF target genes. When it comes to long-term adaptation, a series of hypoxia-stimulated angiogenesis processes are initiated through induction of pro-angiogenic genes such as VEGF-A, angiopoietin-1, angiopoietin-2, platelet-derived growth factor, basic fibroblast growth factor and monocyte chemoattractant protein-1 (MCP-1/CCL2), which finally induces endothelial cell proliferation, sprouting, migration, adhesion and tube formation as well as vascular permeabilization.45,46 Because these factors are regulated by HIF, HIF can be regarded as the master regulator of angiogenesis. This process would appear to resolve the matter of chronic low oxygen levels in tissues. However, numerous pathological diseases related to hypoxia have a general basis in this process.47,48,49,50,51 Pathological ocular diseases related to hypoxia include diabetic retinopathy (DR), age-related macular degeneration (AMD), glaucoma and retinopathy of prematurity (ROP); these conditions are discussed separately below.
AMD occurs when abnormal new blood vessels develop under the retina, a process called choroidal neovascularization (CNV). AMD leads to severe visual loss in the elderly population. There remains neither a strongly effective preventive treatment nor a cure for AMD.52,53 Although the development of AMD is associated with complex pathological mechanisms, increasing evidence suggests that dysfunction of retinal pigment epithelium (RPE) may be mainly involved in neovascular and atrophic forms of AMD.54 RPE is a monolayer of polarized cells that resides below the retina and interacts with photoreceptor cells to achieve retinal homeostasis.55 The outer blood–retina barrier can be modulated by RPE via regulation of the delivery of nutrients and oxygen to the outer retina and the clearance of metabolic waste from the outer retina. RPE produces growth factors such as VEGF to support the physiologies of the retina and choriocapillaris.54,56,57 However, aging and increased oxidative stress can result in dysfunction of RPE, ultimately leading to RPE producing pathological amounts of growth factors, especially VEGF.58,59 This process may be a pathological cause of the development of CNV, which finally leads to AMD.60 To investigate this process, we attempted to control RPE via inhibition of the HIF/VEGF-A axis in RPE. We found a significant reduction of the CNV volume in a RPE-specific Hif-1α conditional knockout mouse model of CNV compared with that of the CNV volume in the age-matched control mouse model of CNV.61 Furthermore, by carrying out extensive screenings of drugs and food ingredients, we discovered several HIF inhibitors (lactoferrin, rice bran, vitamin B6, Garcinia cambogia and hydroxycitric acid) and demonstrated that diet supplemented with these ingredients can reduce the CNV volume in a mouse model of CNV and reduce VEGFA mRNA expression under CoCl2-induced hypoxia.61,62,63 Lee et al. investigated the effects of resveratrol on HIF-1α and VEGF expressions in human ARPE-19 cells and a mouse model of CNV.64 Resveratrol, a natural compound found in grapes, has been suggested to suppress VEGF in various cancer cells, possibly through HIF-1α inhibition.65,66,67 In ARPE-19 cells, resveratrol significantly reduced HIF-1α and VEGF-A expressions by blocking the PI3K/Akt/mTOR signaling pathway and promoting proteasomal HIF-1α degradation; moreover, oral administration of resveratrol reduced the CNV volume in a mouse model of CNV.64 Song et al. found that chrysin (5,7-dihydroxyflavone), another flavonoid naturally present in many fruits and vegetables, inhibited HIF-1α and VEGF expressions with an accompanying reduction of the CNV volume in a rat model of CNV.68 Using a non-pharmacological approach, Koo et al. attempted to use therapeutic gene editing in a mouse model of CNV.69 The intravitreal delivery of CRISPR RNA-guided endonuclease LbCpf1 (derived from Lachnospiraceae bacterium ND2006) targeted to Hif-1α or Vegfa reduced the volume of CNV as efficiently as aflibercept (an anti-VEGF drugs currently in clinical use).69 Furthermore, LbCpf1 targeted to Hif-1α or Vegfa exerted long-term therapeutic effects, unlike aflibercept.69 In summary, even though the mode of action in vivo requires further investigation, control of the HIF/VEGF-A axis in the eye could have therapeutic value in the management of CNV and ultimately suppress the development of AMD.
Retinopathy of prematurityROP is an ocular disease of prematurely born babies, usually seen in those having received neonatal intensive care in which oxygen therapy is administered because of insufficient lung development.70 In contrast to the damaging vessels found in AMD, retinal blood vessels grow disorganized in ROP and result in scarring and retinal detachment, which may lead to blindness in serious cases.70 The development of retinal neovascularization also involves complex pathological mechanisms similar to that of CNV. The involvement of multiple interlinked structural and functional changes caused by abnormal crosstalk between retinal neurons, glial cells and vasculatures has been suggested as the mechanism of ROP development.71 Neurons (with glial cells) act as sensors of oxygen and regulators of vasculatures by interacting with various types of vessels in the central nervous system,72 and impairment of their crosstalk results in uncontrolled neurovascular homeostasis.72,73 In the eye (a component of the central nervous system), the activation of HIFs in neurons (especially HIF-1α) and glia cells (especially HIF-2α) are suggested to have critical pathological roles in retinal vascular diseases.74 We demonstrated that no change in Hif-2α mRNA expression and only a slight increase in HIF-2α protein expression was seen in the ischemic retina, whereas expressions of Hif-1α mRNA and HIF-1α protein showed significant increases along with upregulation of Vegf mRNA expression in a mouse model of oxygen-induced retinopathy (OIR), a condition that mimics ROP.75,76 Furthermore, we demonstrated that Hif-1α and Vegfa in retinal neuronal cells were responsive to CoCl2-induced hypoxic stress, whereas Hif-2α was not induced.77 This finding suggests that the HIF-1α/VEGF-A axis in retinal neurons may be a major regulator of retinal neovascularization. In contrast, Weidemann et al. demonstrated that astrocyte-derived VEGF may be essential for retinal neovascularization in a model of OIR,78 and Korovina et al. suggested that HIF-2α in hematopoietic cells may contribute to the development of retinal neovascularization in a model of OIR.79 Against this background, for a better understanding of the pathological mechanisms of retinal neovascularization, comprehensive studies need to be continued regarding the effects of the main subtypes of HIF on the development of retinal neovascularization as well as investigations of VEGF-producing cell types and systemic vascular cell conditions.
As a pharmacological treatment for ROP, we suggested topotecan, a topoisomerase I inhibitor that decreased HIF-1α stabilization in a mouse model of OIR with a reduction in upregulated Vegfa and Angpt2 mRNA expressions in the ischemic retina, which ultimately prevented retinal neovascularization.75,76 Doxorubicin (a widely used HIF-1α inhibitor) also prevented retinal neovascularization in a mouse model of OIR, even though the mode of action of doxorubicin was not directly stated in the study.75,76 We also attempted to use marine products to prevent retinal neovascularization in a mouse model of OIR.80 Among various materials from marine fish, Decapterus tabl ingredients were shown to possess HIF inhibitory effects under dimethyloxalylglycine-induced hypoxic conditions.80 Furthermore, oral administration of Decapterus tabl ingredients suppressed retinal neovascularization in a mouse model of OIR even though the mode of action in vivo and the effector compounds in the ingredients have not been clarified.80
In addition to marine products, we focused on investigating novel HIF inhibitors from mushroom products and found that 2-azahypoxanthine (AHX), a fairy chemical from the fungus Lepista sordida,81,82 had an inhibitory effect on HIF activation in retinal cells and suppressed Vegfa mRNA upregulation under CoCl2-induced hypoxic conditions.77 Oral administration of AHX significantly suppressed retinal neovascularization in a mouse model of OIR.77 Yoshida et al. applied digoxin (a cardiac glycoside) to suppress retinal neovascularization by inhibiting HIF activation83 based on the suggestion that digoxin blocks tumor growth via HIF-1α synthesis inhibition.84 In the ischemic retinas in a model of OIR, digoxin selectively inhibited expressions of HIF target genes such as Bnip3, Glut1 and Epo and also inhibited expressions of hypoxia-induced angiogenic growth factors and their cognate receptors such as Vegfa, Sdf-1 (stromal cell-derived factor 1), Scf (stem-cell factor), Pdgf-b (platelet-derived growth factor subunit B), Pgf (placental growth factor), Vegfr2 (vascular endothelial growth factor receptor 2), Cxcr4 (C-X-C chemokine receptor type 4), Cd117 and Pdgfr-b (platelet-derived growth factor receptor subunit B).83 Usui-Ouchi et al. designed an allosteric peptide inhibitor of HIFs to examine whether HIF inhibition peptide therapy is applicable to counteract retinal neovascularization.85 The peptide design concept was the disruption of HIF binding to coactivators CBP/p300 to reduce pathological HIF-mediated hypoxia-responsive gene expressions as well as HIF-related pro-inflammatory gene expressions. The peptide inhibitor of HIFs reduced the upregulated expressions of Vegfa, Epo, Tnf-α, Ccl2, Ccl3 and Il-1β in ischemic retinas and effectively regulated retinal neovascularization in a mouse model of OIR.85 Furthermore, a combination of this peptide inhibitor and a reduced concentration of the anti-VEGF agent aflibercept showed promising therapeutic effects on the regulation of retinal neovascularization.85
By taking a different approach from the direct application of HIF inhibitors, we demonstrated that pemafibrate could prevent retinal neovascularization via upregulation of serum levels of fibroblast growth factor 21 (FGF21), a possible HIF inhibitor, in a mouse model of OIR.75 Pemafibrate is a selective peroxisome proliferator-activated receptor α modulator that was recently approved in Japan as a powerful therapeutic agent against hyperlipidemia. Pemafibrate activated PPARα in the liver, resulting in an increase in serum levels of FGF21.86,87 Previously, Fu et al. showed that FGF21 administration could suppress pathological retinal neovascularization in a mouse model of OIR.88 Following this, we found a significant increase in serum levels of FGF21 by pemafibrate administration in a model of OIR.75 Moreover, we suggested that a decrease in retinal HIF-1α immunoreactivities along with a reduction in Vegf mRNA expression in a mouse model of OIR may have a strong correlation with FGF21 induction by pemafibrate.75 Chen et al. examined whether retinal neovascularization could be modulated by treatment with fenofibrate, another PPARα agonist.89 Fenofibrate also ameliorated retinal neovascularization in a rat model of OIR and inhibited tube formation and migration in endothelial cells.89 The effects of fenofibrate can be partially explained by the blockade of activation of HIF-1α and upregulation of VEGF.89 Taken together, these reports suggest that the control of HIF-1α, VEGF and other pathological angiogenic factors could have a great impact on the regulation of retinal neovascularization.
Diabetic retinopathyDR is one of the most serious and common complications of diabetes in subjects with type 1 or type 2 diabetes mellitus.90 It is a chronic microvascular complication in the retina caused by chronically uncontrolled glucose levels in the body during the development of diabetes.91,92 The chronic microvascular damage leads to severe hypoxic conditions and results in retinal neovascularization.91,92 Chronic severe hypoxic stress in the retina can induce retinal detachment, which may result in loss of vision.92 Rodrigues et al. showed that HIF-1α and VEGF-A were expressed in the retinas of proliferative sickle retinopathy patients, higher than that in the retinas of controls. This finding implies that HIF-1α might be important for the development of DR.93 Wang et al. determined that the levels of VEGF-A and HIF-1α in the vitreous fluid of patients with proliferative DR were higher than those in control subjects.94 This also implies that VEGF-A and HIF-1α could be associated with the pathological angiogenic processes of DR.94 Yamada et al. focused on genetic variation in HIF-1α by examining all regions of the gene for single-nucleotide polymorphisms in type 2 diabetes patients and performed association studies in Japanese type 2 diabetes patients with or without retinopathy.95 Mutated HIF-1α (P582S) was found to be associated with type 2 diabetes and had a higher transcriptional activity than the wild-type, which implies that HIF-1α may have a strong association with the occurrence of type 2 diabetes.95 Even though no association with retinopathy was detected with the P582S HIF-1α mutation, Yamada et al. suggested that P582S HIF-1α should be assessed in larger studies to confirm this finding.95 Ekberg et al. suggested that the P582S HIF-1α mutation may have a protective effect against the development of severe DR with a strong risk reduction, even when adjusting for known risk factors for DR such as diabetes duration, hyperglycemia and hypertension.96 This finding implies that HIF-1α may be important for the development of severe DR. In summary, the potential roles of HIF-1α in the development of DR have been much discussed in clinical studies.
Preclinical animal studies of DR are problematic because the currently available experimental models do not clearly and exactly recapitulate the entire pathophysiology of changes in neurons and vasculatures that occur at each stage of human DR.97,98 For example, the streptozotocin-induced diabetic model (one of the most widely used experimental models for diabetes) has a limited ability to mimic proliferative DR, which means that neovascularization is not clearly seen in the model.99,100,101,102,103 The galactose feeding approach to diabetic modeling requires a long period to develop DR in mammals,104,105,106 and proliferative DR is still rarely formed in the model using this approach. Pancreatectomy, which directly removes the pancreas, is only applicable to large animals such as cats or monkeys, but these have large variations among individuals.107,108 The direct injection of VEGF to the eyes to induce neovascularization is not mediated by hyperglycemia and, after VEGF is metabolized physiologically, the effects of VEGF could easily disappear.109 A mouse model of OIR (also used for studying ROP) has been classically used because neovascularization is highly inducible and applicable to rodents without technical difficulties.110 However, this method is also not DR specific, and only newborn rodents are available for induction of retinal neovascularization. The development of clinically relevant murine models, e.g., combining ischemic experimental models with streptozotocin injection111 or high-fat diet supplementation112 to increase glucose levels, must first be established to better understand how HIF modulation has protective (or deleterious) effects in the development of DR.
GlaucomaGlaucoma is a group of eye conditions that results in damage to the optic nerve, which leads to vision loss. This disease is characterized by high intraocular pressure.113 Elevated eye pressure can be induced by overproduction of the aqueous humor that flows throughout the eye or by dysfunction of the drainage system of the aqueous humor through the trabecular meshwork at the angle where the iris and cornea meet.113 Even though the occurrence of neovascular glaucoma is lower than that of other types of glaucoma, neovascular glaucoma is known as a potentially blinding secondary glaucoma114 characterized by the development of new vessels over the iris that obstruct outflow of the aqueous humor and thereby increase intraocular pressure.115 Interestingly, elevated intraocular pressure was suggested to increase HIF-1α expression in the retina in an experimental model of glaucoma116; furthermore, there was an increase in immunoreactivities of HIF-1α in the retina and optic nerve head of glaucomatous human eyes compared with those of control eyes.117 Consequently, we investigated whether HIF-1α inhibition has protective effects on the retina in a mouse model of glaucoma.118 Using the general HIF-1α inhibitor topotecan, we demonstrated that impairment of visual function in a mouse model of glaucoma could be suppressed by topotecan administration via reduction in stabilized HIF-1α protein levels in the retina.118 Furthermore, we found that another HIF inhibitor, halofuginone (a synthetic derivative of febrifugine), could prevent neurodegeneration in a mouse model of glaucoma via inhibition of HIF-1α activation in the retina.119 Compared with the involvement of HIF in AMD and/or ROP, the effects of HIF modulation in glaucoma have been less studied. Therefore, more studies are needed to better understand how HIF inhibition could have protective effects in glaucomatous retinas.
Corneal diseasesIn addition to the retina and choroid, corneal neovascularization is also a sight-threatening condition caused by new vessel formation from the limbus and its invasion into the cornea, resulting in ocular inflammation, infection and hypoxia.120,121 HIF-1α expression has been detected in corneal neovascularization, and VEGF-A, also known to be a main pro-angiogenic factor in corneal neovascularization, is under regulation of the HIF-1α signaling pathway in the cornea.122,123 Therefore, HIF inhibition has been suggested as a promising target for treating corneal neovascularization. Chen et al. used shRNA targeting HIF-1α to suppress VEGF-A expression and corneal neovascularization in a mouse model of closed-eye contact lens wear.122 Fu and Xin demonstrated that silencing HIF-1α or silencing VEGF-A inhibited corneal neovascularization, while double-target interference of VEGF and HIF-1α improved pathological changes regarding corneal neovascularization to a greater extent.124 Kwon et al. examined whether rapamycin (an effective antifungal agent125) could inhibit corneal neovascularization in an alkaline-burned corneal mouse model.126 As a result, mRNA expressions of substance P and Hif-1α were inhibited by rapamycin treatment (1–3 days) with a reduction in VEGF-A expression on day 7 in corneal tissues.126 This implies that rapamycin could be useful as an angiogenic regulator in the treatment of corneal neovascularization. Zhang et al. focused on identifying the profile of miRNA expressions in neovascularized corneas after alkali burn injuries to the eye and found that miR-21 in alkali-burned corneas showed the greatest increase.127 Using subconjunctival injection of antagomir-21, an inactivator of miR-21, progression of neovascularization was attenuated by inhibiting expressions of VEGF-A and HIF-1α.127 Taken together, this evidence suggests that targeting HIF-1α could also be useful for the management of corneal neovascularization.
In this review article, we summarized recent studies of HIF inhibition therapy mainly for ocular neovascular diseases. Even though the pathogenesis of each disease contains many additional complex mechanisms associated with age, genetics and environmental factors, the activation of HIFs (especially HIF-1α) is shared as a possible major pathological factor in the development of ocular neovascular diseases (Fig. 3A). HIF inhibition could constitute an ideal approach because the pathological role of pro-angiogenic factors (especially VEGF-A) can be directly targeted while the physiological role of these factors can be maintained to facilitate ocular homeostasis (Fig. 3B). We hope that this review promotes a comprehensive understanding of the protective roles of HIF inhibitors in preventing the development of diseases that can cause blindness in humans.

Working hypothesis for the roles of HIF in ocular diseases. (A) Hypoxia and ischemia resulting from various factors such as aging, genetics and environmental factors lead to activation of HIFs (especially HIF-1α) in retinal cells and in retinal pigment epithelium (RPE), depending on the disease. HIF activation induces pro-angiogenic gene expressions of factors such as VEGF, the upregulation of which causes neovascularization. AMD, age-related macular degeneration; DR, diabetic retinopathy; ROP, retinopathy of prematurity; RGC, retinal ganglion cell; CNV, choroidal neovascularization. (B) For the prevention of ocular neovascular diseases, HIF can be targeted to reduce pathological levels of VEGF while maintaining physiological levels of VEGF needed for ocular homeostasis.
We thank K. Kurosaki and A. Kawabata for critical discussions. This work was supported by Grants-in-Aid for Scientific Research (KAKENHI, number 15K10881 and 18K09424) from the Ministry of Education, Culture, Sports, Science and Technology (MEXT) to T.K.
The authors declare that there are no conflicts of interest.