2014 Volume 62 Issue 1 Pages 122-124
2014 Volume 62 Issue 1 Pages 122-124
A new spiroindene pigment, phelliribsin A, was isolated from the medicinal fungus Phellinus ribis, and its structure was determined by two dimensional (2D)-NMR methods. Phelliribsin A is an unprecedented spiroindene compound, and was found to have cytotoxic activity against PC12 cells at a concentration of 30 µM.
Mushrooms are important sources of physiologically beneficial molecules, and are therefore considered to be functional foods. They produce a variety of classes of secondary metabolites with interesting biological activities, and thus have the potential to be used as valuable chemical resources for drug discovery.1–3) Several mushrooms that belong to the genera Phellinus have been used as traditional medicines for the treatment of gastrointestinal cancer, liver or heart diseases, and stomach ailments.4) Polysaccharides, in particular, β-glucan are considered to be responsible for their main biological activity, and there are many reports in the literature on the isolation and biological activity of polysaccharides derived from the Phellinus bodies.5–7) Interestingly, the genera Phellinus commonly elaborates a number of yellow antioxidant pigments that are composed of hispidin derivatives and polyphenols.8–12)
Phellinus ribis, which is distributed throughout East Asia, is a white-rot fungus that prefers to live on stumps of Rosa polyantha and Weigela subsessilis. Recently, we reported that the MeOH extract of the fruiting body of P. ribis was found to promote the neurite outgrowth of nerve growth factor (NGF)-mediated PC12 (pheochromocytoma) cells, and four benzofuran ribisins A–D showed neurotrophic activities.13) In our ongoing efforts to search for neurotrophin-like natural products, we have isolated a new compound named phelliribsin A (1) and two known yellow pigment phelligridin F (2)10) and inoscavin B (3).14) In this paper, we report the structure of 1 and its cytotoxic activity on PC12 cells.
Phelliribsin A (1) was obtained as an orange amorphous material. The molecular formula C22H16O7 with 15 degrees of unsaturation was established by high-resolution (HR)-FAB-MS (m/z 393.0988 [M+1]+). The IR spectrum of 1 revealed the presence of a hydroxyl group at 3275 cm−1, an α,β-unsaturated carbonyl group at 1627 cm−1, and aromatic groups at 1594, 1543 cm−1. The 1H-NMR spectrum of 1 showed signals attributed to a 1,3,4-trisubstituted phenyl moiety at δH 6.79 (1H, d, J=8.1 Hz), 7.01 (1H, dd, J=8.1, 2.0 Hz), 7.11 (1H, d, J=2.0 Hz); a trans-disubstituted double bond at δH 6.91 and 7.46 (1H each, d, J=15.9 Hz); four uncoupled aromatic and/or olefinic protons at δH 5.95, 6.61, 7.02, and 7.92 (1H each, s); and a methyl ketone group at δH 2.35 (3H, s). The protonated carbons and their bonded protons were unambiguously assigned by the heteronuclear multiple quantum coherence (HMQC) experiment, which provided 12 quaternary carbons (δC 96.3, 128.3, 133.8, 137.2, 143.0, 147.0, 148.4, 149.5, 150.0, 187.5, 194.6, and 202.5). To establish the connectivities of the carbons through two or three bonded long-range couplings, heteronuclear multiple bond correlation (HMBC) experiments were carried out. The HMBC correlations of H-8 (δH 7.11, d, J=2.1 Hz) with C-6, C-9, and C-12, H-12 (δH 7.01, dd, J=8.1, 2.0 Hz) with C-6, H-11 (δH 6.79, d, J=8.1 Hz) with C-9 and C-10, H-5 (δH 6.91, d, J=15.9 Hz) with C-6 and C-7, H-6 (δH 7.46, d, J=15.9 Hz) with C-5, C-7, C-8, and C-12 revealed the presence of a trans-9,10-dihydroxystyryl unit in 1. Moreover, the HMBC correlations of two uncoupled aromatic protons H-8′(δH 6.61)/C-6′, 7′, and C-9′ and H-5′(δH 7.02)/C-4′ and C-6′ established the presence of a tetrasubstituted benzene ring. In addition, the HMBC correlations of H-3′/C-1, C-4′, and C-9′ and H-8′/C-1 allowed us to form a 1,1-disubstituted −6′,7′-dihydroxyindene moiety. Furthermore, the HMBC correlations of H-3 with C-1, C-2, C-2′ and C-4 in combination with the chemical shift values of C-1–4 (δC 96.3, 202.5, 104.4, and 187.5, respectively), and the peak at 1627 cm−1 in the IR spectrum, indicated the presence of a 1,1-disubstituted-2(3H)-furanone moiety. In consideration of the 15 degrees of unsaturation of 1, C-1 of the indene moiety had to be bonded through an oxygene atom of C-4 to form a spiroindene structure unit, which was supported by the chemical shift values of the C-1 (δC 96.3) and C-4 (δC 187.5). Additionally, HMBC correlations of both H-5 and H-6 with C-4 and H-3 with C-5, indicated that the trans-9,10-dihydroxystyryl unit was linked to C-4. Compound 1 was found to be optically inactive, a racemate due to no specific rotation. This suggests that the biosynthesis of the spiroindene moiety of 1 is nonstereoselective. Therefore, phelliribsin A was represented as 1.
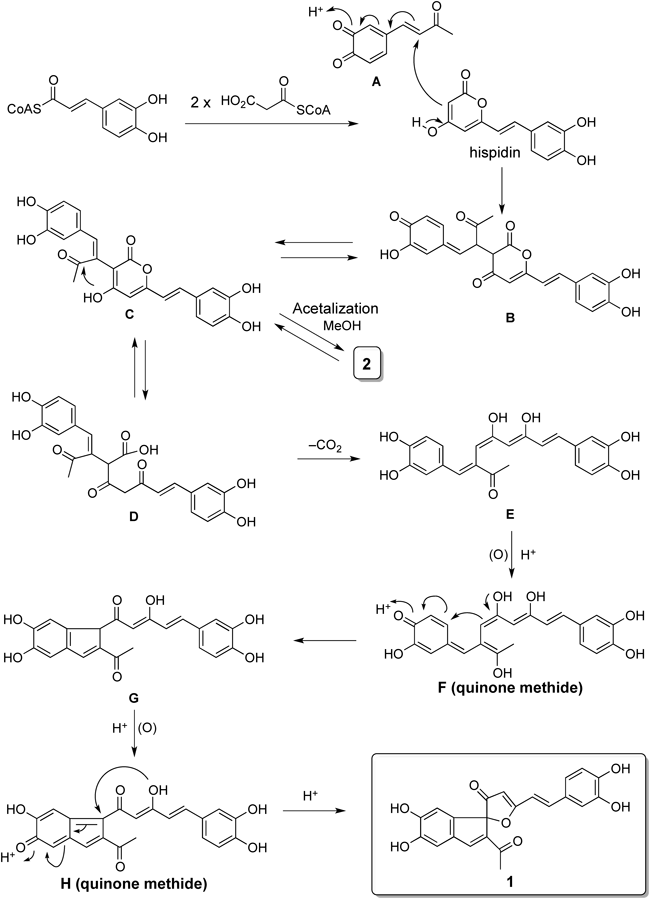
A plausible biosynthetic route for 1 is proposed as follows: caffeoyl-SCoA would bind to two malonyl-SCoAs to give the common biogenetic precursor, hispidin,15,16) which may then couple with A giving rise to B. In addition, the decarboxylation of D, which is in equilibrium with B, would lead to E. The E would undergo a sequential oxidation and intermolecular coupling reaction, giving rise to quinone methide H, which would then undergo a spiro ring fusion to produce 1 (Chart 1).
The effects of compounds 1–3 on the PC12 cells were evaluated according to previously reported methods.17) None of the compounds had any morphological effects on PC12 cells in the absence and/or presence of NGF at 30 µM, but all of the isolated compounds exhibited cytotoxicity against PC12 cells at 30 µM.


| Position | δH | δC | Position | δH | δC |
|---|---|---|---|---|---|
| 1 | 96.3 | 1′ | 194.6 | ||
| 2 | 202.5 | 2′ | 143.0 | ||
| 3 | 5.95 (1H, s) | 104.4 | 3′ | 7.92 (1H, s) | 150.2 |
| 4 | 187.5 | 4′ | 137.2 | ||
| 5 | 6.91 (1H, d, J=15.9 Hz) | 113.7 | 5′ | 7.02 (1H, s) | 113.2 |
| 6 | 7.46 (1H, d, J=15.9 Hz) | 142.5 | 6′ | 148.4 | |
| 7 | 128.3 | 7′ | 149.5 | ||
| 8 | 7.11 (1H, d, J=2.0 Hz) | 115.3 | 8′ | 6.61 (1H, s) | 110.6 |
| 9 | 147.0 | 9′ | 133.8 | ||
| 10 | 150.0 | CH3-1′ | 2.35 (3H, s) | 26.3 | |
| 11 | 6.79 (1H, d, J=8.1 Hz) | 116.6 | |||
| 12 | 7.01 (1H, dd, J=8.1, 2.0 Hz) | 123.4 |
In conclusion, we have isolated a new spiroindene pigment from the fruiting body of Phellinus ribis. Phelliribsin A (1) is an unprecedented spirocyclic compound. Moreover, compound 1 was found to have no neurotrophic activity but to have cytotoxic activity against PC12 cells.
IR and UV spectra were recorded on a JASCO FT-IR 410 infrared and Shimadzu UV-1650PC spectrophotometer, respectively. HR-FAB-MS was taken on a MStation JMS-700. The NMR experiments were performed on a Varian Unity 600 MHz NMR spectrometer. Chemical shifts are given as δ (ppm) and deuterated solvent peaks as references for 1H- and 13C-NMR spectra. Silica gel column chromatography (CC) was carried out on Wako C-300 and Merck Silica gel 60 (70–230 and 230–400 mesh). TLC was carried out with silica gel 60 F254 and PR-18 F254 plates. HPLC was performed on a JASCO PU-1580 pump equipped with a JASCO UV-1575 detector, and all peaks were detected at 254 nm.
Fungus MaterialThe fruiting bodies of P. ribis were collected in Jinan, Shandong Province, China, in October 2010. A voucher specimen (1807FB) was indentified by Prof. Lingchuan Xu at Shandong University of Traditional Chinese Medicine and was deposited at the Institute of Pharmacognosy, Tokushima Bunri University.
Extraction and IsolationThe fruiting bodies of P. ribis (1.5 kg) were powdered and extracted with MeOH (5 L) at room temperature for one month to give 17 g of crude extract, following by solvent removal. The MeOH extract (17 g) was chromatographed on a Si gel column (350 g) eluted with a step gradient of CH2Cl2 (A: 100%), CH2Cl2–EtOAc (B: 1 : 9, C: 1 : 1), EtOAc (D: 100%), EtOAc–MeOH (E: 1 : 9, F: 7 : 3, G: 1 : 1) to yield seven fractions (A–G). Fraction D (1.5 g) was first subjected to Shephadex LH-20 chromatography eluted with MeOH to give fractions 1–9 (Fr. 1: 114.9 mg, Fr. 2: 376.6 mg, Fr. 3: 216.4 mg, Fr. 4: 317.3 mg, Fr. 5: 123.9 mg, Fr. 6: 68.8 mg, Fr. 7: 54.5 mg, Fr. 8: 116.1 mg, Fr. 9: 80.3 mg). Fraction 5 was further separated by reverse-phase HPLC (Cosmosil 5C18-MSII) eluted with MeOH–H2O (1 : 1) to give fractions 10–16. Fraction 12 was purified by reverse-phase HPLC (Cosmosil 5C18-MSII) eluted with MeOH–H2O (45 : 55) to give 1 (2.3 mg).
Phelliribsin A (1) 1H-NMR (CD3OD) δ: 2.35 (3H, s), 5.95 (1H, s), 6.61 (1H, s), 6.79 (1H, d, J=8.1 Hz), 6.91 (1H, d, J=15.9 Hz), 7.01 (1H, dd, J=8.1, 2.0 Hz), 7.02 (1H, s), 7.11 (1H, d, J=2.0 Hz), 7.46 (1H, d, J=15.9 Hz), 7.92 (1H, s). 13C-NMR (CD3OD) δ: 26.3, 96.3, 104.4, 110.6, 113.2, 113.7, 115.3, 116.6, 123.4, 128.3, 133.8, 137.2, 142.5, 143.0, 147.0, 148.4, 149.5, 150.0, 150.2, 187.5, 194.6, 202.5. IR (ATR) cm−1: 3275, 1627, 1594, 1543. UV λmax (MeOH) nm (log ε): 264 (4.23), 378 (4.39). [α]D ±0 (c=0.014, MeOH). HR-FAB-MS m/z: 393.0988 [M+1]+ (Calcd for C22H17O7: 393.0974).
We thank Drs. Masami Tanaka and Yasuko Okamoto (TBU) for measuring the 600 MHz NMR and mass spectra. This work was supported in part by a Grant-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science and Technology of Japan (22590029, 23790036) and a Grant from MEXT-Senryaku for the Promotion and Mutual Aid Corporation for Private Schools of Japan.