2026 年 49 巻 3 号 p. 402-407
2026 年 49 巻 3 号 p. 402-407
Antimicrobial resistance (AMR) has become a major global health threat, prompting the implementation of antimicrobial stewardship (AS) programs to optimize antimicrobial use. Pharmacists play a vital role in these efforts. In Japan, a series of national policy measures has been launched as part of the 2016 National Action Plan on AMR, and systems have been established in parallel to support the active involvement of pharmacists with advanced expertise in infectious diseases. This narrative review provides an overview of the expanding contributions of pharmacists to AS across diverse healthcare settings in Japan. In large hospitals with relatively abundant human resources, pharmacists have promoted post-prescription review, dose optimization, and de-escalation, resulting in reduced use of broad-spectrum antibiotics, shortened treatment durations, cost savings, and even lower resistance rates. Further, in small- and medium-sized hospitals, where infectious disease specialists are often unavailable, pharmacist-led stewardship efforts are gradually emerging. Community pharmacists have also expanded AS efforts outside hospital settings by surveilling outpatient antibiotic prescriptions and identifying leftover antibiotics during home visits. In addition, multicenter collaborative studies and analyses using large-scale administrative claims databases are contributing to the generation of robust real-world evidence, facilitating sustained surveillance and policy evaluation. To maintain and further enhance these efforts, fostering the next generation of AS professionals and developing novel indicators and predictive models are essential. Pharmacist-led AS in Japan is expected to continue progressing, with sustainable contributions needed across healthcare situations of all field.
Antimicrobial resistance (AMR) has become a critical global public health threat. In 2019, drug-resistant pathogens were estimated to be responsible for approximately 1.27 million deaths. Importantly, nearly 5 million deaths were associated with AMR, positioning it as one of the leading causes of mortality worldwide.1) If current trends continue, AMR-related deaths could reach 10 million annually by 2050.1) Resistant infections not only prolong hospital stays, but also incur higher treatment costs, placing a substantial economic burden on healthcare systems.2) Therefore, AMR presents both serious health and economic challenges, requiring comprehensive and coordinated countermeasures.
Antimicrobial stewardship (AS) has been recognized as a fundamental strategy to preserve the effectiveness of antimicrobial agents. AS programs aim to support the optimization of antimicrobial selection, dosing, and duration through multidisciplinary teams including infectious disease specialists.3) This approach improves clinical outcomes, reduces unnecessary antibiotic exposure, and provides economic benefits.3,4) Among the healthcare professionals involved in AS efforts, pharmacists play a key role owing to their expertise in pharmacotherapy and accessibility. Pharmacist-led interventions—such as prospective audit and feedback, dose optimization, and education—have been shown to significantly improve the quality of antibiotic prescribing and reduce inappropriate use.4)
In Japan, the national policy framework has increasingly emphasized AS in recent years. Particularly, the government’s National Action Plan on AMR (2016–2020) outlined strategic goals to curb AMR through prudent antibiotic use5) (Fig. 1). Building on this foundation, a 2017 multisociety guidance on appropriate antimicrobial use provided a blueprint for implementing hospital AS programs. In 2018, AS-specific reimbursement incentives (a dedicated “Antimicrobial Stewardship Fee” alongside existing infection control premiums) were introduced in the healthcare system to encourage formal AS activities. This has bolstered pharmacist engagement in stewardship.6)

In addition, professional bodies established pharmacist certification programs in infectious diseases—exemplified by the Infection Control Pharmacy Specialist credential launched in 2005 and the Antimicrobial Chemotherapy Pharmacist certification by the Japanese Society of Chemotherapy in 2008—to develop AS experts.6) Pharmacists now play vital roles in AS across hospitals, community pharmacies, and educational settings. This narrative review aimed to provide an overview of pharmacist contributions to AS in these various settings in Japan.
Until Japan’s first National Action Plan on Antimicrobial Resistance (2016–2020), pharmacist-led AS activities in large Japanese hospitals primarily focused on inpatient settings and centered around post-prescription review and feedback, antimicrobial restriction, and guideline adherence. Key interventions included reviewing prolonged or inappropriate intravenous therapy, optimizing empirical selections, and promoting de-escalation or intravenous-to-oral conversion. These efforts resulted in tangible outcomes: significant reductions in the use of broad-spectrum agents, such as carbapenems and antipseudomonal antibiotics,7–11) shorter treatment durations,9,10) and lower antibiotic-related costs.7,11) Resistance patterns were also improved; these included decreased isolation rates of methicillin-resistant Staphylococcus aureus (MRSA) and enhanced susceptibility of Pseudomonas aeruginosa to fluoroquinolones.8,10) Moreover, the introduction of rapid diagnostic tools such as matrix-assisted laser desorption/ionization time-of-flight mass spectrometry combined with pharmacist-led interventions has improved the time to effective therapy and reduced clinical failure in bloodstream infections.12) Metrics such as defined daily doses (DDDs), days of therapy (DOT), and incidence of resistant pathogens were commonly employed.7–9,11) During this phase, pharmacists were positioned primarily within infectious disease or infection control teams, playing a consultative role to prescribers.
However, since 2020, pharmacist-driven AS in large hospitals has expanded in scope, depth, and integration. In addition to inpatient stewardship, newer interventions have targeted specific populations and settings (e.g., urology, ward based and outpatient), introduced behavioral audits, and implemented hospital-wide collaborative system.13–15) Pharmacists now proactively participate in real-time feedback, multidisciplinary ward rounds, and dosing optimization based on pharmacokinetics. Recent studies have also adopted broader and more diverse indicators, such as days of antibiotic spectrum coverage (DASC) and the WHO’s Access, Watch, and Reserve (AWaRe) classification, to quantify the appropriateness of antimicrobial selection over time.16,17) Notably, AS teams have demonstrated adaptability in managing antimicrobial use during external pressures, such as the coronavirus disease 2019 pandemic.18) In large-scale hospitals, pharmacists are increasingly immersed in clinical workflows—not only enforcing AS policies, but also designing data-driven AS strategies that balance spectrum narrowing with clinical safety. Compared to small- and medium-sized hospitals, larger hospitals have relatively greater availability of human resources, enabling pharmacists to take on these expanded roles.19,20) Overall, recent studies reaffirm the evolving and essential leadership role of pharmacists in optimizing antimicrobial use across various clinical departments.
2.2. Small- and Medium-Sized HospitalsIn contrast to large hospitals, small- and medium-sized institutions in Japan often face limited human and material resources. Consequently, pharmacist contributions to AS in these settings have historically been underreported. However, recent studies have highlighted the important role pharmacists play in facilities where infectious disease (ID) physicians are frequently unavailable. Current evidence suggests that AS initiatives in these hospitals can lead to substantial improvements, including reduced antimicrobial expenditures,21,22) improved susceptibility rates,21) shortened durations of therapy,22,23) and increased rates of de-escalation.22,23) Notably, even in the absence of ID physicians, these initiatives have demonstrated meaningful clinical and operational impact, underscoring the feasibility of pharmacist-led AS in resource-constrained environments.22,24) Furthermore, amid global shortages of antimicrobials, AS teams involving pharmacists have been shown to effectively manage alternative antibiotic selection, ensuring continuity of care without compromising outcomes.25) With respect to evaluation metrics, emerging indicators such as DASC24,25) are being increasingly adopted alongside traditional measures such as DDD21) and DOT24,25) regardless of institutional size.
2.3. Multicenter Collaborative StudiesMulticenter collaborative studies are essential when single-center investigations are insufficient to evaluate the effectiveness of AS or when enhancing external validity is required. Despite their importance, such studies have remained relatively scarce in Japan. Nevertheless, collaborative efforts between pharmacists and physicians have begun to clarify important topics, such as the effectiveness of AS in therapeutic drug monitoring (TDM) and actual prescribing practices for outpatients.26,27) In addition, multicenter settings have enabled the evaluation of novel indicators (e.g., the modified antibiotic heterogeneity index28,29) and point-prevalence surveys30)), demonstrating their utility in AS research and suggesting the value of broader and more sustained surveillance initiatives.
2.4. Community PharmaciesPromoting AS requires the engagement of not only hospital pharmacists, but also community pharmacists. Recent nationwide investigations by community pharmacy-based pharmacists have clarified patterns of antibiotic use based on outpatient prescription data,31,32) as well as the extent of leftover antibiotics found during home visits.33) These findings highlight the growing importance of community pharmacists in AS and underscore the need to further strengthen their involvement in national stewardship efforts.
2.5. Real-World Evidence Based on Large-Scale DatabasesIn recent years, Japan has shifted from evaluating AS efforts primarily at the institutional level to conducting broader, population-based analyses using large-scale administrative databases. These studies have enabled researchers to assess the impact of nationwide policy interventions and prescribing trends across the healthcare system. For instance, claims-based studies have demonstrated that the introduction of a new reimbursement policy that rewards physicians for not prescribing antibiotics for pediatric upper respiratory tract infections led to a significant nationwide reduction in antibiotic prescriptions.34,35) Japan has also established systems such as the Japan Surveillance of Antimicrobial Consumption,36) which monitors antibiotic use over time based on sales and claims data, allowing for longitudinal evaluations of stewardship activity. Furthermore, a nationwide surveillance system known as the Japan Surveillance for Infection Prevention and Healthcare Epidemiology37) has been developed to automatically collect data on antimicrobial use and resistance patterns directly from hospital information systems. This platform has facilitated regional evaluations of the relationship between antimicrobial consumption and resistance trends.38) Collectively, these improvements illustrate the growing role of database-driven research in providing a comprehensive picture of prescribing behavior and in evaluating the real-world impact of national AS policies.
Beyond tracking prescription patterns, large-scale databases have enabled more sophisticated evaluations of AS outcomes and metrics. For example, a cluster analysis of 124 Japanese hospitals showed that institutions with high levels of AS and infection control activity had significantly lower MRSA isolation rates, validating the structural impact of well-resourced stewardship programs.39) Other studies have examined pharmacist-led contributions to TDM, including the appropriateness and effectiveness of dosing strategies for antimicrobial agents requiring close monitoring and the incidence of adverse events.40,41) Similarly, real-world data have been used to assess institutional adherence to clinical guidelines.42) These findings highlight the expanding role of large databases in evaluating not only AS interventions, but also clinical outcomes, resistance trends, and variability in clinical practice. As a result, the landscape of AS research in Japan now encompasses the ability to generate hypotheses, conduct benchmarking, and monitor national strategies with a level of precision that complements the efforts of individual hospitals.
Efforts to advance AS in Japan are increasingly focused on establishing standardized metrics and leveraging data infrastructure to evaluate and guide interventions (Fig. 2). A key priority is the development of novel outcome and process indicators that enable benchmarking of antibiotic use and stewardship performance across institutions.43) For example, a predicted, standardized carbapenem consumption metric was developed using a nationwide hospital database, providing a benchmark to promote more appropriate antimicrobial use.43) Similarly, integrating refined indicators such as DASC into surveillance systems has been proposed to better capture de-escalation and spectrum management at scale.44) Prescribing guidance itself also needs to be standardized. A comparative study revealed that only 29% of antimicrobial agents in Japan include recommended treatment durations in their package inserts, markedly lower than the 84% rate in the United States.45) This lack of clarity can lead to unnecessarily prolonged therapy and undermine pharmacist-driven stewardship efforts. Real-world data systems will play a key role in tracking adherence to such indicators, thereby supporting data-driven AS evaluation.
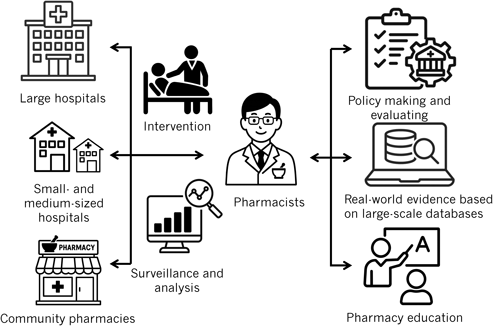
Another priority is the reinforcement of AS human resources and the development of educational infrastructure. Surveys indicate that a significant proportion of Japanese medical students and trainees harbor misconceptions about appropriate antimicrobial use. For instance, nearly half incorrectly believe antibiotics are effective for the common cold. Further, awareness of the national AMR Action Plan remains limited.46) Similar gaps exist among pharmacy students, where AS content is not uniformly included in the curriculum, and few institutions have infectious diseases faculty to support experiential learning.47) To address this, expert panels have called for nationwide implementation of a core AS curriculum for pharmacy education and policy support to increase the number of infectious disease educators for pharmacists.6,47)
At the institutional level, AS remains unevenly implemented. While tertiary hospitals have well-established teams, small- and medium-sized hospitals often lack trained staff or financial resources to support stewardship.6) To address this disparity, it is essential to strengthen the deployment of certified pharmacists in small- and medium-sized hospitals. Further, AS efforts should be expanded into under-resourced settings, such as outpatients and long-term care facilities, to ensure equity in stewardship practices. Finally, economic evaluations of AS in Japan remain limited. Although most intervention studies report cost savings and stable or improved clinical outcomes, few have comprehensively assessed AS program costs or compared economic impact across settings. A recent scoping review emphasized the need for robust, Japan-specific health economic data to strengthen stewardship investment decisions and facilitate international comparisons.48)
Japan’s experience to date highlights that pharmacists, through their specialized pharmacotherapy knowledge and collaborative roles, are indispensable leaders in the fight against AMR. To maintain and further enhance AMR efforts, fostering the next generation of AS professionals and developing novel indicators and predictive models are essential. Pharmacist-led AS in Japan is expected to continue progressing, with sustainable contributions needed across healthcare situations in all fields.
This work was supported by the Ministry of Health, Labour, and Welfare (Grant Number: 23HA2002).
Yuichi Muraki has received funding for commissioned research from Kowa Company, Ltd. and a medical education grant from Pfizer Japan, but this study is not directly related to that funding. Norio Ohmagari has no conflict of interest.