2018 Volume 41 Issue 8 Pages 1299-1302
2018 Volume 41 Issue 8 Pages 1299-1302
Monolaurin is a natural compound that has been known for its broad antimicrobial activities. We evaluate the antifungal activity of monolaurin against Candida albicans biofilms in vivo using a novel bioluminescent model to longitudinally monitor oral fungal infection. Oral fungal infection in vivo was performed using bioluminescent engineered C. albicans (SKCa23-ActgLUC) biofilms on Balb/c mice. The antifungal activity of monolaurin was determined by comparing three groups of mice (n=5/group): monolaurin, vehicle control, and positive control (nystatin). All mice were immunosuppressed with cortisone acetate and oral topical treatments were applied for 5 d. In vivo imaging system (IVIS) imaging was used to monitor the progression of infection over a 5-d period. Total photon flux and ex vivo microbiological analysis of the excised tongues were used to determine the overall fungal burden. Oral topical treatments of monolaurin have resulted in a significant decrease (p<0.05) in the total photon flux over 4 and 5 d post-infection in comparison to the vehicle control group. Furthermore, monolaurin treated group had a significant decrease in colony formation unit of tongue tissue compared to the vehicle control. Our findings support monolaurin as a promising antifungal compound in vivo, which may translate to its future use in the treatment of oral candidiasis.
Oral candidiasis (OC) is a common fungal infection that affects the oral cavity.1,2) Candida albicans is often implicated in OC, which can cause oral thrush, especially among immunocompromised individuals.3) Due to the limitation of the current antifungal drugs as a result of an increase in drug resistance to azole antifungal agents as well as the side effects of those medications, the need for new effective antimicrobials with minimal side effects is on the rise.4) One important virulence factor of C. albicans that is often studied as a contributing factor to its pathogenicity and its antifungal resistance is biofilm formation.5) Therefore, the development of new therapeutic agents that can inhibit biofilm formation will likely lead to the treatment of OC.
Natural products present as potential alternative therapeutic options for several infectious diseases, as antimicrobial agents.6–12) Monolaurin (glycerol monolaurate) is a natural compound that is commonly found in coconut oil,13) which is also considered Generally Recognized as Safe14) (GRAS) for use in the food industry by the Food and Drug Administration (FDA).15) Recent studies have shown monolaurin to have broad bioactivity; for example as antibacterial properties.13,16) In fact, monolaurin was reported to inhibit biofilm formation of Gram-positive bacteria, Staphylococcus aureus, with no drug resistance at a sub-growth inhibitory concentration.17) Similarly, our previous antifungal study performed in vitro on C. albicans biofilms, showed monolaurin to have a strong fungicidal activity.18) It was then concluded that monolaurin has potential for future therapeutic use, possibly as a topical oral application. To the best of our knowledge, an in vivo study investigating the antifungal effects of monolaurin against oral C. albicans biofilms has never been reported.
For the first time, this study primarily evaluates the antifungal activity of monolaurin against C. albicans biofilms in vivo. Furthermore, a mouse model of OC using a non-invasive bioluminescent imaging to monitor real-time progression of the fungal infection was used. The significance of this work is to validate the efficacy of monolaurin to treat and/or prevent OC in vivo.
Monolaurin (12.5 mmol/L) (99.9% HPLC grade) was purchased from Sigma-Aldrich (Sigma-Aldrich, MO, U.S.A.). Nystatin (100 mmol/L) (Sigma-Aldrich) and phosphate buffered saline (PBS) (Lonza, MD, U.S.A.), served as positive and vehicle controls, respectively. A bioluminescent C. albicans SKCa23-ActgLUC was used, which is an engineered Wild-type C. albicans SC5314 to express C. albicans codon-optimized Gaussia princeps luciferase (gLuc) at the cell wall under the control of Actin (ACT1) promoter.19) This strain was kindly provided by Dr. Van Dijck (VIB, Leuven, Belgium).
All animal procedures were performed in accordance with the Institutional Animal Care and Use Committee of USC (protocol # 20266), and the Panel on Euthanasia of the American Veterinary Medical Association (A3518-0). In this study, 15 inbred Balb/c mice (6–7 weeks old), were housed in a pathogen-free conditions at USC Animal Vivarium under full supervision of attending veterinarian. Treatment groups (n=5 in each group) were three; monolaurin (12.5 mmol/L), vehicle control group, PBS, and positive control (nystatin 100 mmol/L). The concentration of monolaurin (100×the minimum inhibitory concentration (MIC)) was selected based on data from a previously published18) in vitro study on biofilm inhibition of C. albicans. All mice were immunosuppressed prior to oral fungal infection by administration of subcutaneous injections of cortisone acetate (225 mg/kg) (Sigma-Aldrich). Immunosuppression was maintained throughout the experiment by administering cortisone acetate injections every other day.20) A 2-d old C. albicans (SKCa23-ActgLUC) biofilm inoculum (1×107 cells/mL) was suspended in Hanks’ balanced salt solution (HBSS).20) On the same day of the infection, mice were sedated with isoflurane oral inhalation (2–4%). Calcium alginate swabs saturated with C. albicans suspension (1×107 cells/mL) were placed sublingually in the oral cavities for 75 min.20,21) Starting the next day post-infection, topical applications of 20 µL of the respective treatments were pipetted into the oral cavity and then brushed for 30 s with a sterile camel brush using gentle circular strokes. Monolaurin (12.5 mmol/L) and the control group treatments were applied twice daily at 6-h intervals. For the imaging of infection, mice were sedated with isoflurane inhalation (2–4%) and coelenterazine (10 µL of 0.5 mg/mL in 1 : 10 ethanol : PBS) (Promega) substrate was pipetted into the oral cavities. Consecutive scans (i.e., 3 images) with acquisition time of 20 s were acquired after coelenterazine administration. Total photon emission from oral cavities were quantified region of interest (ROI) for each mouse, using IVIS-200TM Imaging system (Xenogen Inc.) and Living ImageR software package.21) Monitoring the progression of the infection at different time points; 1, 4, and 5 d post-infection, was based on IVIS imaging. Mice were then euthanized on day 6 post-infection. Ex vivo of the microbial load of the tongues was performed using plating of serial dilutions of organ homogenates onto Sabouraud Dextrose Agar (BD, Franklin Lakes, NJ, U.S.A.). Colony formation units were standardized as CFUs/mL/g of tongue tissue weight. The toxicity was investigated through nutrition status (weight gain), development and external examination (skin and natural orifices).22)
Statistical AnalysisAll data were expressed as the mean±standard error of the mean (S.E.M.) and were analyzed using JMP software (version Pro 11.0.0; SAS Institute Inc.). Analysis of CFUs was performed using Kruskal–Wallis test, followed by Dunnett test. Photon emission quantification was analyzed by repeated measures of ANOVA (PROC MIXED), using SAS. Tukey–Kramer test was then used to assess significance (α=0.05, p<0.05).
The activity of monolaurin as an antifungal agent was compared to nystatin as a positive control and PBS as a vehicle control was using quantification of fungal load by IVIS longitudinal imaging over a 5-d period. Images at day one post-infection served as baseline values confirming infection of all the mice were infected and were used to quantify the initial fungal burden of the oral cavities. After treatments started to be applied, images were captured at two different time points to assess efficacy of the tested treatment to diminish OC. It was found that at days 4 and 5 post infection, there was a significant reduction (p<0.05) in the total photon flux in the monolaurin treated and the positive control groups in comparison to the vehicle control, which had a continuous increase in the total photon flux over a 5-d imaging period (Fig. 1).
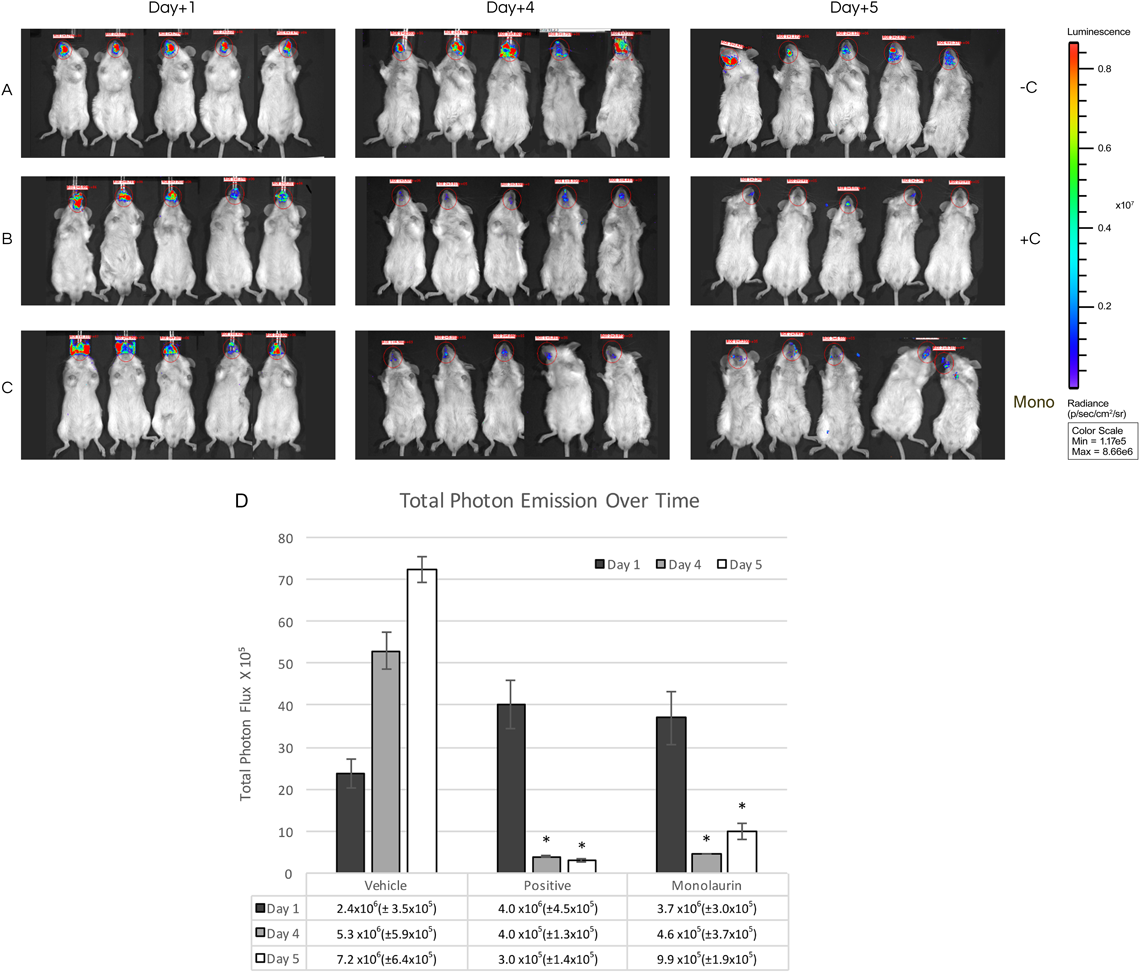
In vivo images from one representative scan (consecutives frames were acquired). (A) Mice (−C) treated with PBS, vehicle control. (B) Treated (+C) with nystatin (100 mmol/L), positive control. (C) Treated (Mono) with monolaurin (12.5 mmol/L). (D) Total photon flux quantification from the oral cavities of the mice, grouped as vehicle control, positive control (treated with nystatin), and monolaurin treated group at days 1, 4, and 5 post-infection. Mean±standard deviation. Significant decrease in total photon flux was observed at days 4 and 5 in monolaurin and nystatin treated groups (* p<0.05). (Color figure can be accessed in the online version.)
At day 6 post-infection, mice were euthanized, and the tongues were excised for to evaluate the microbial load (CFU). The microbiological analysis confirmed the significant reduction (p<0.05) of CFUs/mL/g of tissue in the monolaurin and nystatin treated groups in comparison to the vehicle control group (Fig. 2).

Fungal load of the tongues was expressed as colony formation unit (CFU/mL/g of tissue). Monolaurin group showed reduction in colony count compared to vehicle control group (* p<0.05).
In addition, the external examination of the mice was assessed to determine the nutritional status and normal development. There were no signs of disease in the treated group in comparison to the controls as well as no significant differences in the weight gains among the groups (data not shown). Furthermore, macroscopic observation of vital organs such as heart, lung, liver, stomach, intestine, and kidneys showed no pathological signs in the experimental group.
The aim of this study was to examine monolaurin as an antifungal agent against C. albicans biofilms in vivo using longitudinal imaging of oral infection.7,8,21) Natural compounds have been known to have antimicrobial benefits, rendering them as promising therapeutic agents against infectious disease.23) Monolaurin is an FDA approved as food additive substance and classified as GRAS.14) We have previously tested the antifungal efficacy of monolaurin in eradicating C. albicans biofilms in vitro18) and found strong fungicidal activity compared to the vehicle control (PBS), and the positive control (nystatin) groups. In this animal model, we compared the antifungal effects of monolaurin (12.5 mmol/L or 100×MIC) to the vehicle control (PBS), and the positive control (nystatin 100 mmol/L).
In our in vivo experiments, a bioluminescent strain of C. albicans (SKCa23-ActgLUC) was used in combination with coleranterazine substrate in order to image and monitor OC progression of infections longitudinally.21) Under continuous administration of cortisone acetate, mice were maintained susceptible to fungal infection.20) It was found that with oral topical treatments of monolaurin (12.5 mmol/L) there was a significant reduction in the fungal load of the OC over a period of 5 d. At day 4-post infection, monolaurin was as effective in reducing the total photon flux as nystatin, the most commonly used oral antifungal used for treatment OC24) (Fig. 1D). At day 5 post-infection, there was a noted slight increase in the total photon emission in the monolaurin treated group. However, the photon flux for monolaurin group at day 5 was still significantly (p<0.05) lower than that the vehicle control group. The overall microbiology analysis of the ex-vivo tongue samples confirmed the efficacy of monolaurin as a potent antifungal therapeutic agent, as there was a significant reduction in CFUs/mL/g of tongue tissue in the monolaurin treated group in comparison to the vehicle control group (Fig. 2).
For the first time, the findings of this in vivo study, support monolaurin as a promising antifungal natural compound, which may translate into future clinical trials to test its efficacy in the treatment and/or the prevention of OC.
Research was supported by National Institute of Dental and Craniofacial Research (NIDCR)/National Institutes of Health (NIH) T90 (Grant DE021982), Capes (Grant #006211/2015-01), and the National Center for Complementary and Integrative Health (NCCIH) of NIH (R00AT006507). We are grateful to Dr. Van Dijck (VIB, Leuven, Belgium) for kindly providing SKCa23-ActgLUC strain.
The authors declare no conflict of interest.