2017 Volume 40 Issue 4 Pages 510-515
2017 Volume 40 Issue 4 Pages 510-515
Many zinc (Zn) complexes have been developed as promising oral antidiabetic agents. In vitro assays using adipocytes have demonstrated that the coordination structures of Zn complexes affect the uptake of Zn into cells and have insulinomimetic activities, for which moderate stability of Zn complexes is vital. The complexation of Zn plays a major role improving its bioavailability. However, investigation of the speciation changes of Zn complexes after oral administration is lacking. A dual radiolabeling approach was applied in order to investigate the speciation of bis(5-chloro-7-iodo-8-quinolinolato)zinc complex [Zn(Cq)2], which exhibits the antidiabetic activity in diabetic mice. In the present study, 65Zn- and 131I-labeled [Zn(Cq)2] were synthesized, and their biodistribution were analyzed after an oral administration using both invasive conventional assays and noninvasive gamma-ray emission imaging (GREI), a novel nuclear medicine imaging modality that enables analysis of multiple radionuclides simultaneously. The GREI experiments visualized the behavior of 65Zn and [131I]Cq from the stomach to large intestine and through the small intestine; most of the administered Zn was transported together with clioquinol (5-chloro-7-iodo-8-quinolinol) (Cq). Higher accumulation of 65Zn for [Zn(Cq)2] than ZnCl2 suggests that the Zn associated with Cq was highly absorbed by the intestinal tract. In particular, the molar ratio of administered iodine to Zn decreased during the distribution processes, indicating the dissociation of most [Zn(Cq)2] complexes. In conclusion, the present study successfully evaluated the speciation changes of orally administered [Zn(Cq)2] using the dual radiolabeling method.
In recent years, various types of metal complexes have been developed for the treatment of a variety of diseases such as cancer, infection, and diabetes mellitus (DM).1–6) Zinc (Zn) complexes have attracted much attention in the treatment of type 2 DM as several Zn complexes show potent antidiabetic activity in experimental diabetic animals.6–10) The Zn atom is an essential component of this complex’s antidiabetic activity because its ligands are generally inactive. The complexation of Zn with a ligand potentiates its antidiabetic activity, partially due to the improved gastrointestinal absorption of Zn.8,11–13) However, in vitro assays with adipocytes have demonstrated that Zn complexes with stability constants (log β) values higher than 11.0 exhibited essentially no insulinomimetic activity.14) This suggests that what Zn and its ligands are dissociated from the complex in target tissues has a significant role in the exertion of insulinomimetic activity. Therefore, it is important to investigate whether Zn complex is dissociated in vivo in order to predict the antidiabetic activity.
Fluorescence probes that discriminate between coordination environments and X-ray absorption near edge structure (XANES) imaging can aid in the investigation of the speciation of Zn complexes.15) However, fluorescence probes may perturb metal homeostasis and they are often not detected in areas deep within the body due to the background of biological molecules. XANES imaging may cause chemical disruption, and it has difficulty analyzing the thick samples because of technological limitation. In contrast, a dual radiolabeling approach for tracking metal complexes was recently employed in order to investigate the speciation of copper bis(thiosemicarbazonates) in vitro and in vivo.16) This method enables researchers to analyze the distribution of ligands as well as that of metal, lending insight into whether the metal is coordinated with the ligands in biological samples.
The distribution, accumulation, and metabolism of most Zn complexes in the body are poorly understood. We have developed a novel methodology to analyze the distribution of Zn complexes noninvasively using gamma-ray emission imaging (GREI) to detect the distribution of the radioisotope 65Zn in the body after an injection of 65Zn-labeled compounds.17) We succeeded in visualizing differences in 65Zn distribution among a few Zn complexes. In addition, GREI allows for the visualization of multiple radionuclides simultaneously.18–20) Therefore, the dual radiolabeling approach was applied to noninvasive analysis using GREI in order to investigate the speciation changes of Zn complexes.
Zn complexes have previously been developed as oral antidiabetic agents; one of the main purposes of Zn complexation was to improve bioavailability. However, investigation of speciation changes of Zn complexes after oral administration is lacking. In this study, the bis(5-chloro-7-iodo-8-quinolinolato)zinc complex [Zn(Cq)2] was selected as a dual radiolabeled complex, because the complex shows antidiabetic activity in KK-Ay mice21) and its ligands can be radiolabeled with radioactive iodide.22) We synthesized 65Zn- and 131I-labeled [Zn(Cq)2] and investigated their distribution after a single oral administration both invasively with usual biodistribution experiments and noninvasively with GREI in order to estimate the speciation in vivo.
Sodium hydroxide (NaOH), chloramine T, sodium disulfite (Na2S2O5), acetonitrile, and tetrahydrofuran (THF) were purchased from Nacalai Tesque Inc. (Kyoto, Japan). 5-Chloro-7-iodo-8-quinolinol (clioquinol), and 5-chloro-8-quinolinol (cloxyquin) were purchased from Sigma-Aldrich Inc. (St. Louis, MO, U.S.A.). Sodium dihydrogen phosphate (NaH2PO4) and zinc acetate dihydrate ((CH3COO)2Zn·2H2O) were obtained from Wako Pure Chemical Industries, Ltd. (Osaka, Japan). Zinc chloride (ZnCl2) was purchased from Tokyo Kasei Inc. (Tokyo, Japan). Iodine-131 radionuclide (131I) was obtained from PerkinElmer, Inc. (MA, U.S.A.).
AnimalsMale KK-Ay mice were purchased from CLEA Japan, Inc. All animals were housed under a 12-hour light/dark cycle in a temperature-controlled animal room and allowed free access to food and tap water. All animal experiments were approved by the Ethics Committee on Animal Care and Use of RIKEN and Okayama University, and were performed in accordance with the Guide for the Care and Use of Laboratory Animals.
Synthesis of [Zn(Cq)2](CH3COO)2Zn·2H2O (329 mg, 1.5 mmol) in water (4.0 mL) was added dropwise to clioquinol (5-chloro-7-iodo-8-quinolinol) (Cq) (611 mg, 2.0 mmol) in THF (10.0 mL), and the reaction mixture was stirred at room temperature for 90 min. The yellow precipitate was filtered, rinsed with acetone, and evaporated in a vacuum to obtain the pure product as a yellow solid. Yield: 523 mg (77.6%); 1H-NMR (dimethyl sulfoxide (DMSO)-d6), δ: 7.69 (1H, dd, J=4.2, 8.4 Hz), 7.90 (1H, s), 8.45 (1H, dd, J=4.2, 8.4 Hz), 8.47 (1H, dd, J=4.2, 4.2 Hz); electron ionization (EI)-MS m/z: 674 (M+) Anal. Calcd for C18H8Cl2I2N2O2Zn: C, 32.05; H, 1.19; N, 4.15. Found: C, 32.31; H, 1.05; N, 4.11.
Synthesis of 65Zn-, and 131I-Labeled [Zn(Cq)2]No-carrier-added Na131I (14 MBq) in 0.1 M NaOH (20 µL) was added to cloxyquin (20 µg, 0.11 µmol) in ethanol (25 µL). The solution was buffered to pH 5 by adding 20 µL 0.1 M NaH2PO4. A solution of chloramine-T (20 µg, 0.071 µmol) in water (8 µL) was then added and the reaction mixture allowed to stand at room temperature for 2 min. After quenching the reaction with Na2S2O5 (100 µg, 0.66 µmol) in water (20 µL), the mixture was purified by reverse phase (RP)-HPLC using a COSMOSIL 5C18-AR-ΙΙ, 5 µm, 4.6×150 mm column (Nacalai Tesque Inc.). A mobile phase of acetonitrile/0.1% TFA in water (80/20%, v/v) was used and the flow rate was 0.10 mL/min. The radioactivity peak corresponding to [131I]Cq was collected between 4.2 and 5.2 min in a volume of 0.10 mL. The [131I]Cq solution was evaporated and dissolved in THF. The resultant solution was mixed with non-radiolabeled Cq in THF.
The 65Zn nuclide was produced by a previously described method.14) The purified 65Zn in HNO3 was dried using a heater, and the residue was then dissolved in acetic acid. The solution was dried again and dissolved in water. The resulting solution was mixed with (CH3COO)2Zn·2H2O aqua. (CH3COO)2Zn·2H2O (3.58 mg, 16.3 mmol) containing 65Zn (8.28 MBq) in water (110 µL) was added dropwise to carrier-added [131I]Cq (9.97 mg, 16.3 mmol, 3.43 MBq) in THF (250 µL), and the reaction mixture was stirred at room temperature for 90 min. The yellow precipitate was filtered, rinsed with acetone, and dried up in a vacuum to yield 10.2 mg (92.7%) of [Zn(Cq)2] containing 7.04 MBq of 65Zn (85.0%) and 2.82 MBq of 131I (82.4%). Chemical structure of 65Zn- and 131I-labeled [Zn(Cq)2] is shown in Fig. 1. Before the administration, non-radiolabeled [Zn(Cq)2] was added to 65Zn-, 131I labeled [Zn(Cq)2], and dissolved in PEG400. ZnCl2 was dissolved in PEG400 containing 5% water.

Nine-week-old male KK-Ay mice were orally administered a single dose of 65Zn-, 131I labeled [Zn(Cq)2] or [65Zn]ZnCl2 at a dose of 10 mg Zn/kg of body weight (65Zn: 150 kBq, 131I: 50 kBq per head). Two or six hours after administration, the mice were sacrificed under isoflurane anesthesia. Blood was collected, and the organs (heart, stomach, pancreas, liver, small intestine, kidney, thigh muscle, femur bone, and adipose tissue) were removed. Their radioactivities due to 65Zn and 131I were measured using a calibrated Ge detector or gamma counter. Results were expressed as percent injected dose per gram of tissues (%ID/g).
GREI ExperimentNine-week-old male KK-Ay mouse was orally administered a single dose of 65Zn-, 131I labeled [Zn(Cq)2] at 10 mg Zn/kg of body weight (65Zn: 1.5 MBq, 131I: 0.5 MBq per head). Fifteen minutes after administration, the GREI experiments were carried out under isoflurane anesthesia for 30 min, and for 60 min at 2, 4, 6, and 24 h after administration. The acquired data were recorded in list mode with real-, and live-time information. The distribution images were reconstructed from the acquired data by the adoption of the image-reconstruction methods as previously described.18) The mouse was sacrificed under isoflurane anesthesia right after the GREI experiments at 24–25 h. Blood was collected, and the organs described above in addition to the large intestine were removed. The radioactivity in each tissue was determined using a Ge detector. The radioactivities in the stomach, small intestine, and large intestine were measured with or without their contents. Results were expressed as percent injected dose (%ID).
Image AnalysisTo evaluate the biodistribution of 65Zn and 131I and the speciation of [Zn(Cq)2] noninvasively, regions of interest (ROIs) were drawn around the radioactivity accumulated areas. The radioactivity in each ROI was calculated by integrating the pixel values inside them. In the ROI analysis, the whole body radioactivity at 0–0.5 h after an administration was taken as the administered radioactivity, because the images were acquired before excretion. The ratio of radioactivity in each ROI to the administered radioactivity was calculated for 65Zn and 131I, respectively. The I/Zn ratio that corresponded to the molar ratio of administered I to Zn was calculated by following equation:
 |
 |
The radioactivities of 65Zn and 131I in the resected organs and blood were determined 2 and 6 h after the oral administration of 65Zn-, and 131I-labeled [Zn(Cq)2] or [65Zn]ZnCl2 (Table1). The accumulation of 65Zn in each tissue after an administration of ZnCl2 was much lower than that of [Zn(Cq)2], although the tissue distribution patterns were similar to each other. For [Zn(Cq)2], accumulation of 65Zn and 131I in the stomach wall was the highest at both time points. Accumulation in the stomach wall was probably due to direct absorption and binding to stomach wall after oral administration, as the accumulation of 65Zn in stomach wall was much higher than that in pancreas and liver, locations where Zn physiologically accumulates. Accumulation of both 65Zn and 131I in the stomach wall, small intestine wall, liver, and kidney was higher than that in other tissues. In the pancreas, high accumulation of 65Zn was observed, whereas little accumulation of 131I was found. The accumulation of 65Zn and 131I in most tissues other than the stomach wall and blood at 2 h after administration was as high as that at 6 h. The accumulation of 65Zn and 131I in the stomach wall tended to decrease from 2 to 6 h after an administration. The concentration of 65Zn in the blood at 2 h was significantly higher than that at 6 h.
| Organ | 65Zn (Zn(Cq)2) | 131I (Zn(Cq)2) | 65Zn (ZnCl2) | |||
|---|---|---|---|---|---|---|
| 2 h | 6 h | 2 h | 6 h | 2 h | 6 h | |
| Blood | 0.55±0.06 | 0.28±0.12* | 0.26±0.04 | 0.36±0.10 | 0.07±0.03 | 0.04±0.02 |
| Heart | 0.64±0.17 | 0.64±0.06 | 0.18±0.02 | 0.18±0.03 | 0.04±0.01 | 0.09±0.03* |
| Stomach | 15.34±8.03 | 10.27±3.17 | 29.14±5.61 | 17.39±6.28 | 1.17±0.84 | 0.81±0.91 |
| Pancreas | 4.16±1.19 | 4.15±1.19 | 0.21±0.05 | 0.20±0.05 | 0.42±0.14 | 0.61±0.22 |
| Liver | 1.41±0.35 | 1.77±0.46 | 0.78±0.43 | 0.76±0.25 | 0.18±0.03 | 0.30±0.10 |
| Small intestine | 3.43±1.05 | 3.03±0.50 | 1.09±0.49 | 1.07±0.29 | 0.68±0.23 | 0.64±0.24 |
| Kidney | 1.26±0.35 | 1.48±0.23 | 0.46±0.11 | 0.66±0.15 | 0.25±0.11 | 0.24±0.07 |
| Thigh muscle | 0.10±0.03 | 0.14±0.03 | 0.07±0.02 | 0.11±0.03 | 0.02±0.01 | 0.02±0.01 |
| Femur bone | 0.54±0.01 | 0.64±0.13 | 0.11±0.03 | 0.11±0.04 | 0.05±0.02 | 0.12±0.09 |
| Adipose | 0.05±0.00 | 0.05±0.01 | 0.12±0.03 | 0.17±0.05 | 0.01±0.00 | 0.01±0.00* |
Data are expressed as mean±standard deviation (S.D.) for 3–4 mice. Significance: * p<0.05 vs. 2h. %ID/g, percent injected dose per gram of tissue.
Table 2 shows the molar ratio of administered I to Zn, which suggests speciation of [Zn(Cq)2]. Before administration, [Zn(Cq)2] consisted of Zn and Cq at a molar ratio of 1 : 2, with the I/Zn ratio=2. If Zn and Cq had formed [Zn–Cq], the complex of Zn and Cq would have a molar ratio of 1 : 1 with the I/Zn ratio=1. The I/Zn ratios in the stomach wall and adipose tissue at both time points were higher than 2. In most tissues, the I/Zn ratio were lower than 1. In particular, the values in the pancreas were very low (0.10 at both time points). This indicates that more than 90% of administered Zn that was not coordinated with Cq accumulated in the pancreas. On the other hand, values were relatively high in the thigh muscle (1.47 at 2 h and 1.54 at 6 h).
| Organ | 2 h | 6 h |
|---|---|---|
| Blood | 0.97±0.20 | 2.74±0.59 |
| Heart | 0.59±0.19 | 0.55±0.13 |
| Stomach | 4.29±1.48 | 3.36±0.26 |
| Pancreas | 0.10±0.01 | 0.10±0.01 |
| Liver | 1.19±0.75 | 0.86±0.24 |
| Small intestine | 0.63±0.21 | 0.71±0.13 |
| Kidney | 0.75±0.14 | 0.90±0.18 |
| Thigh muscle | 1.47±0.39 | 1.54±0.36 |
| Femur bone | 0.41±0.10 | 0.36±0.08 |
| Adipose | 4.72±1.31 | 6.75±1.01 |
Data are expressed as mean±S.D. for 3 mice.
The ratio of 65Zn concentration in the pancreas, liver, thigh muscle, and adipose to blood is shown in Table 3. The ratio was the highest in the pancreas, and second highest in the liver. The ratio of each tissue to blood for [Zn(Cq)2] was similar as that for ZnCl2 at both time points.
| Organ | 2 h | 6 h | ||
|---|---|---|---|---|
| Zn(Cq)2 | ZnCl2 | Zn(Cq)2 | ZnCl2 | |
| Pancreas | 7.46±1.16 | 6.45±1.98 | 15.88±2.69 | 14.14±1.51 |
| Liver | 2.53±0.29 | 2.94±0.99 | 6.82±1.22 | 7.02±1.22 |
| Thigh muscle | 0.18±0.04 | 0.31±0.17 | 0.56±0.12 | 0.50±0.21 |
| Adipose | 0.09±0.01 | 0.10±0.02 | 0.19±0.05 | 0.27±0.06 |
Data are expressed as mean±S.D. for 3–4 mice.
High accumulation of both 65Zn and 131I in the stomach was found at 0–0.5 h, with changes in the accumulated areas with time. Although it is difficult to determine from the distribution images the precise organ in which 65Zn and 131I accumulated, the distribution pattern of 65Zn was obviously different from that of 131I at 2–3 and 4–5 h after administration (Fig. 2).
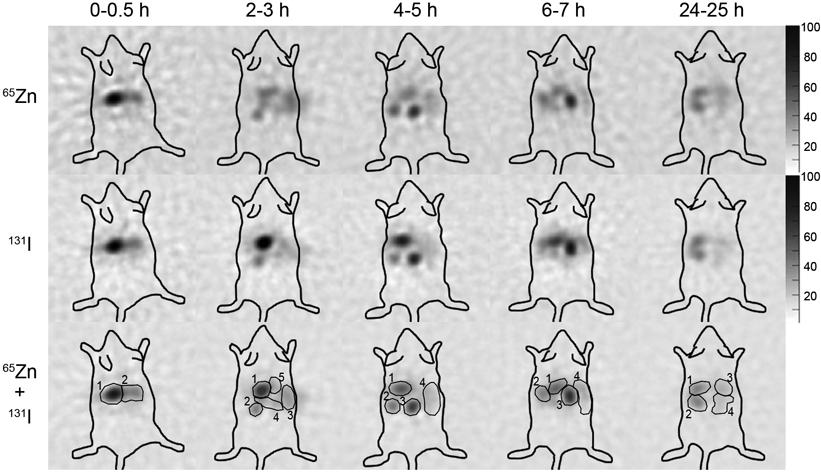
Circles in merge images indicate the ROI around the high accumulation of 65Zn and/or 131I.
Table 4 shows the ratio of image intensity of 65Zn and 131I in each ROI to that in the whole body at 0–0.5 h after administration, the I/Zn ratio, and the MDR of [Zn(Cq)2] in each ROI. The ROI numbers at each time point corresponded to those shown in Fig. 2. At 0–0.5 h, more than a half of 65Zn and 131I in the ROI of No. 1 would correspond to the stomach, and the I/Zn ratio=2.32. At 2–3 h, the highest accumulation of 65Zn was found in ROI of No. 2 with the I/Zn ratio=0.78 in the ROI. At 4–5 h, the I/Zn ratio was 2.50 in the ROI of No. 3 in which 65Zn accumulated most highly, whereas the I/Zn ratios were 1.58 and 1.41 in the ROIs of No. 2 and No. 4, respectively. At 6–7 h, the I/Zn ratios were ˃2.0 in the all studied ROIs. At 24–25 h, the I/Zn ratios were ˃2.0 in the ROIs of No. 1 and No. 2 where a higher accumulation of 65Zn was found. The sum of the MDR at any given time point shows the MDR of [Zn(Cq)2] in the murine body; it was the highest at 2–3 h (11.7%).
| Time | ROI | 65Zn (%) | 131I (%) | I/Zn | MDR (%) |
|---|---|---|---|---|---|
| 0–0.5 h | No. 1 | 51.4 | 59.6 | 2.3 | 0 |
| No. 2 | 22.7 | 25.1 | 2.2 | 0 | |
| 2–3 h | No. 1 | 16.5 | 57.4 | 6.9 | 0 |
| No. 2 | 8.2 | 9.5 | 2.3 | 0 | |
| No. 3 | 19.2 | 7.5 | 0.8 | 11.7 | |
| No. 4 | 10.1 | 10.0 | 2.0 | 0 | |
| No. 5 | 6.8 | 7.2 | 2.1 | 0 | |
| 4–5 h | No. 1 | 12.7 | 39.7 | 6.3 | 0 |
| No. 2 | 13.1 | 10.3 | 1.6 | 2.8 | |
| No. 3 | 22.0 | 27.4 | 2.5 | 0 | |
| No. 4 | 13.8 | 9.7 | 1.4 | 4.1 | |
| 6–7 h | No. 1 | 12.8 | 24.7 | 3.9 | 0 |
| No. 2 | 12.1 | 12.6 | 2.1 | 0 | |
| No. 3 | 28.7 | 41.5 | 2.9 | 0 | |
| No. 4 | 9.5 | 12.9 | 2.7 | 0 | |
| 24–25 h | No. 1 | 13.7 | 13.8 | 2.0 | 0 |
| No. 2 | 13.9 | 14.5 | 2.1 | 0 | |
| No. 3 | 9.2 | 5.0 | 1.1 | 4.2 | |
| No. 4 | 7.4 | 6.0 | 1.6 | 1.4 |
The ROI numbers at each time point correspond to those indicated in Fig. 2.
Table 5 shows the radioactivities of 65Zn and 131I in the resected organs and blood as determined after the GREI experiments at 24–25 h. Accumulation of both 65Zn and 131I in the large intestine was the highest in all studied tissues. Almost all of the accumulation was due to accumulation in its contents.
| Organ | 65Zn | 131I |
|---|---|---|
| Feces/25 h | 8.03 | 12.68 |
| Urine/25 h | 0.36 | 6.96 |
| Heart | 0.27 | 0.03 |
| Stomach* | 1.12 | 1.69 |
| Stomach wall | 0.54 | 0.73 |
| Pancreas | 1.88 | 0.05 |
| Liver | 9.18 | 0.58 |
| Small intestine* | 10.79 | 5.05 |
| Small intestine wall | 3.56 | 0.95 |
| Large intestine* | 32.53 | 25.11 |
| Large intestine wall | 1.62 | 0.39 |
| Kidney | 1.43 | 0.30 |
| Thigh muscle | 0.13 | 0.04 |
| Femur bone | 0.68 | 0.06 |
| Adipose | 0.14 | 0.15 |
*Data shows the accumulation with its contents. %ID, percent injected dose.
In the present study, we investigated the speciation changes of 65Zn- and 131I-labeled [Zn(Cq)2] after a single oral administration by analyzing the distribution of [131I]Cq as well as 65Zn. Conventional invasive assays showed that most [Zn(Cq)2] was dissociated, particularly when examining the distribution to target tissues. The GREI experiments visualized the behavior of 65Zn and [131I]Cq from the stomach to large intestine through the small intestine, and found that most administered Zn was transported together with Cq. These results imply that Zn associated with Cq during absorption phase was highly absorbed by the intestinal tract, with the majority of [Zn(Cq)2] dissociated through the distribution processes to the target tissues. We successfully evaluated the speciation changes of orally administered [Zn(Cq)2] using this dual radiolabeling method.
After the single oral administration of 65Zn- and 131I-labeled [Zn(Cq)2], the highest accumulation of 65Zn and 131I was observed in the stomach wall, with the I/Zn ratio=4.29 at 2 h. This implies that more than half of Cq dissociated from the [Zn(Cq)2] complex in the stomach wall. It is well known that the environment within the stomach is acidic (approximately pH 2) and that metal ions of metal complexes tend to be dissociated in such strongly acidic conditions. Although the species distribution of [Zn(Cq)2] was not determined, the ligand Cq is highly protonated under acidic conditions.23) The [Zn(Cq)2] complex in the stomach wall might have dissociated to Zn and Cq. Van Campen and Mitchell demonstrated that Zn was not absorbed from the stomach wall, but rather from the wall of small intestine, especially the duodenum.24) It has also reported that Cq is not absorbed from the stomach wall.25) However, the highest accumulation of 65Zn also observed after an administration of [65Zn]ZnCl2 was also in the stomach wall. Thus, we hypothesize that the high accumulation in the stomach wall in the present study is due to binding to the surface of gastric mucosa. In the blood, at 2 h after an administration, the I/Zn ratio was almost 1.0. This implies that at least half of the [Zn(Cq)2] was dissociated when absorbed into the systemic circulation, although the ratio of [Zn(Cq)2], [Zn–Cq], and dissociated Zn or Cq in blood was unknown. In the distribution phase, we focused on the accumulation in the pancreas, because it is the only organ, except the metabolic organs, in which 65Zn highly accumulates (Table 1). In the pancreas, the I/Zn ratios were almost 0.1, indicating that almost all the [Zn(Cq)2] was dissociated through distribution to the pancreas. In contrast, the I/Zn ratios were relatively high in the thigh muscle, and extremely high in adipose tissue, which is another promising target tissue of Zn.11,26) This suggests that the distribution of Zn to such tissues is affected by its association with Cq in the blood. However, the ratios of organ to blood of 65Zn was similar between [Zn(Cq)2] and ZnCl2. This implies that almost all the 65Zn distributed to such organs was not coordinated with Cq. High I/Zn ratios in the thigh muscle and the adipose tissue are due to higher levels of dissociated Cq that demonstrate a higher degree of lipophilicity than that noted for the levels of dissociated Zn in the tissue. Noteworthy, the accumulation of 65Zn in all studied tissues for [Zn(Cq)2] was much higher than that for ZnCl2, which indicates higher bioavailability of [Zn(Cq)2]. These results suggest that Zn associated with Cq is highly absorbed by the intestinal tract, and this association is dissociated during distribution to the target tissues of Zn.
Nuclear medicine imaging is powerful tool for early drug development.27) GREI is a novel one that enables the simultaneous imaging of multiple radionuclides including many radionuclides not detected by conventional imaging modalities.19,20) Nuclear medicine imaging is generally performed after an intravenous administration of radiolabeled compounds in order to investigate the tissue distribution pattern of drugs. In recent years, it was reported that this technique is also useful for the assessment of the oral drug absorption process in vivo.28–30) In the present study, the accumulation of 65Zn and 131I were noninvasively visualized using GREI after an oral administration of 65Zn- and 131I-labeled [Zn(Cq)2]. The distribution assays after GREI experiments showed the highest accumulation of both 65Zn and 131I in the large intestine, especially in its contents. This implies that the behavior of 65Zn and 131I from the stomach to large intestine through the small intestine was accurately visualized in the GREI experiments. Differences in the distribution of 65Zn and 131I were not observed at 0–0.5 h, but were observed at 2–3, and 4–5 h after an administration. The ROI analysis shows that at least 11.7% of [Zn(Cq)2] was dissociated in the body at 2–3 h after an administration (Table 2). However, the distribution of the majority of 65Zn was similar to that of 131I. This suggests that Zn is associated with Cq during the absorption process. This might contribute to the higher absorption rate of 65Zn for [Zn(Cq)2] than ZnCl2, as observed in distribution analysis (Table 1).
In conclusion, we synthesized 65Zn- and 131I-labeled [Zn(Cq)2], and investigated the speciation changes of the complex after a single oral administration by analyzing the distribution of [131I]Cq as well as 65Zn. Typical invasive assays showed that most [Zn(Cq)2] was dissociated, particularly as it was distributed from the systemic circulation to the target tissues. The behavior of 65Zn and [131I]Cq from the stomach to large intestine through the small intestine was visualized by GREI. The ROI analysis showed that the majority of the administered Zn was transported together with Cq. Higher accumulation of 65Zn for [Zn(Cq)2] than ZnCl2 was also observed. These results imply that Zn coordinated with Cq at the absorption phase was highly absorbed by the intestinal tract with most [Zn(Cq)2] dissociated during the distribution to target tissues. We successfully evaluated the speciation changes of orally administered [Zn(Cq)2] using dual radiolabeling method. These results suggest that an improvement in Zn bioavailability is essential in producing a potent antidiabetic effect for [Zn(Cq)2].
This work was supported by a Grant-in-Aid for the Japan Society for the Promotion of Science Fellows to M. Munekane (No. 26·8219).
The authors declare no conflict of interest.