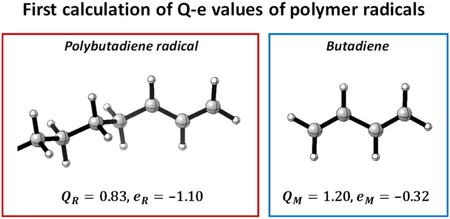
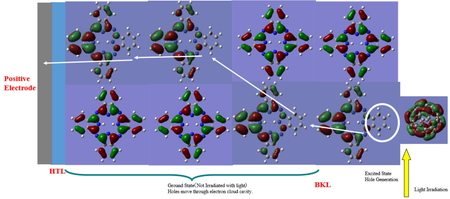
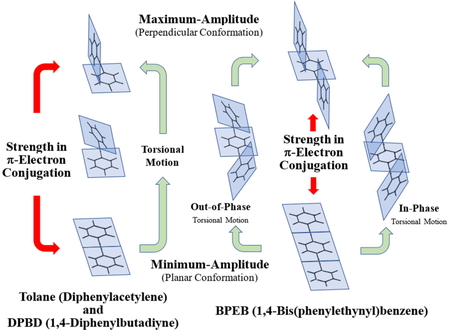
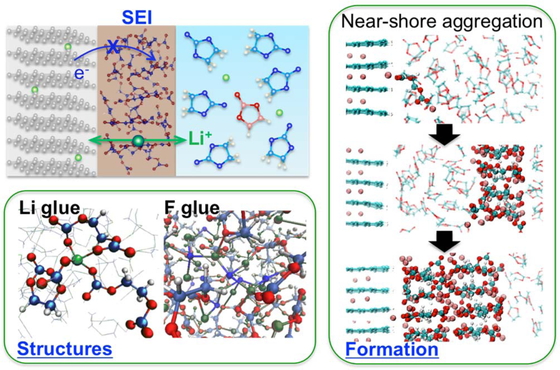
Journal of Computer Chemistry, Japan
Published by
Society of Computer Chemistry, Japan
1,071 registered articles
(updated on July 27, 2024)
(updated on July 27, 2024)
Online ISSN : 1347-3824
Print ISSN : 1347-1767
ISSN-L : 1347-1767
Print ISSN : 1347-1767
ISSN-L : 1347-1767
JOURNAL
PEER REVIEWED
FREE ACCESS
FULL-TEXT HTML
ADVANCE PUBLICATION
J-STAGE Data
J-STAGE Data