2018 Volume 59 Issue 1 Pages 61-65
2018 Volume 59 Issue 1 Pages 61-65
Mechanical and tribological properties of powder metallurgy (PM) α-titanium (Ti) materials with dissolved nitrogen atoms were evaluated in this study. Pin-on-disk wear test was carried out under dry condition, where a SKD61 disk specimen was used as a counter material. The elemental mixture of Ti and TiN powders was compacted and sintered in vacuum, and then extruded to the full-dense PM Ti rods. During sintering in vacuum, TiN particles were completely decomposed via reaction with Ti powder. Nitrogen atoms originated from TiN were dissolved into α-Ti matrix, and resulted in the remarkable improvement of micro-hardness and tensile strength. The additional heat treatment on the sintered Ti materials was effective to improve further elongation in tensile test because the localization of dissolved nitrogen atoms was decreased. The friction coefficient of nitrogen dissolved Ti material was extremely lower and more stable compared to pure Ti specimen employed as a reference material. The wear loss of the former was significantly smaller than that of the latter specimen. This is because of superior wear resistance of α-Ti material with nitrogen solid-solution due to a large increment of micro-hardness of Ti matrix.
This Paper was Originally Published in Japanese in J. Jpn. Soc. Powder Powder Metallurgy 64 (2017) 275–280.
Titanium (Ti) is rich in the earth as oxide minerals, and the ninth largest number of the crust elements. Since Ti alloys have high specific strength, excellent corrosion resistance and biocompatibility, they are widely used in various industries, for example aircraft components, chemical plant products, medical devices. The alloying elements are necessary to improve the mechanical properties of Ti materials, however, they often mean expensive rare metals or harmful and toxic elements for human1). Recently, instead of such elements, a new materials design for Ti materials strengthening by using oxygen, nitrogen, hydrogen and carbon (named as “ubiquitous elements”) has been studied2). In our research group, the solid-state process based on powder metallurgy (PM) route to fabricate Ti materials containing ubiquitous elements by solid-solution strengthening was established3–6). It is reported that mechanical strength of PM Ti materials is extremely improved by nitrogen solid-solution mechanism5) because α-Ti has a large solubility of nitrogen atoms7). On the other hand, Ti materials have some disadvantages regarding tribological wear properties such as severe wear damages, high and unstable friction coefficient under dry condition sliding8). This is because when Ti materials wear in contact with metal materials, seizure phenomena easily occur due to their reactive property and low thermal conductivity. In the previous studies, the surface modification techniques such as strengthening method of the most-top surface and surface hard coating process are developed to improve the tribological properties of Ti materials9,10). For example, the application of titanium carbide (TiC) hard coating film on Ti material is available to obtain a low and stable friction coefficient because TiC has a good coherence with Ti matrix11). That is, in case of the surface modification process by thin coating films, the friction behavior and wear resistance are strongly dependent on both mechanical properties of the coating films and bonding coherence between the coating film and the matrix.
In this study, instead of the previous surface modification method, the matrix strengthening process by using nitrogen solid solution was investigated to improve the tribological property of PM pure Ti based materials. The previous study clarified the extreme improvement of tensile yielding stress of pure Ti material by using nitrogen solid solution via solid-gas reaction process5). In this process, a large input energy is necessary to dissolve nitrogen atoms into the matrix of pure Ti powders by heating them at high temperature for a long time in nitrogen gas atmosphere, and results in an increase in the materials cost. Therefore, the elemental mixture of pure Ti powder and titanium nitride (TiN) particles is used as starting materials in this study, and nitrogen atoms originated from TiN are dissolved into α-Ti matrix of the Ti sintered material after TiN decomposition in sintering. Nitrogen solution strengthening effect on the mechanical and tribological properties of PM pure Ti based materials is investigated in detail.
Pure Ti powder, having a mean particle size of 21.9 μm, produced by hydride dehydride (HDH) process12) (TC-450: Toho Technical Service Co.) and TiN particles with a mean particle size of 3.1 μm (Mitsuwa Chemicals Co.) were employed as raw materials. A naphthene oil (Nippon Oil & Energy Co.) of 0.004 g was added to Ti powder of 200 g to uniformly coat TiN fine particles on Ti powder surface by using a ball milling equipment (AV-2, Asahi). A very little additive oil can be thermally decomposed during sintering, and then the effect of its element on mechanical properties of Ti sintered materials is possibly negligible. The above pure Ti powder was mixed with TiN particles for 3.6 ks by using a rocking mill equipment (RM-05, SEIWA Co.), where TiN content ratio to the mixture powders was 0~4 mass%. The elemental mixture of Ti-TiN powders were consolidated to the Ti billets with 42 mm diameter by spark plasma sintering (SPS, SPS-1030s, SPS Syntex Co.) at 1000℃ for 10.8 ks under 30 MPa applied pressure in vacuum (~6 Pa). Heat treatment (HT) in vacuum (~40 Pa) was applied to the billet to obtain the uniform solution of nitrogen atoms, where the temperature of 1000℃ and heating time of 10.8 ks were employed. After pre-heating the Ti billets at 1000℃ for 300 s in argon gas atmosphere (flowing rate; 5 L/min.), immediately hot extrusion process (extrusion ratio; 18.5) was applied to them to fabricate the rods with 10 mm diameter by using 2000 kN hydraulic pressing machine (SHP-200-450, Shibayama Kikai Co). Optical microscope (BX-51P, Olympus), field emission scanning electron microscope (FE-SEM, JSM-6500F, JEOL) and x-ray diffraction (XRD, XRD-6100, SHIMADZU) were used for microstructures and phase analysis of PM Ti sintered and extruded materials. In addition, the element distribution analysis of these specimens was carried out by using electron probe micro analyzer (EPMA, JXA-8530F, JEOL). Oxygen and nitrogen contents of them were measured by using oxygen, nitrogen and hydrogen elemental analyzer (EMGA-830, HORIBA). Micro-hardness test was performed at room temperature by using a microhardness tester (HMV-2T, SHIMADZU), and the average Hv of 20 measurements was used as a representative value of the samples. A tensile test specimen, having a gauge length of 20 mm and diameter of 3 mm, and columnar shape specimen (named as pin specimen) used for the wear test with 3.5 mm diameter and 21 mm length, were machined from the extruded rod with the tensile axis perpendicular to the extrusion direction. Each average value of ultimate tensile strength (UTS), 0.2% yielding strength (0.2% YS), and elongation was measured at room temperature from 3 specimens by using a universal testing machine (AUTOGRAPH AG-X 50 KN, SHIMADZU) at a strain rate of 5 × 10−4 s−1. Pin-on-disk type sliding wear test (FPR-2100, RHESCA Co.) was carried out in atmosphere under dry condition to evaluate the friction coefficient and wear loss, where the counter material (disk specimen) was SKD61 alloy steel (Hv; 650). The applied load of 2.5 N, rotating speed of 200 mm/s, and test time of 3.6 ks were used in this study. After the sliding wear test, the damaged sliding surface of each specimen was investigated by using SEM with energy dispersive X-ray spectrometry (EDS, JED-2300, JEOL) to discuss the effect of nitrogen atoms solution into Ti material on the wear phenomena.
Figure 1 shows SEM observation result of (a) pure Ti powder and (b) the elemental mixture of pure Ti powder and 4 mass% TiN particles (Ti-4% TiN). TiN fine particles uniformly exist on Ti powder surface, and no agglomeration of additive TiN particles was observed. Figure 2 indicates XRD profiles of Ti-4% TiN mixture powder and extruded Ti materials using pure Ti, Ti-2% TiN and Ti-4% TiN powders. No TiN diffraction peak was detected in all extruded specimens because additive TiN particles were completely decomposed during SPS and hot extrusion process. According to the previous study5), (0002) Ti peak shift to lower diffraction angle indicates that nitrogen atoms originated from TiN are interstitially soluted between basal planes of α-Ti (hcp) crystal and results in the lattice expansion in c-axis direction. Figure 3 reveals the dependence of the lattice constant changes15) on the TiN content of PM extruded Ti materials by using Ti peak shift measurements of XRD profiles shown in Fig. 2. With increase in the TiN content, the lattice constant in c-axis obviously increases while that in a-axis is almost constant. This result corresponds well to the behavior in case of the interstitial solution of atoms into hcp crystals16), and then it is concluded that this lattice expansion in c-axis is due to the nitrogen atoms solution into α-Ti. As shown in Fig. 4, there is no significant increase in the oxygen contents of each material, that is, no oxidation occurred in the specimen preparation. On the other hand, the nitrogen contents proportionally increase to the TiN addition. The theoretical values of Ti-2% TiN and Ti-4% TiN mixture powders are 0.57 mass% and 1.14 mass%, respectively. The measurement of these specimens are 0.61 mass% and 1.23 mass%, respectively. A good agreement was found through the comparison of the theoretical and analytical values of nitrogen contents. Therefore, almost all of nitrogen atoms originated from the additive TiN particles exist as interstitial elements in α-Ti crystal, and results in the lattice expansion in c-axis direction.

SEM micrographs of pure Ti powder (a) and Ti-4%TiN powder (b).

XRD profiles of extruded Ti-TiN with various TiN contents and Ti-TiN mixed powder.

Lattice constant of a-axis and c-axis of extruded Ti-0-4%TiN materials as function of TiN content.
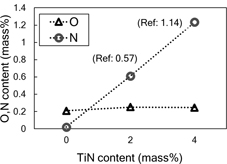
N content and O content of extruded Ti-TiN materials as function of TiN content.
Figure 5 shows EPMA mapping results of (a) SPSed and (b) extruded Ti materials using Ti-4% TiN mixture powder. Both reveals no TiN particle dispersion in the matrix, that is, they are completely dissolved in specimen preparation. In Fig. 5(a) of the SPSed specimen, the primary Ti powder boundaries (PPBs) are obviously observed, and nitrogen atoms locally exist at the boundaries. The extruded material shown in (b) also indicates some agglomeration of nitrogen elements at the boundaries of the Ti primary powder elongated along the extrusion direction. These results suggest that soluted nitrogen atoms are not uniformly distributed in each Ti material after the complete decomposition of TiN additive particles.

EPMA mapping image of Ti-4%TiN specimen after SPS (a) and extruded specimen (b).
Mechanical properties of PM Ti materials with nitrogen solution are shown in Figs. 6 and 7. Nitrogen atoms solution is effective to improve the tensile strength and micro-hardness of Ti materials as same as the evaluation results in the previous study5). The increment of TiN addition, however, caused a remarkable decrease of elongation to failure, that is, from a viewpoint of maintaining enough ductility, a suitable nitrogen content is limited, for example in Japanese Industrial Standards (JIS) H 4600-Class 1~417). In addition, some agglomeration of solute nitrogen elements in the matrix shown in Fig. 5 also cause a significantly hardening behavior, and result in the brittleness of Ti materials.

Dependence of tensile properties on TiN content of extruded Ti-0-4%TiN specimens.

Vickers micro-hardness of extruded Ti-0-4%TiN materials as function of TiN content.
The heat treatment (HT) in vacuum (~40 Pa) was applied to SPSed Ti billets to cancel some agglomeration of nitrogen elements, where HT temperature of 1000℃ and heating time of 10.8 ks were employed in this study. Figure 8 shows EPMA mapping results of (a) SPSed Ti billet after vacuum HT and (b) its extruded sample by using Ti-4% TiN mixture powder. Compared to EPMA images of Ti-4% TiN materials without HT shown in Fig. 5, the SPSed billet via HT and its extruded rod revealed very little agglomeration of nitrogen elements at PPBs, and the uniform distribution of nitrogen elements was observed in both materials after the vacuum HT. Figure 9 indicates the effect of the vacuum HT on the tensile properties of PM Ti materials with nitrogen elements. A remarkable improvement of ductility was performed in both specimens after vacuum HT while no decrease of UTS and 0.2% YS. Additionally, in case of Ti-4% TiN extruded material via vacuum HT, a micro-hardness difference between the maximum and minimum values (ΔHv) was 171 Hv and much smaller, compared to the same Ti material with no HT having ΔHv of 264. This is due to the uniform distribution of nitrogen solution atoms in α-Ti matrix after cancellation of nitrogen agglomeration by HT as mentioned in EMPA results (Fig. 8).

EPMA mapping image of Ti-4%TiN specimen with heat treatment (HT) after SPS (a) and extruded specimen (b).

Stress-Strain curve comparison of extruded Ti-1-2%TiN specimens with HT and without HT.
Figure 10 shows the friction coefficient changes in the sliding wear test using the pin type specimen of (a) PM pure Ti and (b) Ti-4% TiN materials. The latter reveals a low and stable friction coefficient change, compared to pure Ti material in (a). For example, a mean friction coefficient (μ0) and its mean variation (Δμ) of pure Ti were 0.553 and 0.518, respectively. Menwhile, Ti material with nitrogen solution had μ0 of 0.277 and Δμ of 0.115, which were smaller than those of pure Ti. As shown in Fig. 11, both of μ0 and Δμ values gradually decreased with increase in the additive TiN content, that is, nitrogen solution strengthening phenomenon of Ti materials caused the improvement of their tribological properties and a stable sliding wear behavior under dry condition. Figure 12 shows surface morphology observation results of wear damages of pin type specimens. A lot of wear debris were observed on the surface of Ti-0% TiN (a) and its weight loss was 5.4 mg, meanwhile Ti-4% TiN specimen (b) revealed a few debris and a small wear loss of 0.3 mg. It also indicated that the former showed many scratches during sliding while the latter sample (b) had a very slight sliding damages. According to EDS analysis results in Fig. 12, both pin type specimens showed a small amount of wear debris from the SKD61 disk counter material. In general, the matrix hardening is effective to prevent abrasive wear phenomena18), and then it is concluded that a remarkable decrease of wear loss of Ti-4% TiN specimen is due to the matrix hardness improvement by nitrogen atoms solid solution as shown in Fig. 7. Figure 13 reveals SEM-EDS analysis results of the surface damages and tracks of SKD61 disk specimens in using Ti-0% TiN (a) and Ti-4% TiN (b) pin type specimens. Many Ti debris were stuck throughout the disk surface in use of Ti-0% TiN material (a), while such Ti debris were very limited by using Ti-4% TiN specimen (b). For example, the area ratio of the Ti seizure on SKD61 disk surface in using was Ti-0% TiN (a) and Ti-4% TiN (b) pin type specimens was 34.0% and 10.6%, respectively. It is well known adhesive wear phenomena were caused by severe shear failure at the material surface18). As mentioned above, nitrogen solid solution strengthening caused a significant improvement of the matrix hardness, and resulted in control of adhesive wear behaviors at the contacting surfaces between Ti pin and SKD61 disk specimens. According to these analysis results, PM Ti material with nitrogen solution possibly prevents both abrasive and adhesive wear damages by the hardening, and showed a low and stable friction coefficient.

Changes of the friction coefficient of extruded Ti materials strengthened by nitrogen solid-solution of 0 mass% (a) and 4 mass% (b).

Changes of the friction coefficient of extruded Ti materials strengthened by nitrogen solid-solution as function of TiN content.

Surface morphology observations on wear surfaces of Ti-0%TiN (a) and Ti-4%TiN (b).

Surface morphology observations on wear tracks of SKD61 disks using for Ti-0%TiN (a) and using for Ti-4%TiN (b).
To improve the mechanical and tribological properties of Ti materials with no rare metals, nitrogen solid solution strengthening was applied in the fabrication of pure Ti based materials by powder metallurgy route. When the SPS and hot extrusion processes were employed to consolidate the elemental mixture of Ti-TiN powders, the additive TiN particles were completely decomposed and nitrogen atoms were interstitially soluted into α-Ti crystal. The mechanical properties such as 0.2%YS, UTS and micro-hardness significantly increased with increase in the nitrogen content. The agglomerated nitrogen elements caused an elongation decrease of Ti materials, meanwhile the heat treatment application to the sintered Ti billet was effective to improve their elongation by reduction of such concentrated nitrogen elements. The matrix hardness improvement also successfully inhibited both abrasive and adhesive wear phenomena, and resulted in the stably low friction coefficient and reduction of wear loss after sliding test.
This study was financially supported by A-STEP program (AS2715084T), Research and Development Programs Focused on Technology Transfer, Japan Science & Technology Agency (JST) and the research funds by The Light Metal Educational Foundation, Inc.