Abstract
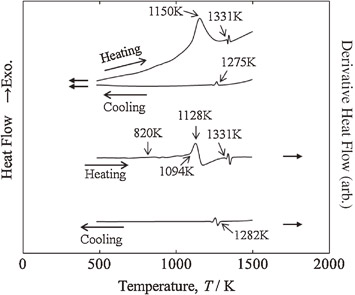
Oxidation behavior and subsequent phase decomposition of Au-55 mol%Ti high temperature shape memory alloy are investigated for heating oxidation up to 1773 K in Ar-50 vol%O2 using a simultaneous thermogravimetry combined with differential scanning calorimetry (TG-DSC). Au-55 mol%Ti powder specimen begins to be oxidized at 802 K and the oxidation reaction is most active at 1150 K. Since the oxidation start temperature is comparable to the martensitic transformation temperatures, the oxidation must be a problem for practical high-temperature long-term applications. An equilibrium phase reaction appears near 1346 K which is melting of Au produced by the reaction of AuTi + O2 → Au + TiO2. The oxidation reaction and products assessed by the TG-DSC measurement are in good agreement with those obtained by X-ray diffraction analysis.