2017 Volume 65 Issue 3 Pages 261-267
2017 Volume 65 Issue 3 Pages 261-267
Radioactive nuclides leak into the surrounding environment after nuclear power plant disasters, such as the Chernobyl accident and the Fukushima Daiichi Nuclear Power Plant disaster. Cesium-137 (137Cs) (t1/2=30.1 year), a water-soluble radionuclide with a long physical half-life, contaminates aquatic ecosystems and food products. In humans, 137Cs concentrates in muscle tissue and has a long biological half-life, indicating it may be harmful. myo-Inositol-hexakisphosphate (IP6) is a compound found in grain, beans, and oil seeds. IP6 has the ability to form insoluble complexes with metals, including lanthanum (La) and zinc (Zn). We hypothesized that La-IP6 and Zn-IP6 may promote the elimination of 137Cs from the body through the adsorption of La-IP6 and Zn-IP6 to 137Cs in the gastrointestinal tract. Therefore, in this study, we evaluated the adsorptive capacity of La-IP6 and Zn-IP6 complexes with 137Cs in vitro and in vivo. La-IP6 and Zn-IP6 complexes were stable in acidic solution (pH 1.2) at 37°C. In vitro binding assays indicated that La-IP6 and Zn-IP6 complexes adsorbed 137Cs, with the adsorption capacity of Zn-IP6 to 137Cs greater than that of La-IP6. To evaluate the usefulness of La-IP6 and Zn-IP6 in vivo, La-IP6 or Zn-IP6 was administrated to mice after intravenous injection of 137Cs. However, the biodistribution of 137Cs in the La-IP6 treated group and the Zn-IP6 treated group was nearly identical to the non-treated control group, indicating that La-IP6 and Zn-IP6 were not effective at promoting the elimination of 137Cs in vivo.
Radionuclides are released into the atmosphere and the ocean in considerable amounts as the result of nuclear power plant disasters, such as the accidents of the Chernobyl Nuclear Power Plant and the Fukushima Daiichi Nuclear Power Plant.1–4) Among released radionuclides, cesium-137 (137Cs), which is one of the most important nuclear fission elements, is considered harmful to human because it has a long physical half-life (t1/2=30.1 year) and can contaminate aquatic ecosystems and food products due to its high water-solubility.4) Indeed, an extreme minority of people displayed internal 137Cs levels of more than 50 Bq/kg in 2012 at Minamisoma Municipal General Hospital and Hirata Central Hospital located in 23 and 43 km from the Fukushima Daiichi Nuclear Power Plant, respectively. This exposure was likely due to the intake of homegrown vegetables and wild mushrooms without proper radiation inspection.5)
137Cs decays to barium-137m (137mBa) with the emission of β-particles (0.51 MeV), and 137mBa (t1/2=2.55 m) decays through an isomeric transition to the stable isotope, 137Ba, with emission of gamma ray (0.66 MeV). Cesium is one of the alkaline metals in the periodic table and has chemical properties similar to potassium. 137Cs is immediately absorbed after inhalation or oral exposure and distributed throughout the whole body, with a particularly high accumulation in muscle tissue. The biological half-life of 137Cs is long and is approximately 100 d in male human adults.6–9) These properties, coupled with internal exposure of 137Cs, could be very harmful to humans. When exposure to 137Cs occurs in humans, decorporation agents are useful for decreasing the absorbed radiation dose. Decorporation agents bind to 137Cs and inhibit its absorption into the bloodstream from the gastrointestinal tract, and enhance its elimination into feces. These agents should be especially effective against 137Cs since 137Cs undergoes enterohepatic circulation. Indeed, insoluble ferric(III) hexacyanoferrate (Fe4III[FeII(CN)6]3)·xH2O (Radiogardase®, insoluble Prussian blue, PB) (Fig. 1A), a compound that promotes the elimination of 137Cs from the body, has been used successfully for the treatment of radiocesium poisoning10) and has been approved in Japan and by the U.S. Food and Drug Administration (FDA) and European Medicines Agency (EMA). PB was used to treat to 137Cs exposure in people after the Chernobyl nuclear reactor accident and the Goiania accident in Brazil,11) and was shown to reduce the biological half-life of 137Cs in humans.12) PB binds to 137Cs in the gastrointestinal tract, inhibiting its reabsorption into the blood and, consequently, enhancing its excretion into feces.13) However, potassium may be also taken up by PB because of the similarity of potassium to cesium. Mild cases of hypokalemia resulting from PB treatment has been reported,14) thus serum potassium levels should be monitored whenever PB is administrated.
Meanwhile, myo-inositol-hexakisphosphate (phytic acid: IP6) (Fig. 1B), found in cereals, nuts, legumes, spores, needles, pollen, and oil seeds at 1–5% weight, possesses a high chelation potential with many kinds of metal cations due to its specific structure.15,16) It is known that one IP6 molecule can coordinate up to six divalent metal cations, and the metal cations could form cross-linking with at least two IP6 molecules depending on the redox state.16) For example, it has been reported that an excessive IP6 intake can cause dwarfism in humans due to lack of zinc.17) This zinc deficiency is caused by the formation of an insoluble complex between IP6 and zinc in gastrointestinal tract, thus inhibiting zinc absorption. Namely, IP6 forms insoluble complex with zinc ion (Zn2+). Moreover, lantern ion (La3+) selected from some kinds of metals by our preliminary study formed insoluble complex with IP6. Although the Cs ion is monovalent, its ion radius is large. Thus, the chelation potential of the Cs ion with IP6 should be higher than that of Na ion or K ion due to the cross-linking chelate. Therefore, we hypothesized that insoluble La-IP6 and Zn-IP6 complexes could potentially chelate with Cs in the gastrointestinal tract to inhibit absorption and enhance excretion of radioactive Cs into feces. In this study, we prepared La-IP6 and Zn-IP6 complexes and evaluated their in vitro and in vivo potential for 137Cs decorporation.
The relationship of M/IP6 (M=La or Zn) mole ratios between the reaction mixture and products are shown in Fig. 2. When the mole ratios of La/IP6 and Zn/IP6 in the reaction mixtures were 2, the mole ratios of La/IP6 and Zn/IP6 in the produced La-IP6 and Zn-IP6 complexes were approximately 2. When the mole ratios of La/IP6 and Zn/IP6 in the reaction mixtures were >3, the mole ratio of Zn/IP6 in the produced Zn-IP6 complex was approximately 3; however, the mole ratio of La/IP6 in the produced La-IP6 complex remained 2. The coordination forms of the La-IP6 and Zn-IP6 complexes were suggested from these results. When the mole ratios of M/IP6 in produced M-IP6 complexes were 2, the coordination numbers of Zn and La were 6. Since the IP6 contains 6 phosphate groups, the coordination per phosphate group was considered 2. When the mole ratios of Zn/IP6 in the produced Zn-IP6 complex were 3, the coordination numbers of Zn decrease to 4.
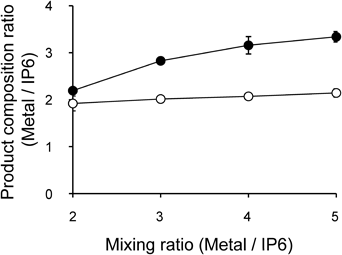
Data of La/IP6 ratios (open circles) and Zn/IP6 ratios (closed circles) are expressed as the mean±standard deviation (S.D.) of three experiments.
The stability results of M-IP6 in acidic solution (pH 1.2) are shown in Fig. 3. After incubation at 37°C in acidic solution for 6 h, over 88% of the La-IP6 and Zn-IP6 complexes remained intact. The ratio of soluble lanthanum (La) or zinc (Zn) from the decomposition of these complexes was detected by inductively coupled plasma atomic emission spectrometry (ICP-AES), and was comparable to that of soluble phosphorus (P) derived from IP6. These results indicate that the M/IP6 mole ratios in the M-IP6 complexes were maintained. We hypothesized that the observed insolubility of La-IP6 and Zn-IP6 complexes was based on the electron configurations of La3+ and Zn2+. The electron configurations of La3+ and Zn2+ are [Xe] and [Ar] 3d10, respectively. La3+ has the same electron configuration of Xe as a noble gas. The 3d orbit, the outermost electronic shell, is full in the electron configuration of Zn2+. Thus, La3+ and Zn2+ do not need to form fixed coordination geometry as predicted by the d-orbital crystal field theory. It makes the direction of the coordination flexible, and cross-linked chelates derived from the phosphate groups of IP6 theoretically can extend to intermolecular coordination infinitively. Therefore, the complexes could be insoluble by the formation of a polymer. Meanwhile, after incubation at 37°C in neutral solution (pH 6.8) for 6 h, 94.6 and 97.5% of La-IP6 and Zn-IP6 complexes remained intact, respectively. In the case of using M-IP6 complexes as agents for promoting the excretion of 137Cs from body, these results indicate that oral administration of M-IP6 may proceed through the stomach and intestine without decomposition.
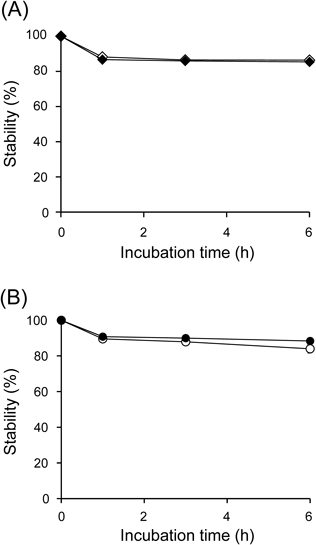
Data calculated from the concentration of P (open diamonds and open circles), La (closed diamonds), and Zn (closed circles) in insoluble M-IP6 are expressed as the mean±S.D. of four samples.
Adsorption capacity of M-IP6 or PB with cesium (Cs) was evaluated by the Langmuir model, defined as follows:
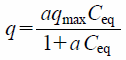 | (1) |
The binding of cesium to M-IP6 and PB increased linearly at low metal concentrations and showed a plateau at high concentrations (Fig. 4), although values and slopes are different, indicating saturation of the adsorption sites. These data match with the Langmuir model and indicate that the adsorption occurs on a uniform surface with finite identical sites involving a monolayer adsorption of the adsorbate. The maximum sorption capacity of Zn-IP6, La-IP6, and PB was estimated to be 203.0, 113.9, and 17.8 mg Cs/g, respectively. In previous reports, the maximum sorption capacity of PB has been reported to be 16.5 mg Cs/g at pH 8.6,6) 158.5 mg Cs/g,18) 12.5 mg Cs/g at pH 7.7 in seawater,19) and 131.6 mg Cs/g,20) depending on the study. While these values from previous study are different, the results of this study indicate that Zn-IP6 and La-IP6 possess much higher adsorption potency compared to PB, which has been approved as a decorporation agent for radiocesium.
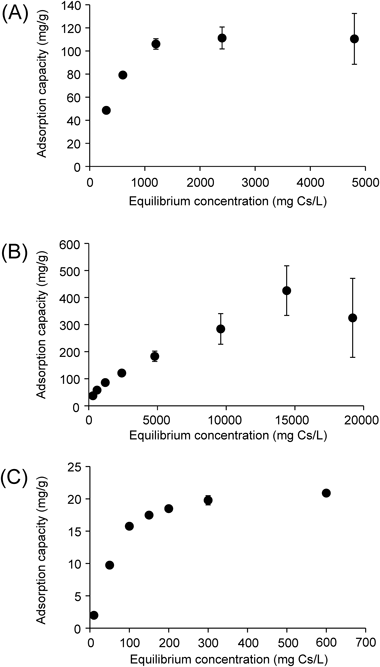
Adsorption capacity of (A) La-IP6, (B) Zn-IP6, and (C) PB to cesium. Data are expressed as the mean±S.D. of three samples.
Zn-IP6 and La-IP6 per one IP6 molecule unit used in the experiments could be 2Zn-IP6·4Na·4Cl·8H2O and 2La-IP6·7H2O, respectively, which were determined by ICP-AES and elemental analysis. Thus, 1 g of each complex per one IP6 molecule unit is calculated as 0.86 and 0.95 mmol, respectively. As above-mentioned, the maximum sorption capacity of Zn-IP6 and La-IP6 was estimated to be 203.0 and 113.9 mg Cs/g, namely 1.53 and 0.86 mmol (Cs)/g (M-IP6), respectively. The maximum sorption capacity of Zn-IP6 and La-IP6 with Cs was calculated as 1.78 Cs/Zn-IP6 and 0.91 Cs/La-IP6, respectively.
Biodistribution ExperimentsTo evaluate the potential of M-IP6 (M=La or Zn) to enhance the elimination of 137Cs from the body in vivo, La-IP6 or Zn-IP6 was repetitively administrated to mice after intravenous injection of 137Cs. The biodistribution of radioactivity in mice at 48 h post-injection is shown in Table 1. It has been reported that 137Cs is recycled via the enterohepatic circulation.13) Thus, we hypothesized that for M-IP6 to work effectively as decorporation agents, M-IP6 may inhibit the reabsorption of 137Cs in the intestine and enhance its excretion into feces, even if 137Cs was intravenously injected. It is assumed that decorporation agents should be effective for enhancing the excretion of 137Cs even after absorption, such as in the case of the existence of 137Cs in the body. Contrary to our expectation, the biodistribution pattern of radioactivity in M-IP6 administrated groups was nearly identical to a non-treatment control group. These results indicated that 137Cs excretion was not enhanced by oral administration of M-IP6 after 137Cs was intravenously injected. The biodistribution pattern of radioactivity in mice after injection of 137Cs and repeated administration of PB, instead of M-IP6, was also evaluated. In this case, the radioactivity of all tissues prominently decreased with PB treatment, indicating that PB enhanced the excretion of 137Cs radioactivity from the body (Table 1).
| Tissue | Control | La-IP6 | Zn-IP6 | PB |
|---|---|---|---|---|
| Blood | 0.31 (0.02) | 0.33 (0.01) | 0.31 (0.02) | 0.16 (0.01)*** |
| Liver | 0.68 (0.07) | 0.88 (0.04)* | 0.82 (0.17) | 0.23 (0.03)*** |
| Kidney | 1.39 (0.13) | 1.78 (0.06)** | 1.44 (0.18) | 0.44 (0.05)*** |
| Small-intestine† | 2.43 (0.25) | 2.58 (0.31) | 2.48 (0.49) | 0.70 (0.08)*** |
| Large-intestine† | 0.85 (0.09) | 1.11 (0.13) | 1.07 (0.14) | 0.56 (0.22) |
| Spleen | 1.14 (0.13) | 1.38 (0.13) | 1.27 (0.19) | 0.35 (0.03)*** |
| Pancreas | 1.45 (0.16) | 1.93 (0.09)* | 1.75 (0.29) | 0.49 (0.10)*** |
| Lung | 0.81 (0.12) | 0.93 (0.23) | 0.76 (0.12) | 0.27 (0.02)*** |
| Heart | 1.27 (0.13) | 1.41 (0.04) | 1.28 (0.21) | 0.34 (0.08)*** |
| Stomach† | 0.37 (0.05) | 0.48 (0.03) | 0.46 (0.04) | 0.20 (0.07)** |
| Bone (Femur) | 0.84 (0.07) | 0.69 (0.11) | 0.80 (0.13) | 0.29 (0.07)*** |
| Muscle | 2.34 (0.26) | 2.98 (0.07)* | 2.87 (0.31) | 1.61 (0.20)** |
| Brain | 0.70 (0.02) | 0.75 (0.01) | 0.68 (0.05) | 0.39 (0.01)*** |
| Rest† | 19.93 (1.76) | 24.78 (0.31)** | 22.07 (2.87) | 10.62 (0.65)*** |
Data are expressed as % injected dose per gram tissue. Each value represents the mean (S.D.) for three or four animals. Rest means whole body without stomach, small-intestine, and large-intestine. †Data are expressed as % injected dose. * p<0.05, ** p<0.01, *** p<0.001 vs. control.
To further evaluate the possibility of M-IP6 as decorporation agents, 137Cs was orally administrated to mice just after oral administration of M-IP6. The biodistribution of radioactivity at 24 h after oral administration of 137Cs is shown in Table 2 and was the same among a non-treatment control group and M-IP6 treated groups. These results indicate that M-IP6 are not an effective decorporation agent for 137Cs. On the other hand, there was little radioactivity remaining in tissues with PB treatment (Table 2), indicating that PB prominently inhibited the absorption of 137Cs.
| Tissue | Control | La-IP6 | Zn-IP6 | PB |
|---|---|---|---|---|
| Blood | 0.39 (0.04) | 0.45 (0.07) | 0.36 (0.12) | 0.01 (0.01)*** |
| Liver | 1.51 (0.25) | 1.14 (0.05) | 1.58 (0.34) | 0.04 (0.01)*** |
| Kidney | 2.39 (0.52) | 2.22 (0.23) | 2.53 (0.58) | 0.08 (0.02)*** |
| Small-intestine† | 4.21 (0.69) | 4.76 (0.33) | 4.25 (1.09) | 0.11 (0.02)*** |
| Large-intestine† | 1.69 (0.26) | 2.14 (0.17) | 1.66 (0.49) | 0.12 (0.08)*** |
| Spleen | 2.22 (0.50) | 2.12 (0.17) | 2.13 (0.53) | 0.05 (0.02)*** |
| Pancreas | 3.07 (0.70) | 3.53 (0.28) | 3.14 (0.71) | 0.09 (0.03)*** |
| Lung | 1.21 (0.21) | 1.52 (0.07) | 1.46 (0.42) | 0.03 (0.01) |
| Heart | 2.10 (0.47) | 2.24 (0.67) | 1.90 (0.51) | 0.06 (0.02)** |
| Stomach† | 0.68 (0.11) | 1.15 (0.32)* | 0.80 (0.17) | 0.03 (0.01)** |
| Bone (Femur) | 1.19 (0.12) | 1.15 (0.18) | 1.12 (0.30) | 0.05 (0.01)*** |
| Muscle | 2.57 (0.48) | 2.68 (0.37) | 2.39 (0.76) | 0.06 (0.00)*** |
| Brain | 0.60 (0.04) | 0.58 (0.03) | 0.51 (0.16) | 0.01 (0.00)*** |
| Rest† | 31.30 (5.38) | 26.80 (4.34) | 23.78 (6.56) | 0.71 (0.15)*** |
Data are expressed as % injected dose per gram tissue. Each value represents the mean (S.D.) for three animals. Rest means whole body without stomach, small-intestine, and large-intestine. †Data are expressed as % injected dose. * p<0.05, ** p<0.01, *** p<0.001 vs. control.
In in vitro experiments, the binding affinity of M-IP6 for 137Cs was higher than that of PB, but, PB was much better than M-IP6 for decorporation of 137Cs in vivo. However, it was not entirely clear whether M-IP6 could adsorb 137Cs at all in vivo. Thus, we performed biodistribution experiments after oral administration of 137Cs-M-IP6 complexes, which were bound together in advance, to evaluate their stability in vivo. Table 3 lists the biodistribution pattern of radioactivity at 1 and 24 h after oral administration of 137Cs-M-IP6 or 137Cs-PB. In the case of 137Cs-PB, the existence of radioactivity was nominal in any organs at 24 h. This indicates that 137Cs was not released from the 137Cs-PB complex, and it was not absorbed into the blood. In contrast, in the case of 137Cs-M-IP6, the radioactivity was distributed to the whole body, suggesting that the 137Cs-M-IP6 complexes decomposed and 137Cs was absorbed into the blood.
| Tissue | 137Cs-PB | 137Cs-La-IP6 | 137Cs-Zn-IP6 | |||
|---|---|---|---|---|---|---|
| 1 h | 24 h | 1 h | 24 h | 1 h | 24 h | |
| Blood | 0.00 (0.00) | 0.02 (0.00) | 0.30 (0.05) | 0.50 (0.01) | 0.29 (0.02) | 0.52 (0.08) |
| Liver | 0.04 (0.01) | 0.06 (0.01) | 4.14 (0.59) | 1.90 (0.08) | 3.80 (0.78) | 1.97 (0.43) |
| Kidney | 0.12 (0.01) | 0.11 (0.01) | 11.13 (2.23) | 3.26 (0.18) | 13.09 (2.20) | 3.42 (0.81) |
| Small-intestine† | 47.24 (2.09) | 0.16 (0.01) | 28.23 (5.78) | 4.71 (0.43) | 29.57 (3.45) | 5.70 (1.15) |
| Large-intestine† | 0.10 (0.10) | 0.07 (0.00) | 4.03 (1.19) | 2.28 (0.38) | 3.79 (0.24) | 2.39 (0.50) |
| Spleen | 0.01 (0.01) | 0.12 (0.01) | 2.01 (0.41) | 2.67 (0.05) | 2.18 (0.31) | 2.88 (0.39) |
| Pancreas | 0.01 (0.01) | 0.16 (0.01) | 3.58 (1.08) | 4.18 (0.50) | 3.61 (0.49) | 5.01 (1.09) |
| Lung | 0.01 (0.00) | 0.06 (0.02) | 3.73 (0.87) | 1.55 (0.30) | 2.99 (0.15) | 1.77 (0.16) |
| Heart | 0.05 (0.02) | 0.14 (0.04) | 6.29 (1.42) | 2.63 (0.45) | 6.74 (1/47) | 2.85 (0.89) |
| Stomach† | 49.84 (2.04) | 0.04 (0.01) | 18.14 (14.43) | 1.02 (0.14) | 13.53 (8.57) | 1.00 (0.28) |
| Bone (Femur) | 0.05 (0.08) | 0.13 (0.02) | 0.95 (0.45) | 1.53 (0.06) | 1.40 (0.15) | 1.44 (0.39) |
| Muscle | 0.00 (0.00) | 0.15 (0.03) | 0.94 (0.35) | 3.31 (0.38) | 0.99 (0.16) | 3.55 (0.90) |
| Brain | 0.00 (0.00) | 0.03 (0.00) | 0.09 (0.05) | 0.72 (0.08) | 0.10 (0.03) | 0.72 (0.18) |
| Rest† | 0.33 (0.06) | 1.06 (0.13) | 36.01 (8.62) | 32.97 (2.73) | 31.69 (5.35) | 35.84 (5.59) |
Data are expressed as % injected dose per gram tissue. Each value represents the mean (S.D.) for three animals. Rest means whole body without stomach, small-intestine, and large-intestine. †Data are expressed as % injected dose.
The effectiveness of M-IP6 for enhancing the elimination of 137Cs was expected since M-IP6 highly bound 137Cs in in vitro experiments; however, the elimination of 137Cs was not enhanced by the oral administration of M-IP6 in biodistribution experiments. The ineffectiveness of M-IP6 may be due to a low binding potential between 137Cs and M-IP6 in vivo. The existence of other metals in in vivo situations may be one of factors; M-IP6 may preferentially bind multivalent metal cations, which could have a higher binding potential with M-IP6. We hypothesized that various other metals displaced 137Cs in the 137Cs-M-IP6 complexes. Thus, we investigated the effects of other metals on the in vitro adsorption of 137Cs to M-IP6 or PB.
Effects of Cations (Na+, K+, or Ca2+) on the in Vitro Adsorption of 137Cs to M-IP6 or PBFigure 5 shows the binding of cesium to M-IP6 (M=Zn or La) or PB in the presence of cations (Na+, K+, or Ca2+). The presence of high concentrations of cations tended to decrease the binding of cesium to M-IP6, with Ca2+ having the largest effect. These results support the hypothesis that the existence of other cations in the gastrointestinal tract may inhibit the adsorption of M-IP6 to 137Cs. Meanwhile, the presence of Na+, K+, or Ca2+ minimally affected the binding of cesium to PB, suggesting that the binding affinity of PB for cesium is more specific.
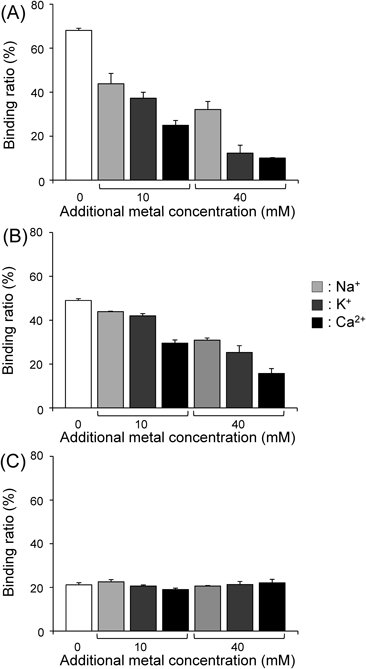
Zero additional metal concentration (white columns) means using HEPES buffer containing 10 mM Na+. Data are expressed as the mean±S.D. of three samples.
The crystal structure of PB is a cubic lattice with Fe(II) and Fe(III) occupying the corners and the cyano group at the sides. The Fe(II) and Fe(III) bind to the carbon and nitrogen atom in the cyano group, respectively. Additionally, 14–16 water molecules are coordinated to PB.21) The adsorption mechanism of PB to cesium still has not been completely clear. It was proposed that Cs+ adsorption of PB is caused by the monovalent cesium ion exchange with hydrogen ion (H+) or from water bound hydronium ion (H3O+) in the PB lattice.22) Recently, it has been proposed that lattice defect sites in PB are important for Cs+ adsorption ability because Cs+ adsorption of PB could be caused via the hydrophilic lattice defect sites with proton-exchange from coordination water, which exist in the lattice defect sites.23) It was also reported that the adsorption ability of PB for alkali metal ions increased in the order of Cs+ ≫ K+ ≥Na+.23) The high ion selectivity of PB to Cs+ should be derived from the similar size of Cs+, which could be easy to fit, to the lattice defect site.24)
These results indicated that the uselessness of M-IP6 for decorporation of 137Cs in vivo was caused by the lower selectivity to Cs+.
M-IP6 (M=Zn or La) possess much higher adsorption potency against 137Cs compared to PB in in vitro experiments. However, the binding potential between 137Cs and M-IP6 was low in vivo, and M-IP6 did not effectively work as decorporation agents to 137Cs. While M-IP6 cannot be used as decorporation agents of 137Cs in the human body, it may be useful as absorbent for the decontamination of 137Cs in the environment.
137CsCl was purchased from Eckert & Ziegler Isotope Products Inc. (Valencia, CA, U.S.A.). ICP-AES was obtained with iCAP-6300 (Thermo Fisher Scientific, Waltham, MA, U.S.A.). Elemental analysis was carried out on LECO CHN-900 (LECO Corporation, St. Joseph, MI, U.S.A.). IP6 was purchased from Sigma-Aldrich (St. Louis, MO, U.S.A.) as phytic acid sodium salt hydrate, IP6·6Na+·6H2O. Prussian blue was purchased from Sigma-Aldrich. Other reagents were of reagent grade and used as received.
Preparation of La-IP6 ComplexesIP6 (906 mg, 1.0 mmol) was dissolved in 10 mL of water, and 10 mL of La2O3 solution (100 mM). The La2O3 solution was prepared from the dissolution of 1.6 g of La2O3 (5.0 mmol) in 50 mL of water containing 5 mL (added dropwise) of concentrated HCl aqueous solution; this solution was added to the IP6 solution while stirring. After centrifugation at 10000×g for 5 min at room temperature, the supernatant was removed and water was added to the precipitation. After the suspension was vortexed, it was centrifuged at 10000×g for 5 min at room temperature. The supernatant was removed by filtration to obtain La-IP6 (La : IP6=2 : 1) (0.44 g) as a white powder.
La-IP6 (La : IP6=3 : 1), La-IP6 (La : IP6=4 : 1), and La-IP6 (La : IP6=5 : 1) were prepared by the above-mentioned procedure using 15, 20, and 25 mL of the La2O3 aqueous solution (100 mM), respectively. The yields of La-IP6 (La : IP6=3 : 1), La-IP6 (La : IP6=4 : 1), and La-IP6 (La : IP6=5 : 1) were 0.39, 0.25, and 0.08 g, respectively.
La-IP6 samples (10 mg) were dissolved in 50 µL of concentrated nitric acid, and 10 µL of the La-IP6 solution was diluted with 10 mL of 0.1 M nitric acid. The concentrations of La, Na, and P in each sample were determined using ICP-AES. The mole ratios of La/IP6 in the produced La-IP6 samples were calculated.
Preparation of Zn-IP6 ComplexesIP6 (906 mg, 1.0 mmol) was dissolved in 10 mL of water, and 10 mL of 0.4 M NaOH aqueous solution was added to IP6 solution at stirring. ZnCl2 (272 mg, 2.0 mmol) was dissolved in 10 mL of water, and the solution was added the IP6 solution. After centrifugation at 10000×g for 5 min at room temperature, the supernatant was removed. Water was added to the precipitation. After the suspension was vortexed, it was centrifuged at 10000×g for 5 min at room temperature. The supernatant was removed by filtration to obtain Zn-IP6 (Zn : IP6=2 : 1) (353 mg) as a white powder.
Zn-IP6 (Zn : IP6=3 : 1), Zn-IP6 (Zn : IP6=4 : 1), and Zn-IP6 (Zn : IP6=5 : 1) were prepared by the above-mentioned procedure using 15, 20, and 25 mL of the ZnCl2 aqueous solution (200 mM), respectively. The yields of Zn-IP6 (Zn : IP6=3 : 1), Zn-IP6 (Zn : IP6=4 : 1), and Zn-IP6 (Zn : IP6=5 : 1) were 1.07 g, 775, and 815 mg, respectively.
Each Zn-IP6 samples (10 mg) were dissolved in 100 µL of concentrated nitric acid, and 10 µL of the Zn-IP6 solution was diluted with 10.0 mL of 0.1 M nitric acid. The concentrations of Zn, Na, and P in each sample were determined using ICP-AES. The mole ratios of Zn/IP6 in the produced Zn-IP6 samples were calculated.
In Vitro Stability of M-IP6 Complexes (M=Zn or La)To evaluate the stability of La-IP6 and Zn-IP6 in acidic and neutral conditions, 10 mg of La-IP6 (La : IP6=2 : 1) or Zn-IP6 (Zn : IP6=2 : 1) was suspended in 1.0 mL of the first test solution (artificial gastric juice, pH 1.2) or the second test solution (artificial intestinal juice, pH 6.8) defined in the Japanese Pharmacopoeia, and was incubated for 6 h at 37°C. After incubation of 1, 3, and 6 h, 100 µL of each sample was taken and centrifuged at 10000×g for 3 min at room temperature. The supernatant (50 µL) was diluted using 0.1 M nitric acid aqueous solution, and the concentrations of P, La, and Zn in the supernatant were determined using ICP-AES. Control group underwent the same procedure with the dissolution of 10 mg of M-IP6 in 50 µL of concentrated nitric acid and dilution with 950 µL of the Japanese Pharmacopoeia collapse examination first solution (pH 1.2) or the second solution (pH 6.8). The rate of insoluble M-IP6 (%) was determined using the following equation: Insoluble M-IP6 (%)=(1−[concentration (ppm) of supernatant of each sample]/[concentration (ppm) of of supernatant of control])×100.
In Vitro Adsorption of 137Cs to M-IP6 (M=Zn or La) or PBLa-IP6 (La : IP6=2 : 1), Zn-IP6 (Zn : IP6=2 : 1), or PB samples (10 mg/mL) containing various concentration of Cs and 2 kBq/mL of 137Cs as a final concentration, in 20 mM N-(2-hydroxyethyl)piperazine-N′-2-ethanesulfonic acid (HEPES) buffer (pH 7.4) containing 10 mM Na, were prepared by mixing radioactive cesium and non-radioactive cesium. After shaking at 1000 rpm and 37°C for 1 h, the samples were centrifuged at 10000×g at room temperature for 10 min. The radioactivity of the supernatant was measured using an auto-well gamma counter (ARC-380; Hitachi Aloka Medical, Ltd., Tokyo, Japan), and the counts were corrected for background radiation. Control experiments were performed using the same procedure but without M-IP6 or PB. The binding ratios were determined as follows: Binding ratio to M-IP6 or PB (%)=(1−[radioactivity of supernatant of each sample]/[radioactivity of supernatant in the respective control])×100.
Biodistribution Experiments after Intravenous Injection of 137Cs and M-IP6 (M=Zn or La) or PB TreatmentExperiments with animals were conducted in strict accordance with the Guidelines for the Care and Use of Laboratory Animals of Kanazawa University. The animal experimental protocols used were approved by the Committee on Animal Experimentation of Kanazawa University (Permit Number: AP-143039). The animals were housed with free access to food and water at 23°C with a 12-h alternating light/dark schedule. Groups of three or four 6-week-old male ddY mice (27–32 g, Japan SLC, Inc., Hamamatsu, Japan) were intravenously administered 100 µL of saline solution of 137CsCl (37 kBq). One hundred milligrams of La-IP6 (La : IP6=2 : 1), Zn-IP6 (Zn : IP6=2 : 1), or PB suspension in 5% glucose aqueous solution was orally administrated at just after 12, 24, and 36 h after the injection of the 137CsCl solution. In the control group, 5% glucose aqueous solution was orally administrated instead of the M-IP6 suspension. Mice were sacrificed 48 h post-injection. Tissues of interest were removed and weighed, and radioactivity was determined with an auto well gamma counter, and counts were corrected for background radiation.
Biodistribution Experiments after Orally Administration of 137Cs with M-IP6 (M=Zn or La) or PB PretreatmentLa-IP6 (La : IP6=2 : 1), Zn-IP6 (Zn : IP6=2 : 1), or PB suspension in 5% glucose aqueous solution (100 mg) was orally administrated to 6-week-old male ddY mice. In the control group, 5% glucose aqueous solution was orally administrated instead of the M-IP6 or PB suspension. Then, just after administration of M-IP6 suspension, a saline solution of 137CsCl (37 kBq/100 µL) was orally administrated. Mice were sacrificed 24 h post-administration. Tissues of interest were removed and weighed, and radioactivity was determined.
Biodistribution Experiments of 137Cs-M-IP6 (M=Zn or La) and 137Cs-PBTo evaluate the properties of 137Cs-M-IP6, 137Cs-M-IP6, and 137Cs-PB were prepared before administration. Namely, 30 mg of La-IP6 (La : IP6=2 : 1), Zn-IP6 (Zn : IP6=2 : 1), or PB was suspended in 500 µL of water and 137CsCl (37 kBq) was added to the suspension. After the suspension was shaken at 37°C for 10 min, the suspension was centrifuged at 10000×g for 5 min, and the supernatant was removed. The precipitation was resuspended in 500 µL of 5% glucose aqueous solution, and the suspension as 137Cs-La-IP6, 137Cs-Zn-IP6, or 137Cs-PB was orally administrated to 6-week-old male ddY mice. Mice were sacrificed 1 and 24 h post-injection. Tissues of interest were removed and weighed, and radioactivity was determined.
Effects of Cations (Na+, K+, or Ca2+) on the in Vitro Adsorption of 137Cs to M-IP6 (M=Zn or La) or PBCs solutions (600 ppm at final concentration) containing 2 kBq/mL of 137Cs in 20 mM HEPES buffer (pH 7.4) containing 10 mM Na+, 20 mM Na+, 50 mM Na+, 10 mM Na++10 mM K+, 10 mM Na++40 mM K+, 10 mM Na++10 mM Ca2+, and 10 mM Na++40 mM Ca2+ were prepared by dissolution of NaCl, KCl, or CaCl2. M-IP6 or PB samples (10 mg) were suspended in 1 mL of each Cs solution. After shaking the suspensions at 1000 rpm and 37°C for 1 h, the binding ratio of each sample to M-IP6 or PB was determined using the methods described above.
Statistical EvaluationA one-way ANOVA followed by Dunnett’s post hoc tests compared with the control group were used for the biodistribution experiments of 137Cs. Results were considered statistically significant at p<0.05.
The authors declare no conflict of interest.