2014 Volume 232 Issue 4 Pages 255-261
2014 Volume 232 Issue 4 Pages 255-261
Gastric acid measurement is useful in assessing the effectiveness of antisecretory drugs, however, the conventional tests involve invasive nasogastric intubation. Orally administered 13C-labeled calcium carbonate (Ca13CO3) reacts with gastric acid to produce 13C-labeled carbon dioxide (13CO2), which is then excreted in the breath. The objective of this study was to evaluate the suitability of Ca13CO3 breath test for estimating gastric acid secretion in human noninvasively. First, the Ca13CO3 breath test and the measurement of pooled gastric acid under a fasting condition were performed in 6 healthy volunteers to evaluate the correlation between the two parameters. Next, endoscopic gastric acid collection and the Ca13CO3 breath test were performed on different days after pentagastrin injection in 20 subjects to evaluate the correlation between the tests and the reproducibility. Finally, the same studies were repeated in 4 subjects before and after 1-week rabeprazole, a proton pump inhibitor, administration. The maximum 13CO2 concentration (Cmax) correlated very well with the amount of pooled gastric acid (r = 0.95), suggesting that Ca13CO3 breath test values well reflected the fasting intragastric acidity. The 13CO2 concentration after pentagastrin injection correlated well with pentagastrin-stimulated maximal acid output (r = 0.79 at 20 min). The reproducibility of the Ca13CO3 breath test under pentagastrin-stimulation was good (coefficient of variation = 0.11). Rabeprazole administration markedly reduced the values of the Ca13CO3 breath test, suggesting that it can sensitively assess the efficacy of rabeprazole. The Ca13CO3 breath test can potentially be a useful method for non-invasive estimation for gastric acid secretion in human.
Gastro-esophageal reflux disease (GERD) is a common disease in Western countries (Locke et al. 1997; Stanghellini et al. 1999; Diaz-Rubio et al. 2004), and the number of patients with GERD is increasing in Japan as well (Inamori et al. 2003; Fujimoto et al. 2003; Fujiwara et al. 2005). Gastric acid has an important role as the dominant injurious factor in the pathogenesis of GERD. The primary therapeutic approach is suppression of gastric acid secretion, and stronger and prompter gastric acid suppression is required (Bell et al. 1992). Proton pump inhibitors (PPIs) are commonly used as the first-line treatment because of the high effectiveness and prolonged duration of suppression of gastric acid secretion. Thus, the therapeutic effects of PPIs on GERD are associated with the gastric acidity.
Gastric acidity is different in various disease states and their measurement may be useful in the diagnosis and treatment of these diseases. High levels of acidity are found in duodenal ulcers and patients with Zollinger-Ellison syndrome, whereas low or absent levels of acidity are found in pernicious anemia, atrophic gastritis and gastric carcinoma (McColl et al. 1997). Gastric acid measurement may also be useful in assessing the effectiveness of new antisecretory drugs.
The conventional tests of measuring gastric acid secretion involve nasogastric intubation into the stomach and continuous aspiration of gastric fluid for a few hours (Kay 1953; Johnston and Jepson 1967). The procedure of these invasive tests causes significant distress to the patients and is time consuming. To estimate gastric acid secretion easily and rapidly, we previously devised an endoscopic method of gastric acid secretory testing (endoscopic gastrin test: EGT), which is a simple modification of the conventional gastrin-stimulated maximal acid output test (Iijima et al. 1998). However, the endoscopic procedure still causes some discomfort in the subjects, hence repeating EGT is not always easy. Therefore, the development of a non-invasive method would be desirable.
13C is a non-radioactive stable isotope of carbon and is widely used in clinical settings, such as in the 13C-urea breath test for the diagnosis of Helicobacter pylori infection (Graham et al. 1987). 13C can also be incorporated into calcium carbonate. When 13C-labeled calcium carbonate (Ca13CO3) is orally administered, it then reacts with hydrochloric acid in the stomach producing 13C-labeled carbon dioxide (13CO2).
 |
The 13CO2 is absorbed rapidly by the gastric wall and delivered through the bloodstream to the lungs, and finally excreted in the breath. Thus, the amount of exhaled 13CO2 is expected to correlate with the amount of pooled gastric acid. Then, by measuring the ratio of 13CO2 to 12CO2 in the breath sample via an infrared spectrophotometer, the amount of gastric acid can be estimated.
The objective of this study was to evaluate the suitability of the Ca13CO3 breath test for measuring gastric acid secretion in human. First, we evaluated correlations between the breath response and gastric secretory parameters under fasting and pentagastrin stimulated conditions. In addition, we also evaluated the reproducibility of the breath test results under the same conditions. Then, we evaluated weather the Ca13CO3 breath test results reflected the change in gastric acid secretion induced by PPIs administration.
First, to evaluate the correlation between the results of the Ca13CO3 breath test and the total amount of pooled gastric acid under a fasting condition, 6 healthy volunteers (mean age 23.0 ± 1.3 years) underwent endoscopic examination after overnight fast, and the pooled gastric acid was collected. Two hours after endoscopy, the Ca13CO3 breath test was performed under a fasting condition. Then, the series of studies were repeated six times on different days with different doses of Ca13CO3 to determine the appropriate dose of Ca13CO3.
Next, to evaluate the correlation between Ca13CO3 breath test values and the amount of modified gastric acid output (EGT value) under pentagastrin-stimulation, 20 participants (mean age 39.8 ± 18.0 years) were enrolled. Of them, 15 were healthy volunteers, and the other 5 were diagnosed with atrophic gastritis. The subjects with atrophic gastritis did not receive acid suppressive drugs. They underwent EGT as mentioned below, in which gastric fluid was collected endoscopically after pentagastrin injection. On a different day, the Ca13CO3 breath test was performed 20 min after intramuscular injection of pentagastrin. The reproducibility of the Ca13CO3 breath test was estimated by repeating the same test on different days.
Finally, to evaluate whether the Ca13CO3 breath test results reflected the change in gastric acid secretion by PPIs, 4 healthy volunteers were given oral rabeprazole at a dose of 10 mg twice daily for a week. Then, endoscopic gastric acid collection (EGT) and Ca13CO3 breath test under pentagastrin stimulation were repeated before and after rabeprazole administration.
The study was performed in accordance with the Declaration of Helsinki. The study protocol was approved by the ethics committee of the Tohoku University Graduate School (2010-392). Subjects provided written informed consent before participating.
Measurement of the amount of pooled gastric fluidAfter an overnight fast, pooled gastric fluid was obtained by aspiration through an endoscope. The H+ concentration was determined by titration. The total amount of pooled gastric acid was calculated by multiplying the volume with the concentration, and was expressed as H+ mEq.
Calcium carbonate (Ca13CO3) breath testThe Ca13CO3 breath test was performed six times with various doses of Ca13CO3 on different days. Subjects received a single oral dose of 20, 50, 100, 200, 500, and 1000 mg of Ca13CO3 suspended in 50 ml of carboxymethylcellulose sodium (CMC-Na) on each occasion. The participants were in an upright sitting position during the breath test. Breath samples were collected in breath collection bags before and 5, 10, 15, 20, 25, 30, 40, 50, 60, 80, 100, and 120 min after dosing. The 13CO2 concentration in each breath sample was measured using an infrared spectral analyzer (POCone; Otsuka Electronics Co., Ltd., Hirakata, Japan).
Endoscopic Gastrin Test (EGT)The details of the EGT have been reported previously (Iijima et al. 1998). Briefly, after an overnight fast, subjects were injected intramuscularly with pentagastrin at a dose of 6 μg/kg (pentagastrin; Sigma, St. Louis, MO, USA). 15 min after the injection, an endoscope was inserted into the stomach, and pooled gastric fluid was aspirated and discarded. Gastric fluid secreted between 20 and 30 min after the pentagastrin injection was then aspirated and collected under direct visualization during routine endoscopic examination of the stomach and duodenal bulb. The volume of the sample collected over the 10-min period was recorded, and the H+ concentration was determined by titration. The acid output in the 10-min period was calculated by multiplying the volume by the H+ concentration, and the EGT value was expressed as H+ mEq/10 min.
Calcium carbonate (Ca13CO3) breath test under pentagastrin-stimulated gastric acid secretionAfter an overnight fast, subjects were injected intramuscularly with pentagastrin at a dose of 6 μg/kg. A single oral dose of 500 mg of Ca13CO3 suspended in 0.5% CMC-Na was administered 20 min after the injection. A baseline breath sample was collected before injection. Breath samples were collected 2, 4, 6, 8, 10, 12, 14, 16, 18, 20, 25, 30, 40, 50, 60, 80, 100, and 120 min after dosing. The participants were in an upright sitting position during the breath test. The 13CO2 concentration in each breath sample was measured using an infrared spectral analyzer.
Analysis of 13CO2 in breath samplesThe 13CO2 concentration in each breath sample was measured using an infrared spectral analyzer. The 13CO2/12CO2 ratio was expressed as the δ13C value (permil, ‰) relative to the Pee Dee Belemnite Limestone standard, and the Δ13C value (‰) was calculated using the following equations:
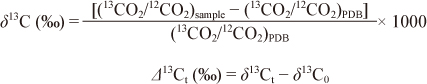 |
Where Δ13Ct is the change in the δ13C value measured at the time t (Δ13Ct) from the baseline (Δ13C0) following the administration of 13C-product.
Statistical analysesThe data are expressed as mean values ± standard deviation (s.d.). The breath test results were defined as the maximum 13CO2 concentration during the 120 min sampling period (Cmax) or a value at a specific time point. A liner regression analysis was carried out to evaluate proportionality between the dose levels of Ca13CO3 and the breath-Cmax. Correlations between the Ca13CO3 breath test values and the EGT values were assessed using linear regression analysis. The reproducibility of the tests was assessed by the coefficient of variation (CV).
The mean 13CO2 concentration (Δ13CO2) in the expired breath versus the time curves after various doses Ca13CO3 are shown in Fig. 1A. The magnitude of the response was related to the Ca13CO3 dose; the Δ13CO2 increased with increases in the Ca13CO3 dose. When 20, 50, 100, 200, or 500 mg of Ca13CO3 was administered, the Δ13CO2 rose sharply within 10 minutes after its oral administration and then decreased gradually. When 1,000 mg of Ca13CO3 was administered, the Δ13CO2 rose sharply within 10 minutes, reached a plateau until 40 minutes and then increased gradually. The excessive amount of Ca13CO3 remaining in the stomach that did not react with gastric acid could be responsible for the delayed breath response to the dose of 1,000 mg of Ca13CO3. The correlation between the Cmax and the Ca13CO3 dose is shown in Fig. 1B and C. Cmax increased linearly with increasing doses of Ca13CO3 up to 200 mg (Fig. 1B), and reached a plateau when 500 and 1,000 mg of Ca13CO3 were administered (Fig. 1C). The Cmax correlated very well with Ca13CO3 doses up to 200 mg (r = 0.87). Hence, 200 mg was considered an appropriate dose of Ca13CO3 in this experimental condition. There was a very good correlation between the Cmax and the total amount of pooled gastric acid, when 200 mg of Ca13CO3 were administered (r = 0.947). This correlation became even slightly higher when the 13CO2 values were modified by height and body weight, as shown in the appendix (r = 0.951, Fig. 1D). Therefore, the following results are presented using 13CO2 values modified by height and body weight, and are expressed as %dose/hr. It was clearly demonstrated that the Ca13CO3 breath test values well reflected the fasting intragastric acidity.
However, the reproducibility of the total amount of pooled gastric acid under a fasting state turned out to be poor in that the CV was 0.74 (data not shown). Because of the poor reproducibility, we could not expect satisfactory reproducibility for the Ca13CO3 breath test under a fasting state, as well. Hence, we employed pentagastrin as a gastric stimulant to make the Ca13CO3 breath test reproducible since high reproducibility of the maximal acid output after pentagastrin injection was reported (Card and Marks 1960). Then, we performed the Ca13CO3 breath test under pentagastirin-stimulated gastric acid secretion. In our previous study (Iijima et al. 1998), the mean EGT value representing the amount of gastric acid secreted between 20 and 30 min after the pentagastrin injection was 3.6 mEq. On the other hand, the mean amount of pooled gastric acid in this study was 1.4 mEq. Hence, we considered that an approximately 2.5 fold higher dose of Ca13CO3 is required under pentagastirin stimulation. Therefore, we used 500 mg of Ca13CO3, which was expected to maintain a linear correlation between the Ca13CO3 dose and the Δ13CO2 of the breath test under pentagastrin stimulation.
The mean Δ13CO2 versus the time curves after oral administration of 500 mg of Ca13CO3 after pentagastrin injection is shown in Fig. 2. The Δ13CO2 under pentagastrin-stimulation rose sharply around 10 min. after oral administration of Ca13CO3 and then decreased gradually. In 4 participants who underwent the Ca13CO3 breath tests with and without pentagastrin, the Δ13CO2 under pentagastrin-stimulation was consistently higher than that without pentagastrin-stimulation at any measuring time point, and the Cmax under pentagastrin-stimulation was 2.7 times higher than that without pentagastrin-stimulation (data was not shown). The Cmax thus acquired in the stimulated Ca13CO3 breath tests well correlated with the EGT value (Fig. 3A, r = 0.72). The high correlation between the stimulated Ca13CO3 breath test and the EGT value was evident even when a one point sampling value of the breath test was applied in place of the Cmax. In particular, the Δ13CO2 at 20 min after dosing showed the highest correlation with the EGT value (r = 0.79), suggesting that the Ca13CO3 breath test can be performed as a simple and reliable procedure with one-point sampling for the measurement of gastric acid secretion in human (Fig. 3B). In addition, the stimulated Ca13CO3 breath tests were repeated in 4 participants to evaluate the reproducibility. Consequently, the reproducibility of the breath test was good with a CV of 0.11, as shown in Fig. 4.
Fig. 5 shows the Δ13CO2 versus the time curves of the stimulated Ca13CO3 breath test before and after rabeprazole administration at a dose of 10 mg twice daily for a week. Compared with Δ13CO2 without rabeprazole, Δ13CO2 after rabeprazole administration dramatically decreased and nearly flattened at each measuring time point, and the Cmax decreased from 61.6 ± 7.5 %dose/hr to 8.1 ± 5.8 %dose/hr. Meanwhile, rabeprazole administration reduced the mean EGT value from 3.7 ± 1.5 mEq/10 min to 0.17 ± 0.25 mEq/10 min in the same participants.

Ca13CO3 breath test under unstimulated conditions.
A): The mean 13CO2 concentrations (Δ 13CO2) in expired air versus time curves after oral administration of Ca13CO3 at 20, 50, 100, 200, 500, and 1000 mg. Data are shown as mean ± s.d. (n = 6) for each dose. B), C): The correlation between the maximum 13CO2 concentration in breath (Cmax) and the Ca13CO3 dose. Correlations between the Cmax and Ca13CO3 dose in the range from 20 to 200 mg (B) and from 20 to 1,000 mg (C). There was a high correlation between the Cmax and Ca13CO3 dose up to 200 mg (r = 0.87). Each symbol represents mean of 6 subjects. D): The correlation between the Cmax and the total amount of pooled gastric acid at 200 mg of Ca13CO3. There was a very high correlation between the two parameters (r = 0.95). Each symbol represents individual data of 6 subjects.

The mean 13CO2 concentrations (Δ 13CO2) in expired air versus time curves of Ca13CO3 breath test under pentagastrin stimulation.
Data are shown as mean ± s.d. n = 20.

The correlation between the Ca13CO3 breath test under pentagastrin stimulation and the EGT value.
A: The correlation between the Cmax of the breath test results and EGT value (r = 0.71). B: The correlation between Δ 13CO2 at 20 min after dosing and EGT value (0.79). n = 20.

Reproducibility of Ca13CO3 breath test under pentagastrin stimulation.
The duplicate results of Ca13CO3 breath test (Δ 13CO2 versus time curves) are shown for each subject (n = 4). The coefficient of variation of Δ 13CO2 at 20 min was 0.11 (0.04-0.20).

The effect of rabeprazole administration on the Ca13CO3 breath test under pentaagastrin stimulation and the EGT values.
A: The mean Δ 13CO2 versus time curves of Ca13CO3 breath test under pentagastrin stimulation; B: the mean EGT value prior to and after 1-week rabeprazole administration. Data are shown as mean ± s.d. (n = 4).
This study shows the usefulness of the Ca13CO3 breath test for estimating the total amount of gastric acid secretion. The Ca13CO3 breath test well reflected the amount of fasting pooled intragastric acid. The Ca13CO3 breath test under pentagastrin stimulation had high reproducibility and correlated well with the modified maximal acid output (EGT). Moreover, the stimulated Ca13CO3 breath test could sensitively detect changes in the intragastric acidity induced by PPIs administration.
There was a very good correlation between the Cmax and the total amount of fasting pooled gastric acid (r = 0.95), suggesting that the Ca13CO3 breath test results well reflected the fasting intragastric acidity at any given time. The chemical property of Ca13CO3, which is poorly water-soluble, but reacts immediately with hydrochloric acid in the stomach producing 13CO2, which is excreted in the breath at a high rate, would be responsible for the high correlation between the breath test and the amount of pooled intragastric acid. These results are consistent with those of a study in rats reported by Inada et al. (2012), in which the Ca13CO3 breath test with or without proton pump inhibitors or pentagastrin showed a high correlation between the breath-Cmax and the total amount of gastric acid (r = 0.994). Thus, the Ca13CO3 breath test can be a potentially promising candidate as a noninvasive test for measuring gastric acid secretion in human.
High reproducibility is essential to establishing a new biological test, and is especially true for gastric secretory testing because it is well-known that the gastric secretory state shows wide-variation in daily life. Nonetheless, the reproducibility of the total amount of pooled gastric acid without any stimulants was poor in the present study (CV = 0.74), consistent with a previous study showing poor reproducibility of a gastric fluid analysis in the basal state (White and Juniper 1973). Therefore, the Ca13CO3 breath test value under fasting could fluctuate in repeating tests, albeit each test value could reflect the fasting intragastric acidity at a given time. Actually, in 4 subjects who repeated the Ca13CO3 breath test three times without gastric stimulants, the test results considerably varied with the CV of 0.25 (data not shown). To enhance the reproducibility, usage of a gastric acid stimulant such as test meals or drugs have been reported (Kay 1953; Ward et al. 1963; Lawrie and Forrest 1965). In particular, pentagastrin has frequently been used as a gastric acid stimulant in gastric secretory testing. Gastric acid measurements using pentagastrin have been found to be reproducible in several studies including our case of EGT (Johnston and Jepson 1967; Iijima et al. 1998). Therefore, in the present Ca13CO3 breath test, we applied pentagastirin injection as a gastric acid stimulant to enhance its reproducibility.
With the aid of pentagastrin stimulation, the reproducibility of the Ca13CO3 breath test became much improved, with a CV as low as 0.11, compared to that without any stimulants (CV = 0.25). At the same time, the stimulated Ca13CO3 breath test (Cmax or the Δ13CO2 at 20 min) retained a good correlation with the EGT values, which is a simplified index of the conventional maximal acid output. In addition, using this stimulated Ca13CO3 breath test, a representative one-point sampling value in place of the Cmax obtained from multiple samplings for 2 hours turned out to be well correlated with the EGT values. Especially, the Δ13CO2 at 20 min after Ca13CO3 administration showed the best correlation with the EGT value (r = 0.79), suggesting that the Ca13CO3 breath test can be performed as a simple and reliable procedure with one-point sampling for the estimation of gastric acid secretion in human.
Further, we explored the effects of PPI administration on the results of the stimulated Ca13CO3 breath test to determine its clinical suitability and found that 1-week PPI administration caused a prominent reduction in the breath response (Δ13CO2), along with a profound reduction of the actual gastric acid secretion estimated by the EGT, suggesting that the Ca13CO3 breath test could be useful for evaluating the antisecretory effect of PPI. In GERD patients, at present, 24-hour intragastric pH monitoring is occasionally employed to determine whether the PPIs effectively inhibit gastric acid secretion. In that procedure, a pH-measuring electrode has to be inserted in the stomach through the nasogastric route, which is uncomfortable and restricts the physical activity of the patient. In contrast, the Ca13CO3 breath test can provide information regarding the efficacy of PPI in a noninvasive and simple manner.
Some attempts to apply breath tests for measuring gastric acid secretion have been previously reported, but the tests have not been employed in clinical practice. Sack and Stephenson (Sack and Stephensen 1985; Stephensen et al. 1987) performed a hydrogen breath test for estimating gastric acid by oral magnesium administration in 1985. In their test, administered magnesium reacts with gastric acid to produce hydrogen gas, the amount of which reflects the gastric acid. The hydrogen concentration was measured by gas chromatography of the breath samples. A relatively good correlation coefficient of 0.70 was found between the conventional stimulated acid output values and the excess amount of hydrogen exhaled during the test. However, it could be difficult for this test to detect gastric acid secretion because substantial amounts of hydrogen gas exist in the exhaled air in some pathological conditions, especially in patients with intestinal bacterial overgrowth (Armbrecht et al. 1985). In addition, magnesium in its metallic form has not yet been approved for clinical use in many countries including Japan, whereas the clinical use of ordinary Ca12CO3 has been widespread as an antacid. Furthermore, exhaled hydrogen monitoring by gas chromatography is less popular than exhaled 13CO2 measurement by spectral analyzer.
Clough and Axon (2009), using the same compositional Ca13CO3 as in our study, first reported the potential usefulness of a calcium carbonate breath test as a noninvasive method of quantifying the maximal acid secretion. In their test, excessive amounts of Ca13CO3 (e.g. 500 mg every 5 min or 750 mg every 10 min up to several hours) were administered to induce maximal gastric acid secretion by the feedback stimulation resulting from the neutralization of gastric acid by the antacid action of Ca13CO3. The procedure seemed to successfully induce near maximal gastric acid secretion without using a pharmacological gastric stimulant such as pentagastrin. However, the test was performed in a single subject and was not compared with a conventional test, and the procedure was somewhat bothersome and time consuming. Moreover, the extensive use of costly Ca13CO3 lacks versatility for daily clinical practice. Compared to this test, the present Ca13CO3 breath test could simply and reliably assess the gastric acid output in a shorter time, although our test requires pentagastrin injection.
There may be a limitation of this study. Although the overall correlation between the stimulated Ca13CO3 breath test and EGT value was good, the regression line did not pass the origin of the coordinates and was relatively gentle (Fig. 3A and B). This suggests that the breath test is not a quantitative test of acid secretion over the range studied although it is useful as a qualitative test to pick up severe hypochlorhydria. Further studies are required to investigate appropriate test conditions to improve the ability of quantitative measurement of wide range of gastric acid secretion level. In addition, gastric motility such as gastric emptying may affect the breath test results by loss of acid secretion through the pylorus prior to the Ca13CO3 administration. However, once Ca13CO3 is administered into the stomach, gastric emptying should rarely affect the rate of the reaction occurring in the stomach since chemical reaction between Ca13CO3 and hydrochloric acid proceeds promptly.
In conclusion, the Ca13CO3 breath test could be a simple, noninvasive method for estimating gastric acid secretion in human, and the test may be suitable for the clinical management of acid-related diseases. The development of ideal test meals as an alternative to pentagastrin injection for achieving high reproducibility of the Ca13CO3 breath test will broadly enhance the clinical suitability of the test.
The authors declare no conflict of interest.
The 13C-excretion rate (%Dose/hr) against the dose is obtained by the following equation (Ghoos et al. 1993).
 |
∆13C: Change in δ 13C value (‰)
V CO2: Velocity of CO2 formation (mmol / hr)
RPDB: 13C abundance ratio in PDB standard gas = 0.01123726
MW: Molecular weight of reagent (g/mol)
A: Dose of reagent (mg)
#: Number of labeled carbons in the molecule of reagent
APE: 13C-enrichment in reagent (atom%)