Abstract
Carbohydrate-restricted diets are prevalent not only in obese people but also in the general population to maintain appropriate body weight. Here, we report that extreme carbohydrate restriction for one day affects the subsequent blood glucose levels in healthy adults. Ten subjects (median age 30.5 years, BMI 21.1 kg/m2, and HbA1c 5.5%), wearing with a continuous glucose monitoring device, were given isoenergetic test meals for 4 consecutive days. On day 1, day 2 (D2), and day 4 (D4), they consumed normal-carbohydrate (63-66% carbohydrate) diet, while on day 3, they took low-carbohydrate/high-fat (5% carbohydrate) diet. The daily energy intake was 2,200 kcal for males and 1,700 kcal for females. On D2 and D4, we calculated the mean 24-hr blood glucose level (MEAN/24h) and its standard deviation (SD/24h), the area under the curve (AUC) for glucose over 140 mg/dL within 4 hours after each meal (AUC/4h/140), the mean amplitude of the glycemic excursions (MAGE), the incremental AUC of 24-hr blood glucose level above the mean plus one standard deviation (iAUC/MEAN+SD). Indexes for glucose fluctuation on D4 were significantly greater than those on D2 (SD/24h; p = 0.009, MAGE; p = 0.013, AUC/4h/140 after breakfast and dinner; p = 0.006 and 0.005, and iAUC/MEAN+SD; p = 0.007). The value of MEAN/24h and AUC/4h/140 after lunch on D4 were greater than those on D2, but those differences were not statistically significant. In conclusion, consumption of low-carbohydrate/high-fat diet appears to cause higher postprandial blood glucose on subsequent normal-carbohydrate diet particularly after breakfast and dinner in healthy adults.
Introduction
Recently, patients with diabetes and/or obesity have begun to take extremely-low-carbohydrate diets, and some studies have suggested that such diets are effective for weight loss and improving insulin resistance (Shai et al. 2008). In addition, carbohydrate-restricted diets are prevalent even among the general population to maintain appropriate body weight.
For healthy subjects, there is a concern that carbohydrate restriction may induce a deterioration in glucose tolerance. From a classical viewpoint, some previous researchers have indicated that carbohydrate restriction decreases glucose tolerance and increases the frequency of false-positive impaired glucose tolerance results in patients undergoing a glucose tolerance test (Himsworth 1940; Wang et al. 1999). Nowadays, despite the popularity of extreme carbohydrate-restricted diets, the actual clinical influences of these diets on glucose fluctuation and their possible adverse effects remain unknown. Specifically, how changes in dietary carbohydrate contents might affect subsequent blood glucose profiles, how long the influence of differences in dietary carbohydrate contents actually persists, and whether these glucose fluctuations have undesirable effects remain uncertain.
In this study, we investigated, with continuous glucose monitoring (CGM), whether extreme carbohydrate restriction for one day in actual life could affect the subsequent blood glucose levels in healthy subjects.
Materials and Methods
Participants
We enrolled healthy volunteer subjects (2 males and 8 females) who were over 20 years and under 65 years of age and had normal hemoglobin A1c (HbA1c) levels (less than 6.5%). Subjects with a history of diabetes and/or any other diseases that might influence glucose fluctuations (e.g., hormonal disease, post-gastrectomy status, and cancer) were excluded. All the subjects were provided written informed consent prior to enrollment in the study. The study protocol was approved by the ethics committee of the National Center of Global Health and Medicine Center Hospital (NCGM-G-001678-00) and was implemented in accordance with the provisions of the Declaration of Helsinki.
Study design
The subjects were provided two types of test meal and observed the glucose fluctuations with continuous glucose monitoring device (CGM; Medtronic iPro®2 Northridge, CA, USA). CGM is often used in both clinical practice and research. Its reliability in normo-glycemic individuals has also been shown (Akintola et al. 2015). Over the course of 4 consecutive days, the subjects consumed a normal-carbohydrate (NC) diet on days 1 (D1), 2 (D2) and 4 (D4) and a low-carbohydrate/high-fat (LC/HF) diet on day 3 (D3). The glycemic excursions on the day following intake of a NC diet (D2) were compared with those on the day following intake of the LC/HF diet (D4). Representative CGM data from one subject are illustrated in Fig. 1. Male and female subjects had an energy intake of approximately 2,200 and 1,700 kcal/day on days 1 to 4. We decided total energy intake referring to the estimated energy requirement meal intake standards of Ministry of Health, Labour and Welfare in Japan (Ministry of Health, Labour and Welfare 2015). On the days of the NC diet intake (D1, D2 and D4), the mean intakes of carbohydrate, fat and protein intakes per meal in the males and females were 120.6 g (66%) and 88.8 g (63%), 18.5 g (22%) and 15.0 g (23%), and 21.7 g (12%) and 19.7 g (14%), respectively. NC diets were prepared by a combination of rice, boiled vegetable, chicken, fish, hamburg, stew, pudding, fruit juice, curry, cheese, nuts, and tea. On the day of the LC/HF diet intake (D3), the corresponding amounts were 10.0 g (5%) and 7.9 g (5%), 62.8 g (77%) and 46.1 g (73%), and 33.4 g (18%) and 32.0 g (22%), respectively. LC/HF diets were prepared by a combination of tohu, cheese, fish, mayonnaise, nuts, and tea. The subjects were instructed to wear a continuous glucose monitoring device for 4 days, so that the 24-hour glycemic excursions could be observed every 5 minutes (glucose levels were recorded 288 times a day).
The subjects were given the following instructions during the observation period: 1) To eat only the meals provided and to avoid eating between meals, 2) To drink water or sugar free beverages, and 3) To eat meals at the same time of the day as much as possible, and 4) undertake the same level of physical activity while avoiding strenuous exercise.
A day was defined as a 24-hour period starting from 4:00 AM on one day and ending at 4:00 AM on the following day. Fasting blood glucose levels were measured between 4:00 AM and 5:00 AM. The mean 24-hr blood glucose level and its standard deviation (the 24-hr SD), the mean fasting glucose levels (from 4:00AM to 5:00AM), the mean amplitude of the glycemic excursions (MAGE) were calculated on D2 and D4. It is also evaluated that the area under the curve (AUC) for glucose over 140 mg/dL within 4 hours (AUC/4h/140) after each meal, incremental AUC of 24-hr blood glucose level above the mean plus one standard deviation of D2 and D4, area over curve of 24-hr blood glucose level below the mean minus one standard deviation of D2 and D4, on D2 and on D4. These continuous variables were analyzed between D2 and D4.
Statistics
For the statistical analysis, continuous variables were analyzed using the Wilcoxon signed-rank test. The statistical significance level was set at P < 0.05. The statistical analysis was performed using Stata 13 software.
Results
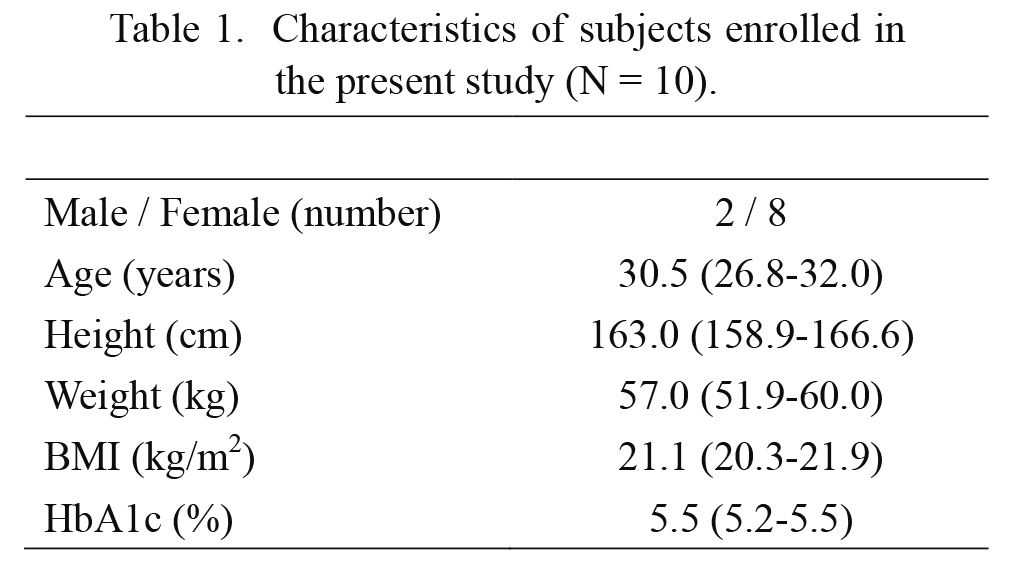
The characteristics of the participants are shown in Table 1. We enrolled 10 healthy subjects (2 males and 8 females). The median age was 30.5 years, the body mass index (BMI) was 21.1, and the HbA1c level was 5.5%. According to CGM data of D2, all of their fasting glucose levels were lower than 110 mg/dL.
The precise blood glucose profiles of the 10 subjects are shown in Table 2. The value of daily mean blood glucose levels on D4 was greater than that on D2 but this difference was not significant. The mean fasting blood glucose levels (4:00 AM to 5:00 AM) on D2 were equivalent to those on D4 (p = 0.139). The SD of the daily mean blood glucose levels and the MAGE were significantly higher on D4 than on D2 (p = 0.009, p = 0.013, respectively). As for the AUC/4h/140, the values obtained after breakfast and dinner were higher on D4 than on D2 (p = 0.006, p = 0.005, respectively), but the values obtained after lunch were not significantly different between D2 and D4 (p = 0.276).
The incremental AUC of 24-hr blood glucose level above the mean blood glucose level plus one SD was significantly greater on D4 than that on D2 (p = 0.007), while the area over curve of 24-hr blood glucose below the mean blood glucose level minus one SD on D4 was comparable to that on D2 (p = 0.226).
Discussion
Previous reports of the effect of dietary carbohydrate restriction on glucose excursion were limited to evaluation by the glucose tolerance test (Himsworth 1940; Wang et al. 1999; Numao et al. 2012). There were unclear points on the influence on glucose metabolism in actual dietary life. The present study by CGM showed that after extreme restriction of carbohydrate, an influence on the blood glucose variability persisted for at least 24 hours in healthy subjects. After dietary carbohydrate restriction for one day, the glucose fluctuation on the following day after the LC/HF diet increased significantly, compared with the fluctuations on days after the ingestion of an NC diet. In particular, the postprandial glucose levels were elevated after breakfast and dinner.
Some researchers have examined glucose loading as a possible cause of the blood glucose fluctuations (Randle et al. 1963; Anderson and Herman 1975; Wang et al. 1999; Numao et al. 2012). Numao et al. (2012) observed that early insulin secretion was lower and post-load blood glucose levels and plasma glucagon-like peptide 1 (GLP-1) levels were higher after the consumption of a low-carbohydrate/high-fat diet for 3 days, compared with those after the consumption of a normal carbohydrate/fat diet for 3 days. Previous studies have indicated that the plasma FFA levels also increase after dietary carbohydrate restriction and that this increase in the FFA levels might be associated with exaggerated post-load blood glucose excursions (Randle et al. 1963; Wang et al. 1999). Anderson and Herman (1975) have suggested that the high fat content associated with low-carbohydrate diets is responsible for the deterioration in the post-load blood glucose levels, rather than the low-carbohydrate content of these diets. Considering these prior studies, both insulin resistance caused by a high FFA plasma level and a decrease in first-phase insulin secretion after a LC/HF diet might have induced the glucose fluctuations observed in this study.
In our results, the AUC/4h/140 after lunch was equivalent on D2 and D4, unlike after breakfast and dinner. According to studies using CGM in patients with type 2 diabetes, exercise significantly decreases the postprandial glucose level, but not the fasting glucose level (MacLeod et al. 2013). Although we did not evaluate the subjects’ activity levels, glucose fluctuations after lunch might be more strongly affected by daytime activity.
Postprandial hyperglycemia has been reported to be a risk factor for cardiovascular events in non-diabetic patients (Tominaga et al. 1999) as well as diabetic patients (Ceriello et al. 2004). Hyperglycemia after glucose loading has been reported to inhibit endothelial flow-mediated dilatation (FMD), which is a measure of the endothelial function that is used to evaluate the cardiovascular risk. Suppression of FMD has been reported by oral glucose loading in subjects with impaired glucose tolerance or diabetes (Kawano et al. 1999) and normal glucose tolerance (Title et al. 2000). In this study, a diet with extreme change in dietary carbohydrate and fat content induced greater blood glucose excursions than a balanced-nutrient diet. These findings suggest that extreme changes in the dietary nutrient balance could induce higher blood glucose fluctuation and have adverse effects in daily life. It also suggests that if an extremely low-carbohydrate diet is stopped abruptly, it could cause a larger degree of postprandial hyperglycemia than an ordinary diet. A gradual re-increase in glucose would probably prevent postprandial hyperglycemia after an extremely low-carbohydrate diet.
This study had several limitations. First, we did not observe the changes in the levels of the insulin, C-peptide, GLP-1, and FFA, and could not assess whether the increase in the postprandial blood glucose levels on the day after carbohydrate restriction was caused by a decrease in insulin secretion or increase in insulin resistance. However, considering the previous studies mentioned above, both elements would affect this result. Second, we did not conduct a glucose tolerance test in the subjects before they were enrolled in this study. Although none of the subjects had a history of diabetes and all of them had HbA1c levels within the normal range (Table 1), we could not distinguish whether the glucose tolerance of the participants was normal or impaired. Third, the observation period was relatively short. An appropriate observation period should include the term during which the influence of the glucose fluctuation recovers after an LC/HF diet. Finally the number of subjects was also small. A larger sample size is needed to compare differences between the sexes. However, we are the first to precisely investigate, by CGM, the around 24-hours effect of a LC/HF diet on the blood glucose profile.
In conclusion, low-carbohydrate/high-fat diets, even for a short duration, can induce increasing blood glucose fluctuations that lasted for at least all of the following day in actual dietary life. Specifically, we found significant increases in the 24-hour SD, MAGE, AUC/4h/140 after breakfast and dinner, and the AUC above the mean blood glucose level plus one SD on the following day after the ingestion of an LC/HF diet, compared with those values on the day after the ingestion of an NC diet.
Further studies on a larger number of subjects and precise glucose and hormonal investigations with varying carbohydrate contents in the test meals are required to confirm the influence of low-carbohydrate/high-fat diets on the blood glucose fluctuation.
Acknowledgments
We are grateful to the physicians of the Department of Diabetes, Endocrinology and Metabolism, nurses on the 11th floor of the East Ward and staff members in the Nutrition Management Department, National Center for Global Health and Medicine Center Hospital, for their sincere cooperation.
KKnamori and NIS equally contributed to this work. KKanamori, RYH, MK, and MN conceptualized the idea for the study; KKanamori, NIS, RYH, MK, and MN designed the study; KKanamori and TN collected the data; KKanamori and NIS performed the statistical analysis; KKanamori, NIS, RYH, MK, and MN wrote the manuscript; CS, SK, HK, and KKawano, joined the discussion. All authors have read and approved the final manuscript.
This work was supported in part by Grants-in-Aid for Research from the National Center for Global Health and Medicine (26A-201).
Conflict of Interest
Koji Kanamori, Noriko Ihana-Sugiyama, Ritsuko Yamamoto-Honda, Tomoka Nakamura, Chie Sobe, Shigemi Kamiya, Miyako Kishimoto, Hiroshi Kajio, and Kimiko Kawano declare that they have no conflicts of interest. Mitsuhiko Noda received grants from Mochida Pharmaceutical Co. LTD., Astra Zeneca K.K., Sanwa Kagaku Kenkyusho Co. Ltd., Takeda Pharmaceutical Co. Ltd., Kowa Pharmaceutical Co. Ltd., Kyowa Hakko Kirin Co. Ltd., Daiichi Sankyo company, and Mitsubishi-Tanabe Pharma Corp. as well as lecture fees from Sanofi K.K., Mitsubishi-Tanabe Pharma Corp., Daiichi Sankyo company, Eli Lilly Japan K.K., MSD K.K., Novo Nordisk Pharma Ltd., Kissei Pharmaceutical Co. Ltd., Ono Pharmaceutical Co. Ltd., Takeda Pharmaceutical Co. Ltd., Astellas Pharma Inc., Kowa Pharmaceutical Co. Ltd., Taisho Toyama Pharmaceutical Co. Ltd., Meiji Seika Pharma Co. Ltd., Kyowa Hakko Kirin Co. Ltd., AbbVie Inc, Sanwa Kagaku Kenkyusho CO. Ltd., AstraZeneca K.K., Shionogi & Co. Ltd., Novartis Pharma K.K., and Johnson & Johnson K.K..
References
-
Akintola,
A.A.,
Noordam,
R.,
Jansen,
S.W.,
de Craen,
A.J.,
Ballieux,
B.E.,
Cobbaert,
C.M.,
Mooijaart,
S.P.,
Pijl,
H.,
Westendorp,
R.G. &
van Heemst,
D.
(2015) Accuracy of Continuous Glucose Monitoring Measurements in Normo-Glycemic Individuals. PLoS One, 10, e0139973.
-
Anderson,
J.W. &
Herman,
R.H.
(1975) Effects of carbohydrate restriction on glucose tolerance of normal men and reactive hypoglycemic patients. Am. J. Clin. Nutr., 28, 748-755.
-
Ceriello,
A.,
Hanefeld,
M.,
Leiter,
L.,
Monnier,
L.,
Moses,
A.,
Owens,
D.,
Tajima,
N. &
Tuomilehto,
J.
(2004) Postprandial glucose regulation and diabetic complications. Arch. Intern. Med., 164, 2090-2095.
-
Himsworth,
H.P.
(1940) Insulin Deficiency and Insulin Inefficiency. Br. Med. J., 1, 719-722.
-
Kawano,
H.,
Motoyama,
T.,
Hirashima,
O.,
Hirai,
N.,
Miyao,
Y.,
Sakamoto,
T.,
Kugiyama,
K.,
Ogawa,
H. &
Yasue,
H.
(1999) Hyperglycemia rapidly suppresses flow-mediated endothelium-dependent vasodilation of brachial artery. J. Am. Coll. Cardiol., 34, 146-154.
-
MacLeod,
S.F.,
Terada,
T.,
Chahal,
B.S. &
Boule,
N.G.
(2013) Exercise lowers postprandial glucose but not fasting glucose in type 2 diabetes: a meta-analysis of studies using continuous glucose monitoring. Diabetes Metab. Res. Rev., 29, 593-603.
-
Ministry of Health, Labour and Welfare
(2015) Overview of Dietary Reference Intakes for Japanese. http://www.mhlw.go.jp/file/06-Seisakujouhou-10900000-Kenkoukyoku/Overview.pdf [Accessed: July 28, 2017]
-
Numao,
S.,
Kawano,
H.,
Endo,
N.,
Yamada,
Y.,
Konishi,
M.,
Takahashi,
M. &
Sakamoto,
S.
(2012) Short-term low carbohydrate/high-fat diet intake increases postprandial plasma glucose and glucagon-like peptide-1 levels during an oral glucose tolerance test in healthy men. Eur. J. Clin. Nutr., 66, 926-931.
-
Randle,
P.J.,
Garland,
P.B.,
Hales,
C.N. &
Newsholme,
E.A.
(1963) The glucose fatty-acid cycle its role in insulin sensitivity and the metabolic disturbances of diabetes mellitus. Lancet, 281, 785-789.
-
Shai,
I.,
Schwarzfuchs,
D.,
Henkin,
Y.,
Shahar,
D.R.,
Witkow,
S.,
Greenberg,
I.,
Golan,
R.,
Fraser,
D.,
Bolotin,
A.,
Vardi,
H.,
Tangi-Rozental,
O.,
Zuk-Ramot,
R.,
Sarusi,
B.,
Brickner,
D.,
Schwartz,
Z., et al.
(2008) Weight loss with a low-carbohydrate, Mediterranean, or low-fat diet. N. Eng. J. Med., 359, 229-241.
-
Title,
L.M.,
Cummings,
P.M.,
Giddens,
K. &
Nassar,
B.A.
(2000) Oral glucose loading acutely attenuates endothelium-dependent vasodilation in healthy adults without diabetes: an effect prevented by vitamins C and E. J. Am. Coll. Cardiol., 36, 2185-2191.
-
Tominaga,
M.,
Eguchi,
H.,
Manaka,
H.,
Igarashi,
K.,
Kato,
T. &
Sekikawa,
A.
(1999) Impaired glucose tolerance is a risk factor for cardiovascular disease, but not impaired fasting glucose. The Funagata Diabetes Study. Diabetes Care, 22, 920-924.
-
Wang,
P.Y.,
Kaneko,
T.,
Wang,
Y.,
Tawata,
M. &
Sato,
A.
(1999) Impairment of glucose tolerance in normal adults following a lowered carbohydrate intake. Tohoku J. Exp. Med., 189, 59-70.