2020 Volume 68 Issue 8 Pages 791-796
2020 Volume 68 Issue 8 Pages 791-796
Because of the complexity of nanomedicines, analysis of their morphology and size has attracted considerable attention both from researchers and regulatory agencies. The atomic force microscope (AFM) has emerged as a powerful tool because it can provide detailed morphological characteristics of nanoparticles both in the air and in aqueous medium. However, to our knowledge, AFM methods for nanomedicines have yet to be standardized or be listed in any pharmacopeias. To assess the applicability of standardization of AFM, in this study, we aimed to identify robust conditions for assessing the morphology and size of nanoparticles based on a polystyrene nanoparticle certified reference material standard. The spring constant of the cantilever did not affect the size of the nanoparticles but needed to be optimized depending on the measurement conditions. The size analysis method of the obtained images affected the results of the analyzed size values. The results analyzed by cross-sectional line profiling were independent of the measurement conditions and gave similar results to those from dynamic light scattering. It was indicated that approximately 100 particles are required for a representative measurement. Under the optimized conditions, there were no significant inter-instrument differences in the analyzed size values of polystyrene nanoparticles both in air and under aqueous conditions.
Recently, nanomedicines have drawn considerable attention as new tools for delivery of various chemical and biological entities.1–3) To promote rapid development of nanotechnology drugs, it is important to develop analytical methods for their quality control.4) Owing to their tiny size (usually less than 1000 nm), new characterization methods should be established in addition to those for conventional drug products because the size and shape of nanomedicines are important factors that affect their behaviors in the body.5,6) Because nanoparticles are complex in terms of size, structure, and surface chemistries, the characterization of the materials with analytical methods based on different measurement principles is recommended.7) In particular, standardization of analytical methods for determining morphology and size of nanomedicines has drawn interest from both scientific and regulatory perspectives.8)
Dynamic light scattering (DLS) is a technique for size measurement of nanoparticles.9) In DLS, the average hydrodynamic diameter of nanoparticles in aqueous solution is measured on the basis of the light intensity scattered from the particles. The major advantages of DLS are its experimental simplicity and its applicability to nanoparticles suspended in aqueous solution. However, this method cannot be used to measure the morphology of particles or particles in an aggregation state. Aggregation or agglomeration might affect the quality, safety, and efficacy of drug products, and should be considered when designing a stability program.10) Moreover, shape and agglomeration of nanoparticles are known to adversely affect the biological action of nanoparticles.11) Therefore, there is a need for imaging techniques that can serve as a complementary method to DLS for characterizing the size of nanoparticles. Electron microscopy is often used to observe the morphology of nanoparticles; however, samples for electron microscope studies require treatments, such as freezing and the use of high vacuum.12) Conversely, atomic force microscope (AFM) offers an imaging technique suitable for various sample types, including soft biological samples and stiff artificial materials both in aqueous media and in air.13–15) There have been reports of AFM applied to observe nanomedicines.8,16–18)
To use AFM as a technique for characterizing nanomedicines for quality control, robust analytical methods should be established. However, to our knowledge, there have been no such reports, which have comprehensively studied the analytical conditions for measuring the size and images of nanoparticles by the use of AFM, including validation of instrument-dependent repeatability nor any listings in pharmacopoeia. Therefore, in this study, we aimed to elucidate the factors that might affect analysis of the morphology and size of nanoparticles, both in air and in aqueous media and to assess the applicability of standardization of AFM as an analytical methodology of quality control of nanomedicines.
A stock suspension of polystyrene nanoparticles (certified reference material (CRM) 5701-a, with a certified mean diameter and uncertainty of 118.5 ± 1.8 nm as measured by DLS, the National Metrology Institute of Japan) was purchased from FUJIFILM Wako Pure Chemical Corporation (Osaka, Japan). The hydrodynamic diameters and polydispersity index (PDI) values of the polystyrene nanoparticles were measured at 25°C by DLS combined with cumulant analysis with a Zetasizer Nano-ZS instrument equipped with Zetasizer Software v.6.01 (Malvern Instruments, Malvern, U.K.). A mean value and standard deviation (S.D.) were obtained from three measurements. It was confirmed that the PDI of polystyrene nanoparticles was less than 0.2, indicating monodispersity.
AFM ApparatusAFM apparatuses used in this study were JPK Nanowizard Ultra Speed microscope, equipped with the JPK Data Processing Software v.6.0 (JPK Instruments AG, Berlin, Germany), Shimadzu SPM-9700 microscope equipped with the SPM software v.4.00 (Shimadzu Co., Kyoto, Japan), Hitachi AFM5000 and AFM5400L microscopes (Hitachi High-Technologies Co., Tokyo, Japan). Both the Hitachi microscopes were equipped with the software Spisel32 v.6.03B1.
CantileverFour kind of cantilevers were used for measurement. Biolever mini (BL-AC40TS, Nominal Spring constant 0.09 N/m) and a OMCL-AC240TS (Nominal Spring constant 2 N/m) were obtained from Olympus Co. (Tokyo, Japan). Alternative cantilevers SI-DF3P2 (Nominal Spring constant: 2 N/m) and SI-DF40P2 (Nominal Spring constant: 26 N/m) were used for the measurements with the Hitachi AFM5000 and AFM5400L microscopes.
Coating of the SubstrateA slide glass (S-2411, 76 × 26 mm, thickness: 0.9–1.2 mm) and cover glass (NEO micro cover glass, 24 × 32 mm, thickness: 0.12–0.17 mm, or micro cover glass 18 mm, thickness: 0.13–0.17 mm) were purchased from Matsunami Glass Ind., Ltd. (Osaka, Japan). All reagents were of analytical grade. Immobilization on mica treated with 3-aminopropyltriethoxysilane (APTES) was performed as follows19): Immediately after mica cleavage, the new surface was incubated with 200 µL of 0.3% (v/v) APTES aqueous solution for 30 min, followed by rinsing with MilliQ-water and air drying. The APTES was purchased from Tokyo Chemical Ind. Co., Ltd. (Tokyo, Japan).
Preparation of SamplesFor the AFM observations of the polystyrene nanoparticles in air, the sample suspensions diluted by water or 5% glucose to 10 or 25 µg/mL were incubated on the glass substrate or APTES modified-mica for 10–30 min. The samples were rinsed with 1–5 mL of milliQ water and then dried in air.
For the AFM observations of the nanoparticle in aqueous medium, we prepared the samples as follows. A cover glass or APTES-modified mica was glued onto the petri dish-type aqueous solution cell (for Shimadzu and Hitachi AFM 5000) or hand-made plastic cell on the slide glass for JPK nanowizard.20) The dilute suspension of polystyrene nanoparticles 25–250 µg/mL in water or 5% glucose was incubated on the glass substrate or APTES-modified mica for 10 min.
Measurement of Nanoparticles by AFMAFM imaging of nanoparticles was performed at room temperature by means of the QI mode of JPK Nanowizard Ultra Speed microscope, dynamic mode of Shimadzu SPM-9700 microscope, or dynamic force mode of Hitachi AFM5000/5400L microscopes with the following measurement conditions. In this study, the scan area was set so that digital resolution was less than 20 nm/pixel enough to detect the nanoparticles in all the AFM measurement. As long as we observe nanoparticles in this condition, there is no significant influence on the measured size of nanoparticles (Table S1).
JPK Nanowizard Ultra Speed MicroscopePrior to taking measurements, the spring constant of the cantilever was calibrated by the thermal noise method.21,22) Under air, AFM images (256 × 256 pixels) were obtained with a BL-AC40TS-C2 cantilever at a set point of 0.10 nN, an extend/retract speed of 85–120 µm/s, and a scan area of 2 × 2 µm2. In aqueous conditions, two kinds of cantilevers were used. In the case of the use of a BL-AC40TS-C2 cantilever, AFM images (256 × 256 pixels) were obtained at a set point of 0.10 nN, an extend/retract speed of 50–90 µm/s, and a scan area of 2 × 2 µm2. When using a OMCL-AC240TS cantilever, AFM images (128 × 128 pixels or 256 × 256 pixels) were obtained at a set point of 2 nN, an extend/retract speed of 40 µm/s, and a scan area of 2 × 2 µm2. All the obtained AFM images were subjected to the flattening process by JPK Data Processing software. We used Gwyddion software v.2.47 to extract the nanoparticles and the area-equivalent diameter (automated particle analysis) and height (cross-sectional line profiling) of nanoparticles were determined (Fig. S1).
Shimadzu SPM-9700 MicroscopeAll AFM images (256 × 256 pixels) were obtained with a Biolever mini cantilever at a scan speed of 0.5 Hz, and a scan area of 1 × 1 µm2 or 3 × 3 µm2. Under air, the operating frequency was set to be 88.4 kHz. Under aqueous conditions, the operating frequency was 23.7–27.5 kHz. By using Particle Analyzer Software (Shimadzu, Kyoto, Japan), all the obtained AFM images were subjected to the flattening process. Then, the area-equivalent diameter and height of nanoparticles were analyzed by automated particle analysis and cross-sectional line profiling, respectively.
Hitachi AFM5000 MicroscopeUnder air, AFM images (256 × 256 pixels) were obtained with the use of a SI-DF3P2 cantilever or a SI-DF40P2 cantilever at a scan speed of 0.19–0.38 Hz, and a scan area of 5 × 5 µm2. For the use of a SI-DF3P2 cantilever, the operating frequency was 74.0–78.0 kHz. For the SI-DF40P2 cantilever, the frequency was 261.5–270.7 kHz. In the case of aqueous conditions, AFM images (256 × 256 pixels) were obtained with the use of a SI-DF3P2 cantilever at an operating frequency of 58.1–64.1 kHz, a scan speed of 0.34–0.71 Hz, and a scan area of 2 × 2 µm2. A flattening process in the Spisel32 software was used to process the obtained AFM images. Then, the area-equivalent diameters and heights of the nanoparticles were analyzed by automated particle analysis and cross-sectional line profiling, respectively.
Hitachi AFM5400L MicroscopeAFM images in air condition (256 × 256 pixels) were obtained with the use of a SI-DF3P2 cantilever or a SI-DF40P2 cantilever at a scan speed of 0.28–0.40 Hz, and scan area of 5 × 5 µm2. For the use of a SI-DF3P2 cantilever, the operating frequency was 56.4–66.2 kHz. As for the SI-DF40P2 cantilever, the frequency was 308.9–309.2 kHz. All the obtained AFM images were subjected to a flattening process within the Spisel32 software. Then, the area-equivalent diameter and height of nanoparticles were analyzed by automated particle analysis and cross-sectional line profiling, respectively.
Statistical AnalysisGraphPad Prism software v.7.04 (GraphPad Software, La Jolla, CA, U.S.A.) was used to analyze the results of reproducibility study by one-way ANOVA, followed by Tukey’s test for comparison of multiple means. Differences were considered statistically significant at p < 0.05.
First, we examined the effect of spring constant of cantilever on the images and the resultant size of the polystyrene nanoparticles by means of JPK nanowizard in aqueous media. As shown in Table 1, there were no large difference between the values obtained using different cantilevers (BL-AC40TS-C2; Nominal Spring constant 0.09 -N/m, OMCL-AC240TS; Nominal Spring constant 2 N/m). The particle morphology obtained from images based on the two cantilevers were similar (Fig. 1).
| Cantilever | BL-AC40 TS-C2 | OMCL-AC240TS | ||
|---|---|---|---|---|
| Analysis method | Automated particle analysis | Cross-sectional line profiling | Automated particle analysis | Cross-sectional line profiling |
| Average (nm) | 133 | 99 | 115 | 100 |
| S.D. (nm) | 25 | 19 | 26 | 21 |
| Particle numbers | 256 | 256 | 256 | 256 |
Analytical conditions: Substrate, APTES-treated mica; Dispersant, 5% glucose; Instrument, JPK Nanowizard.

Scale bars are 500 nm. (Color figure can be accessed in the online version.)
The cantilever SI-DF3P2 (Nominal Spring constant: 2 N/m) was also available for use on the Hitachi AFM. However, the SI-DF40P2 (Nominal Spring constant: 26 N/m) could not be used for this measurement (data not shown), owing to disturbances during the image acquisition. In the case of a cantilever with a large spring constant, the attractive forces in combination with the repulsive elastic type of forces led to unstable scanning of the cantilever on the sample surface in dynamic (tapping/intermittent) contact mode.23,24)
For AFM observations of the nanoparticles, it is preferable to use a cantilever with a high enough stiffness to overcome attractive force such as capillary forces, Van der Waals’s forces, and electrostatic forces.25) However, cantilevers with a higher stiffness than that of sample might destroy or deform the nanoparticle.26) Therefore, there is a need to use a cantilever with a lower stiffness than that of the sample nanoparticles. Furthermore, cantilevers with a large resonance frequency can improve the sensitivity of the scanning and shorten the measurement time but might also damage the sample itself. Therefore, the selection of an optimized cantilever with an appropriate spring constant needs to consider these issues.
Effect of Analysis Method on Measured Size of Polystyrene NanoparticlesAs shown in Tables 2, 3, for the same substrate and cantilever, and dispersion media, the difference in sizes measured by different instruments was 12 nm, for automated particle analysis. Notably, the size difference was only 8 nm through cross-sectional line profiling in air. Conversely, in aqueous media, a 29- and 4-nm discrepancy between the two instruments was observed for automated particle analysis and cross-sectional line profiling, respectively. The cross-sectional line profiling gave the smaller discrepancy between the instruments both in air and aqueous media. Furthermore, in the case of cross-sectional line profiling, the difference of the values in air and aqueous media was smaller than that of the automated particle analysis, regardless of the type of instrument (Tables 2, 3). The values obtained by cross-sectional line profiling were also closer to those measured by DLS, 116 nm. The cross-sectional line profiling, which is not affected by the shape of the cantilever, can probably measure the particle size more accurately than the automated particle analysis by the area-equivalent diameter, which is affected by the shape of the cantilever.25) Therefore, subsequent experiments were performed with the use of cross-sectional line profiling.
| In air | In aqueous medium | |||
|---|---|---|---|---|
| Analysis method | Automated particle analysis | Cross-sectional line profiling | Automated particle analysis | Cross-sectional line profiling |
| Average (nm) | 136 | 108 | 119 | 106 |
| S.D. (nm) | 9.4 | 10 | 17 | 17 |
| Particle numbers | 137 | 34 | 450 | 316 |
Analytical conditions: Substrate, APTES-treated mica; cantilever, BL-AC40TS-C2; Dispersant, MlliQ water; Instrument, Shimadzu SPM-9700.
| In air | In aqueous medium | |||
|---|---|---|---|---|
| Analysis method | Automated particle analysis | Cross-sectional line profiling | Automated particle analysis | Cross-sectional line profiling |
| Average (nm) | 124 | 100 | 148 | 102 |
| S.D. (nm) | 24 | 18 | 26 | 24 |
| Particle numbers | 256 | 256 | 256 | 256 |
Analytical conditions: Substrate, APTES-treated mica; Cantilever, BL-AC40TS-C2; Dispersant, MlliQ water; Instrument, JPK Nanowizard.
Figure 2 shows the particle size distribution dependence on the number of analyzed particles. No. 6 distribution which was depicted with the use of 18 particles led to the result that the particles distribution was bimodal, in the range of 60–80, and 100–120 nm (Fig. 2, arrows). A similar tendency was observed from measurement No. 1 (particle number: 5). Conversely, the particle sizes based on the total particles in measurements Nos. 1 to 7 were normally distributed and the observed frequency from 60 to 80 nm was negligible. For measurement No. 7 based on 46 particles, the normal distribution of particle sizes was close to that of total of Nos. 1–7.
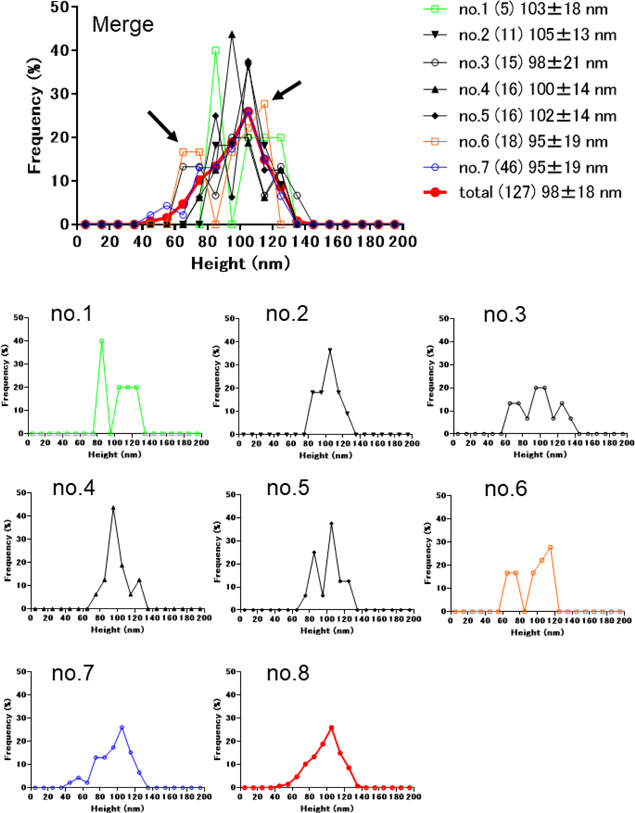
Numbers in parenthesis show the particle numbers used for analysis. The upper graph shows the merged one of the lower graphs. Solvent: 5% glucose, substrate; glass, cantilever, BL-AC 40 TS-C2. Arrows show the bimodal distribution of No. 6. (Color figure can be accessed in the online version.)
The reason for the bimodal distribution in No. 6 in Fig. 2 might be probabilistic. Our results show that approximately 100 particles are required for a representative measurement.
Effect of Solid Substrate and SolvationNext, we studied the effects of the glass substrate and mica substrate on the particle size. In air, it is difficult to observe polystyrene nanoparticles because of coagulation of polystyrene nanoparticles on the glass substrate when the particles were dispersed in water. Conversely, in aqueous media, both substrates enabled observation of the nanoparticles (Table 4). Although the results of the observation of polystyrene nanoparticles showed no notable differences between the glass and mica substrates, there was a tendency that the average sizes are smaller than on glass substrate than those on mica substrate (Table 4). We previously reported that the size of some liposomes adhered on substrates differs depending upon the procedures of immobilizing the liposomes onto a solid substrate.20) Therefore, the different degree of interaction with two substrates might affect the measured size of polystyrene nanoparticles. Table 4 also showed similar results in the sizes obtained from samples dispersed in 5% glucose and milliQ water, both on mica and glass substrates.
| Dispersant | 5% glucose | MIlliQ water | ||
|---|---|---|---|---|
| Substrate | Mica | Glass | Mica | Glass |
| Average (nm) | 99 | 98 | 102 | 95 |
| S.D. (nm) | 19 | 19 | 24 | 24 |
| Particle numbers | 256 | 256 | 256 | 256 |
Analytical conditions: Cantilever, BL-AC40TS-C2; Instrument: JPK Nanowizard.
Different instruments by different analysts and at different laboratories were used to measure the size of polystyrene nanoparticles in air (conditions A) and in aqueous media (condition B) (Table 5). As shown in Table 5, no significant differences occurred between the results obtained by each instrument under these two conditions.
| Condition A (In air) | |||
|---|---|---|---|
| Instrument | 1 | 2 | 3 |
| Average (nm) | 102 | 100 | 100 |
| S.D. | 16 | 16 | 18 |
| Particle numbers | 97 | 101 | 256 |
Analytical Condition: Substrate, APTES-treated mica; Dispersant: milliQ water; Instrument 1, Hitachi AMF 5000 (cantilever: SI-DF3P2); Instrument 2, Hitachi 5400 L (cantilever: SI-DF3P2), Instrument 3, JPK Nanowizard (cantilever: BL-AC40TS-C2).
| Condition B (In aqueous medium) | |||
|---|---|---|---|
| Instrument | 1 | 2 | 3 |
| Average (nm) | 101 | 102 | 106 |
| S.D. | 20 | 24 | 17 |
| Particle numbers | 93 | 256 | 316 |
Analytical conditions: Substrate, APTES-treated mica; Dispersant, milliQ water; Instrument 1, Hitachi AMF 5000 (cantilever: SI-DF3P2); Instrument 2, JPK Nanowizard (cantilever: BL-AC40TS-C2), Instrument 3, Shimadzu SPM-9700 (cantilever: BL-AC40TS-C2).
These results indicate good reproducibility of the method. Considering that the average measured diameters of the polystyrene nanoparticles by DLS were 116, 119, and 123 nm in three difference laboratories, with maximum differences of 7 nm, the maximum difference of 5 nm by AFM cross-sectional line profiling measured in aqueous medium (Table 5 condition B) was acceptable. The consideration points obtained in this study will be useful for the reproducible measurement of nanomedicines by AFM, directed by a consideration of the sample’s physical and chemical properties.
In this research, we studied the effects of various conditions on the measured particle size of a polystyrene nanoparticle CRM standard. The cantilever did not affect the measured size of the nanoparticles once it was optimized for the sample and measurement conditions in aqueous medium. The analytical method used to determine size from the images affected the results of the size measurement values. The cross-sectional line profile was more robust than the automated particle analysis by software and the values of cross-sectional line profiling gave similar result to those obtained by DLS. Approximately 100 particles are required for a representative measurement. There was no effect of the sample dispersant on the results observed in aqueous media. In aqueous media and in air there were no significant instrumental differences under the optimized conditions and showed good reproducibility. These results show that the AFM measurements under the optimized conditions might be a powerful tool for quality control of nanomedicines. Our experiments provide insight into the standardization of AFM for assessing the morphology and size of nanomedicines.
This work was supported in part by the Research on Development of New Drugs from the Japan Agency for Medical Research and Development, AMED (17ak0101074j0801, 18ak0101074j0802, 19ak0101074j0803).
The authors declare no conflict of interest.
The online version of this article contains supplementary materials.