Abstract
The appropriate localization of gastrinoma is still difficult. We aimed to evaluate the diagnostic accuracy of selective arterial calcium injection (SACI) for localization of gastrinomas including multiple lesions. This retrospective study included ten patients with surgically proven gastrinomas (gastrinoma group) and six patients without any findings suggesting Zollinger-Ellison syndrome (non-gastrinoma group). For SACI, calcium gluconate was injected into the arteries supplying pancreas, duodenum, and liver. Blood samples from the hepatic vein were obtained before and 30, 60, and 120 seconds after each injection. The results were considered positive when the increase in serum immunoreactive gastrin (IRG) levels within 60 seconds of calcium gluconate injection were more than 80 pg/mL and more than 20% from baseline. We evaluated the efficacy of SACI by comparing the SACI responses with definitive locations diagnosed by clinical and histopathological findings. In the gastrinoma group, false-positive responses were confirmed in seven of the ten patients. False-negative response was observed in one of the feeding arteries of one patient with gastrinomas in multiple locations. Conversely, the greatest increase in serum gastrin levels from baseline at 30 seconds indicated the true-positive responses in all patients with gastrinomas. In the non-gastrinoma group, calcium gluconate injection into gastroduodenal artery evoked positive responses in five of the six patients. In conclusion, our data suggest the strongest gastrin response evoked by SACI indicates the definitive location in patients with gastrinomas. In contrast, SACI could not accurately locate multiple gastrin-secreting lesions due to poor specificity.
ALTHOUGH the biochemical diagnosis of gastrinomas causing Zollinger-Ellison syndrome (ZES) is straightforward, localization of the tumor remains challenging. Previous studies reported that the sensitivity of conventional imaging studies such as computed tomography (CT), ultrasound, magnetic resonance imaging (MRI), and abdominal arteriography was poor, ranging from 40% to 70% [1]. Although recent guidelines consider positron emission tomography (PET)/CT with 68Ga-labeled somatostatin analogs as the first-line imaging method because of its high sensitivity and specificity for localizing neuroendocrine tumors (NETs) [2], only a small number of institutions can perform this technique. The localizing ability of somatostatin receptor scintigraphy, which can reach up to 91% in preoperative evaluation of gastrinomas [3], depends on tumor size, and tumors less than 1 cm in diameter are missed in more than 50% of the cases [4]. Conversely, duodenal gastrinomas less than 5 mm in diameter often cause hepatic metastases. Therefore, the utility of selective arterial secretagogue injection (SASI) has been proposed as a diagnostic method for localization of invisible microgastrinomas [5, 6]. Recently, efficacy of ultrasound endoscopy (EUS) has been recognized for the detection of small NETs especially in the pancreas [7, 8]. This modality had high sensitivity to detect CT-negative pancreas NETs [9]. However, even if EUS succeeds to localize NETs, there still remain concerns that another gastrinoma may co-exist in another area. In addition, in patients with ZES associated with multiple endocrine neoplasia type 1 (MEN-1), gastrinomas are located mostly in the duodenum, whereas numerous functional and non-functional endocrine tumors often simultaneously occupy pancreas [1, 5]. Therefore, SASI is also expected to distinguish multiple functional lesions from numerous tumors by evaluating gastrin response after stimulation of each feeding artery [10, 11].
SASI comprises selective serial catheterization of splenic, proper hepatic, gastroduodenal, and superior mesenteric arteries followed by gastrin secretagogue infusion into the arteries with simultaneous blood sampling of the hepatic venous effluent for gastrin concentration determination [1, 12, 13]. Imamura et al. was the first to describe SASI as a gastrinoma-localizing technique in 1987 [14]. The authors proposed that the feeding artery of a gastrinoma could be confirmed if the increase in serum level of immunoreactive gastrin (IRG) was more than 80 pg/mL and more than 20% above basal IRG level 40 seconds after the stimulation of each artery in response to 30 units of secretin [1, 15, 16]. Using these criteria, they were able to confirm SASI as a highly reliable localization technique in several case series [5, 14, 15]. While several medical institutions adopted these criteria [10, 17], others proposed different guidelines [18]. In addition, secretin was replaced with calcium gluconate in the early 2000s with the cessation of secretin production [5, 12]. Although several studies reported that calcium gluconate was a highly sensitive and specific alternative secretagogue to replace secretin for SASI, these results were supported by a small number of cases [12, 16].
Therefore, the diagnostic accuracy of selective arterial calcium injection (SACI) using calcium gluconate as the standard secretagogue should be confirmed in case series. In the current study analyzing the outcomes of SACI in patients with and without gastrinomas, we aimed to determine whether the criteria for SASI, which were based on the use of secretin as the secretagogue, were directly applicable to SACI and assess the diagnostic accuracy of SACI in localizing multiple gastrin-secreting lesions.
Patients and Methods
Patients
In this retrospective study, we analyzed data from the medical records of patients with and without gastrinoma who were referred to Tokyo Medical and Dental University Hospital and evaluated for changes in serum gastrin levels by SACI between January 2003 and March 2018. The present study complied with the principles laid by the Declaration of Helsinki and was approved by the ethics committee of Tokyo Medical and Dental University (M2017-309).
During the study period, SACI was performed for functional localization in 16 patients with hypergastrinemia. Patients without ZES, characterized by the presence of symptoms of gastric ulcer or gastro-esophageal reflux due to gastric hypersecretion, were excluded. Similarly, patients who did not undergo curative surgery during the study period were excluded because the definitive locations of the gastrinomas could not be determined. As a result, ten patients were included in the gastrinoma group. A significant reduction in serum IRG levels and subsequent improvement of ZES were confirmed postoperatively in all patients with gastrinomas. In addition, histopathological examinations of the resected specimens were confirmed to exhibit gastrin-positive cells in all patients (Fig. 1a).
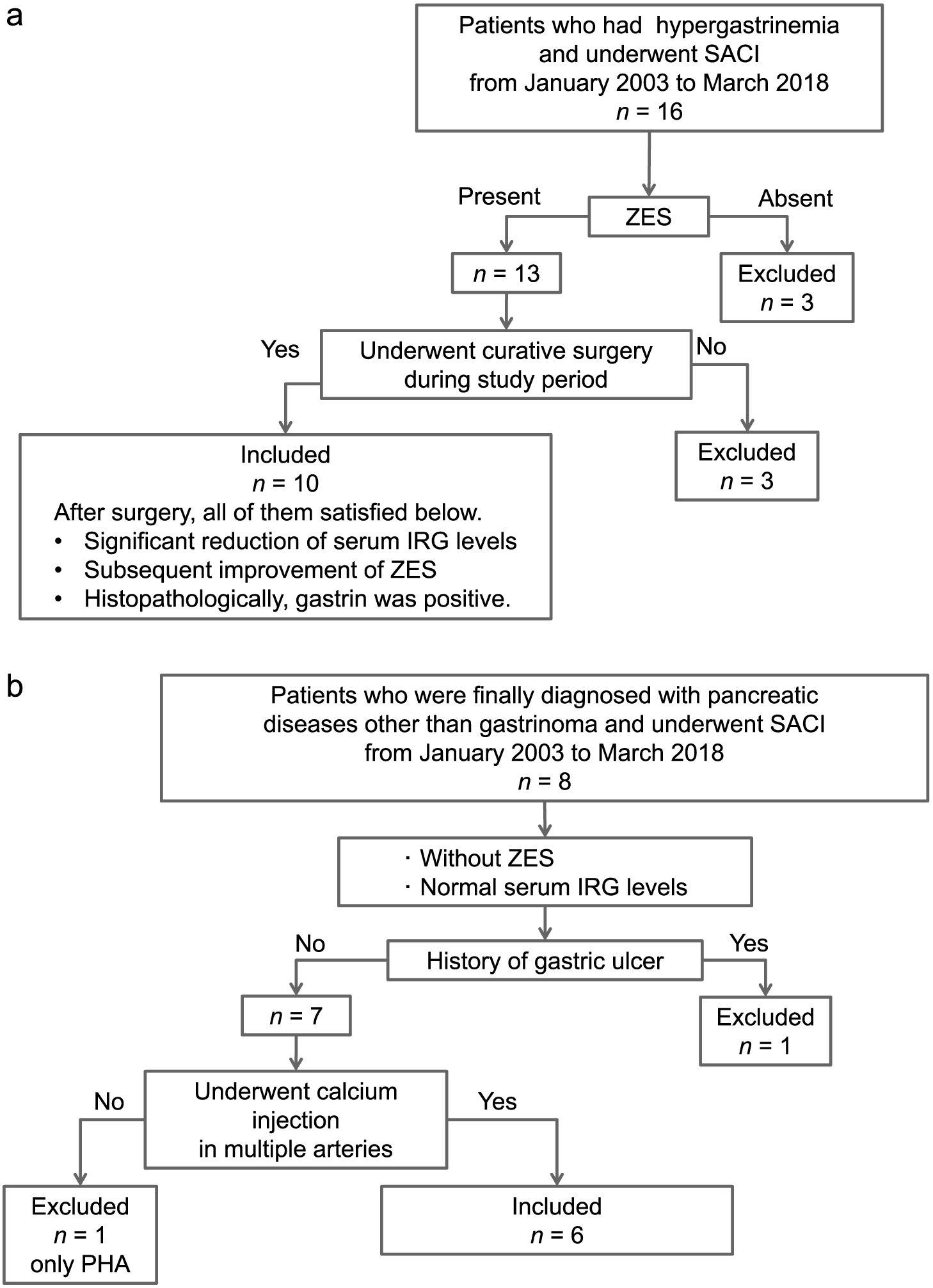
In addition, eight patients with a definitive diagnosis of a pancreatic disease other than gastrinoma underwent SACI for the evaluation of gastrin secretion during the study period. The primary aim of SACI in these patients was localization of insulin-secreting lesions. Therefore, these patients did not develop ZES and had normal serum IRG levels. After the exclusion of one patient with a history of gastric ulcer and one patient in whom calcium gluconate was injected only into proper hepatic artery, the remaining six patients were included in the non-gastrinoma group (Fig. 1b).
Outcome variables
Data regarding age, sex, presence of MEN-1, concomitant use of proton pump inhibitors (PPIs) and H2 receptor antagonists, site of surgical resection, histopathological findings, and pre- and/or postoperative serum IRG levels were collected from the medical records of the patients. Serum IRG levels were determined by a radioimmunoassay (gastrin RIA kit II, Fujirebio, Tokyo, Japan).
SACI
SACI was performed as previously described [19]. Briefly, a venous catheter was placed in right or middle hepatic vein for blood sampling. Standard abdominal arteriography was performed after selective catheterization of gastroduodenal artery (GDA), superior mesenteric artery (SMA), and proximal splenic artery (SpA). In most cases, proper hepatic artery (HA) and distal SpA were also catheterized. In addition, inferior pancreaticoduodenal, posterior superior pancreaticoduodenal, dorsal pancreatic, right hepatic, middle hepatic, and left hepatic arteries were cannulated if appropriate. Calcium gluconate (0.025 mEq/kg) was injected as a bolus into each artery. The interval between each calcium injection was at least 5 minutes. Blood samples were collected at baseline and 30, 60, 90, and 120 seconds after the calcium gluconate injection. Basically, we recognize SpA supplies the pancreatic body and tail, GDA and SMA supply the pancreatic head and neck, and proper HA supplies liver. In addition, to recognize arterial anatomies, the actual vascular territory of each artery was confirmed by arteriography, and selective enhanced CT was performed as needed.
Data analysis
For each artery evaluated by SACI, a maximum serum IRG elevation of more than 80 pg/mL and greater than 20% above the basal serum IRG level within 60 seconds of calcium gluconate injection was defined to indicate the presence of gastrinoma localized to the area supplied by the investigated artery. In the gastrinoma group, these results were compared with the definitive location of the gastrinomas based on the histopathological assessment by the pathologists and clinical outcomes of surgical treatment. In the non-gastrinoma group, arteries that were false-positive for the presence of gastrinoma by SACI were identified. Furthermore, we explored to elucidate other criteria that could predict the location of gastrinomas more accurately.
Results
The basal IRG levels were elevated in all ten patients in the gastrinoma group compared with the normal range (37–172 pg/mL) (Table 1a). None of the patients had undergone gastrectomy before SACI evaluation. One patient had concurrent MEN-1. Morphological imaging studies (enhanced CT, MRI, and EUS) failed to identify tumors in one case (case 1). Treatment with PPIs or H2 receptor antagonists could not be discontinued before SACI due to severe gastrointestinal symptoms in six patients. Serum IRG levels were decreased dramatically after surgery in all patients. During the follow-up period, recurrence was observed in three cases without elevation of serum IRG levels. In one case, CT revealed metastases in lymph nodes 31 months after surgery (case 3). In the other two cases, MRI revealed postoperative recurrence in liver after nine and eight months, respectively (cases 4 and 7). In the non-gastrinoma group, no patient had symptoms of ZES (Table 1b). In addition, the basal IRG levels were within the normal range, and PPIs or H2 receptor antagonists were not administered in these patients. Four patients in the non-gastrinoma group underwent surgical resection, and the histopathological evaluation showed that the resected tissues were negative for gastrin in all cases.
Table 1a
Clinical characteristics of patients in the gastrinoma group
| Case |
Age (years) |
Gender |
MEN-1 |
Preoperative IRG
(pg/mL) |
Antisecretory therapy before SACI |
Imaging studies |
Operation |
Histopathological findings |
Postoperative IRG
(pg/mL) |
Follow-up
(month) |
Recurrence |
| Drug class |
Withdrawal |
Location |
Location |
Size of the largest tumor (mm) |
| 1 |
51 |
F |
– |
2,240 |
PPIs |
not done |
ND |
PPPD |
LN* |
12 |
93 |
153 |
— |
| 2 |
65 |
F |
– |
684 |
PPIs |
not done |
P(B&T) |
DP |
P(B&T), LN |
35 |
57 |
87 |
— |
| 3 |
59 |
M |
– |
1,960 |
PPIs |
not done |
D |
PR |
D, LN |
8 |
225 |
31 |
LN |
| 4 |
55 |
F |
+ |
1,370 |
H2RA |
done (for 1 day) |
D |
SSPPD |
D |
8 |
21 |
9 |
L |
| 5 |
65 |
F |
– |
6,480 |
PPIs |
done (for 4 days) |
P(H), L(S4) |
SSPPD, PH |
P(H), L, LN |
28 |
68 |
25 |
— |
| 6 |
75 |
M |
– |
8,810 |
PPIs |
not done |
P(H) |
SSPPD |
D, LN |
10 |
63 |
6 |
— |
| 7 |
52 |
M |
– |
8,640 |
PPIs |
not done |
D |
SSPPD |
D, LN |
20 |
179 |
8 |
L |
| 8 |
79 |
F |
– |
3,430 |
PPIs |
not done |
LN(#6) |
PD |
LN(#6)** |
11 |
36 |
8 |
— |
| 9 |
48 |
F |
– |
1,700 |
— |
not done |
P(B&T) |
DP |
P(B&T) |
48 |
72 |
31 |
— |
| 10 |
59 |
M |
– |
190 |
PPIs |
done (for 7 days) |
D |
SSPPD |
P(H), D, LN |
20 |
33 |
1 |
— |
*Apparent primary lymph node gastrinoma near the uncinate process of pancreas. **Lymph node metastasis of duodenal neuroendocrine tumor resected ten years earlier.
Abbreviations: M, male; F, female; PPIs, proton pump inhibitors; H2RA, H2 receptor antagonists; MEN-1, multiple endocrine neoplasia type 1; IRG, immunoreactive gastrin; SACI, selective arterial calcium injection; ND, not detected; PD, pancreaticoduodenectomy; SSPPD, subtotal stomach-preserving pancreaticoduodenectomy; PPPD, pylorus-preserving pancreaticoduodenectomy; DP, distal pancreatectomy; PR, partial resection of duodenum; PH, partial hepatectomy; D, duodenum; P(H), pancreas head; P(B&T), pancreas body and tail; L, liver; LN, lymph node
Table 1b
Clinical characteristics of patients in the non-gastrinoma group
| Case |
Age
(years) |
Gender |
MEN-1 |
IRG
(pg/mL) |
Imaging studies |
Operation |
Diagnosis |
| Location |
| 11 |
67 |
F |
– |
105 |
P(B) |
DP |
glucagonoma |
| 12 |
45 |
F |
– |
62 |
ND |
— |
reactive hypoglycemia |
| 13 |
39 |
M |
– |
135 |
P(B&T), L |
DP, PH |
insulinoma |
| 14 |
31 |
F |
– |
68 |
P(T), L |
— |
insulinoma |
| 15 |
56 |
M |
– |
82 |
P(B) |
DP |
hamartoma |
| 16 |
65 |
F |
+ |
65 |
P(H), L |
SSPPD, PH |
insulinoma |
Abbreviations: M, male; F, female; MEN-1, multiple endocrine neoplasia type 1; IRG, immunoreactive gastrin; SACI, selective arterial calcium injection; ND, not detected; SSPPD, subtotal stomach-preserving pancreaticoduodenectomy; DP, distal pancreatectomy; PH, partial hepatectomy; P(H), pancreas head; P(B), pancreas body; P(B&T), pancreas body and tail; P(T), pancreas tail; L, liver
Fig. 2a and 2b show the responses to SACI, and Table 2a and 2b show the maximum increase rate (IRGmax/basal IRG) and the maximum increase in serum IRG levels from baseline (ΔIRGmax) within 60 seconds for each artery in each patient. In the gastrinoma group, 30 of the 50 arteries stimulated using SACI were positive. GDA was not injected with calcium gluconate due to retrograde flow in one case (case 10). Comparison of the SACI responses with the definitive localizations confirmed postoperatively revealed that positive responses were observed not only in the arteries feeding the area of the gastrinomas but also in other arteries in seven of the ten patients (cases 1, 2, 3, 4, 6, 7, and 8). In contrast, an insufficient increase in serum IRG level was observed in one of the feeding arteries of the gastrinoma which was confirmed by histopathological examination in one patient (case 5) (Table 2a). In the non-gastrinoma group, 11 of the 36 stimulated arteries were positive, and calcium gluconate injection into GDA led to a positive response in five of the six patients (cases 11, 12, 13, 15, and 16) (Table 2b), with maximum serum IRG levels reaching up to 636 pg/mL in one patient (case 16) (Fig. 2b).
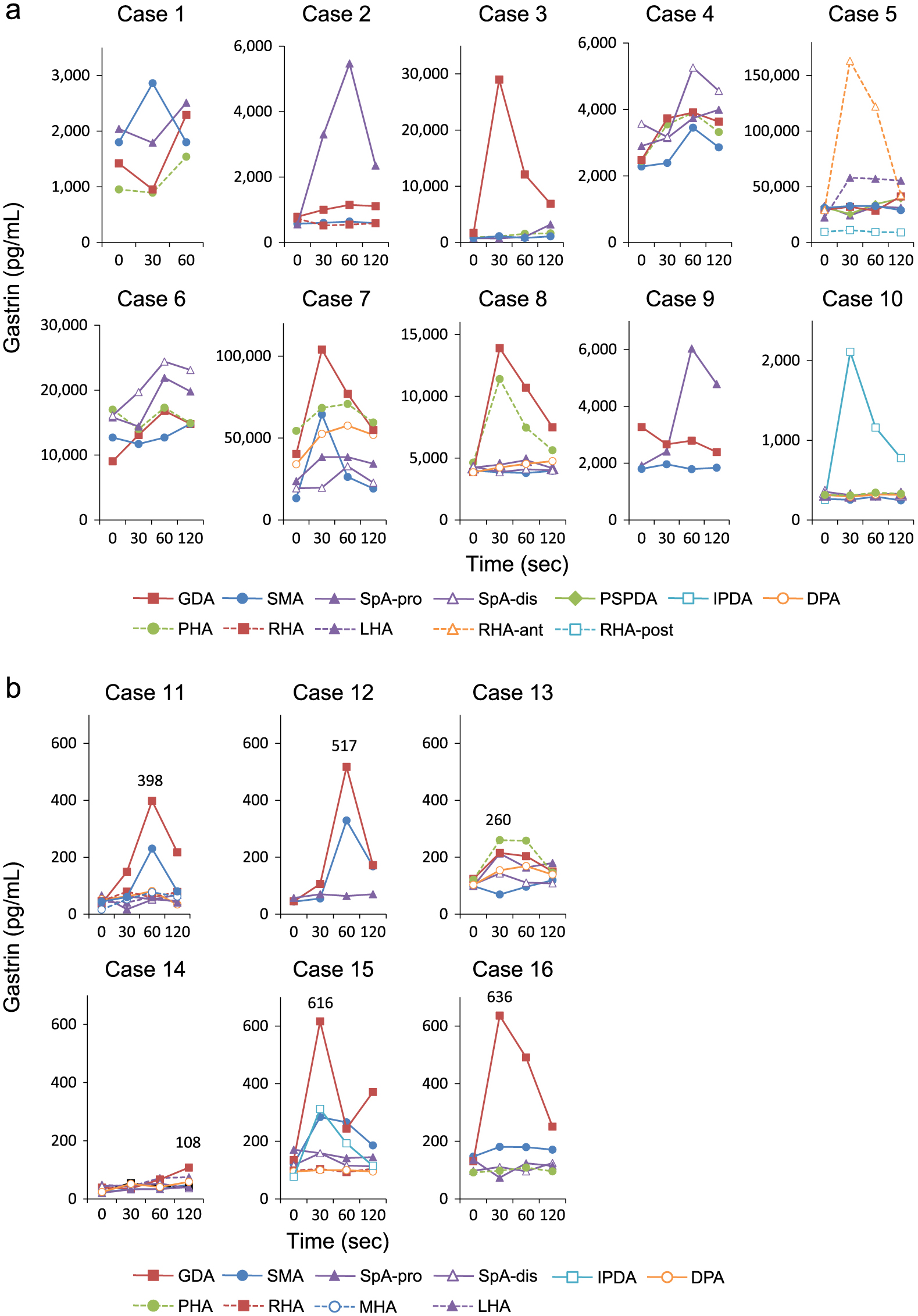
Table 2a
IRG levels and tumor locations determined by SACI in the gastrinoma group
| Case |
|
GDA |
SMA |
SpA-pro |
SpA-dis |
PSPDA |
IPDA |
DPA |
PHA |
RHA |
LHA |
RHA-ant |
RHA-post |
Gastrinoma |
| Location |
| 1 |
IRGmax/basal IRG |
1.61 |
1.59 |
1.23 |
|
|
|
|
1.62 |
|
|
|
|
LN* |
| ΔIRGmax |
870 |
1,060 |
470 |
|
|
|
|
588 |
|
|
|
|
|
| 2 |
IRGmax/basal IRG |
1.47 |
1.12 |
9.91 |
|
|
|
|
|
1 |
|
|
|
P(B&T), LN |
| ΔIRGmax |
366 |
68 |
4,918 |
|
|
|
|
|
0 |
|
|
|
|
| 3 |
IRGmax/basal IRG |
17.16 |
1.45 |
1.31 |
|
|
|
|
1.69 |
|
|
|
|
D, LN |
| ΔIRGmax |
27,310 |
342 |
234 |
|
|
|
|
627 |
|
|
|
|
|
| 4 |
IRGmax/basal IRG |
1.58 |
1.51 |
1.29 |
1.47 |
|
|
|
1.59 |
|
|
|
|
D |
| ΔIRGmax |
1,430 |
1,170 |
840 |
1,690 |
|
|
|
1,450 |
|
|
|
|
|
| 5 |
IRGmax/basal IRG |
1.1 |
1.05 |
1 |
|
1.1 |
|
|
|
|
2.59 |
5.6 |
1.16 |
P(H), L, LN |
| ΔIRGmax |
2,800 |
1,700 |
0 |
|
3,100 |
|
|
|
|
35,700 |
133,900 |
1,520 |
|
| 6 |
IRGmax/basal IRG |
1.86 |
1 |
1.39 |
1.52 |
|
|
|
1.02 |
|
|
|
|
D, LN |
| ΔIRGmax |
7,750 |
0 |
6,100 |
8,300 |
|
|
|
300 |
|
|
|
|
|
| 7 |
IRGmax/basal IRG |
2.58 |
4.84 |
1.61 |
1.7 |
|
|
1.69 |
1.3 |
|
|
|
|
D, LN |
| ΔIRGmax |
63,700 |
51,100 |
14,600 |
13,500 |
|
|
23,600 |
16,400 |
|
|
|
|
|
| 8 |
IRGmax/basal IRG |
3.57 |
1 |
1.14 |
1 |
|
|
1.18 |
2.46 |
|
|
|
|
LN(#6)** |
| ΔIRGmax |
10,010 |
0 |
600 |
0 |
|
|
900 |
6,760 |
|
|
|
|
|
| 9 |
IRGmax/basal IRG |
1 |
1.09 |
3.14 |
|
|
|
|
|
|
|
|
|
P(B&T) |
| ΔIRGmax |
0 |
160 |
4,110 |
|
|
|
|
|
|
|
|
|
|
| 10 |
IRGmax/basal IRG |
|
1.11 |
1.01 |
1 |
|
8.34 |
1.01 |
1.09 |
|
|
|
|
P(H), D, LN |
| ΔIRGmax |
|
29 |
3 |
0 |
|
1,857 |
2 |
29 |
|
|
|
|
|
Bold numerals indicate values higher than 80 pg/mL and greater than 20% above basal serum IRG. Underlined values indicate those with an insufficient increase in the artery feeding the gastrinoma based on postoperative confirmation.
*Apparent primary lymph node gastrinoma near the uncinate process of pancreas. **Lymph node metastasis of duodenal neuroendocrine tumor resected ten years earlier.
Abbreviations: IRG, immunoreactive gastrin; SACI, selective arterial calcium injection; GDA, gastroduodenal artery; SMA, superior mesenteric artery; SpA-pro, proximal splenic artery; SpA-dis, distal splenic artery; PSPDA, posterior superior pancreaticoduodenal artery; IPDA, inferior pancreaticoduodenal artery; DPA, dorsal pancreatic artery; PHA, proper hepatic artery; RHA, right hepatic artery; LHA, left hepatic artery; RHA-ant, anterior branch of right hepatic artery; RHA-post, posterior branch of right hepatic artery; D, duodenum; P(H), pancreas head; P(B&T), pancreas body and tail; L, liver; LN, lymph node
Table 2b
IRG levels and tumor locations determined by SACI in the non-gastrinoma group
| Case |
|
GDA |
SMA |
SpA-pro |
SpA-dis |
IPDA |
DPA |
PHA |
RHA |
MHA |
LHA |
Diagnosis |
| 11 |
IRGmax/basal IRG |
10.76 |
5.23 |
1 |
1.37 |
|
1.56 |
|
1.72 |
4.81 |
1.4 |
glucagonoma |
| ΔIRGmax |
361 |
186 |
0 |
17 |
|
29 |
|
33 |
61 |
17 |
|
| 12 |
IRGmax/basal IRG |
11.49 |
7.48 |
1.23 |
|
|
|
|
|
|
|
reactive hypoglycemia |
| ΔIRGmax |
472 |
285 |
13 |
|
|
|
|
|
|
|
|
| 13 |
IRGmax/basal IRG |
1.73 |
1 |
2.15 |
1.38 |
|
1.64 |
2.15 |
|
|
|
insulinoma |
| ΔIRGmax |
91 |
0 |
114 |
39 |
|
66 |
139 |
|
|
|
|
| 14 |
IRGmax/basal IRG |
1.72 |
1.48 |
1.62 |
1.07 |
|
2.17 |
|
1.8 |
|
1.47 |
insulinoma |
| ΔIRGmax |
28 |
11 |
13 |
9 |
|
28 |
|
24 |
|
23 |
|
| 15 |
IRGmax/basal IRG |
4.56 |
2.33 |
1 |
1.36 |
4.05 |
1.05 |
|
1.05 |
|
|
hamartoma |
| ΔIRGmax |
481 |
162 |
0 |
42 |
235 |
5 |
|
5 |
|
|
|
| 16 |
IRGmax/basal IRG |
3.75 |
1.22 |
1 |
1.13 |
|
|
1.07 |
|
|
|
insulinoma |
| ΔIRGmax |
505 |
34 |
0 |
13 |
|
|
17 |
|
|
|
|
Bold numerals indicate values higher than 80 pg/mL and greater than 20% above basal serum IRG.
Abbreviations: IRG, immunoreactive gastrin; SACI, selective arterial calcium injection test; GDA, gastroduodenal artery; SMA, superior mesenteric artery; SpA-pro, proximal splenic artery; SpA-dis, distal splenic artery; IPDA, inferior pancreaticoduodenal artery; DPA, dorsal pancreatic artery; PHA, proper hepatic artery; RHA, right hepatic artery; MHA, middle hepatic artery; LHA, left hepatic artery
Table 3a shows increases in serum IRG levels from baseline at 30 seconds after the calcium gluconate injection (ΔIRG30sec) in the gastrinoma group. In all cases, the arteries with the greatest ΔIRG30sec were consistent with the tumor-feeding arteries determined by the postoperative definitive localization of the gastrinomas. Table 3b showed the comparisons of IRGmax/basal IRG and ΔIRGmax between feeding arteries and non-feeding arteries shown in Table 2a. The term “definite feeding arteries” means the artery which showed the strongest gastrin response among arteries corresponding to the definitive localization of the gastrinomas in each case. The term “definite non-feeding arteries” means the arteries which are recognized to feed different area from the true locations in each case (SpA and HA for gastrinomas in the pancreas head and the duodenum region, GDA, SMA, and HA for those in the pancreas body and tail region, GDA, SMA and SpA for those in the hepatic region, respectively). In feeding arteries, IRGmax/basal IRG ranged from 1.58–17.16 (median 4.21) and ΔIRGmax ranged from 1,060–133,900 pg/mL (median 6,334 pg/mL), respectively. In non-feeding arteries, IRGmax/basal IRG ranged from 1.00–2.46 (median 1.29) and ΔIRGmax ranged from 0–16,400 pg/mL (median 470 pg/mL), respectively.
Table 3a
Increases in IRG levels from baseline at 30 seconds after calcium injection (ΔIRG
30sec) in the gastrinoma group
| Case |
GDA |
SMA |
SpA-pro |
SpA-dis |
PSPDA |
IPDA |
DPA |
PHA |
RHA |
LHA |
RHA-ant |
RHA-post |
Gastrinoma |
| Location |
| 1 |
–467 |
1,060 |
–250 |
|
|
|
|
–59 |
|
|
|
|
LN* |
| 2 |
213 |
24 |
2,748 |
|
|
|
|
|
–210 |
|
|
|
P(B&T), LN |
| 3 |
27,310 |
342 |
–51 |
|
|
|
|
177 |
|
|
|
|
D, LN |
| 4 |
1,250 |
110 |
240 |
–400 |
|
|
|
1,100 |
|
|
|
|
D |
| 5 |
2,800 |
1,700 |
–8,700 |
|
–5,700 |
|
|
|
|
35,700 |
133,900 |
1,520 |
P(H), L, LN |
| 6 |
4,050 |
–1,000 |
–1,400 |
3,600 |
|
|
|
–3,000 |
|
|
|
|
D, LN |
| 7 |
63,700 |
51,100 |
14,600 |
400 |
|
|
18,600 |
14,000 |
|
|
|
|
D, LN |
| 8 |
10,010 |
–130 |
230 |
–450 |
|
|
390 |
6,760 |
|
|
|
|
LN(#6)** |
| 9 |
–610 |
160 |
490 |
|
|
|
|
|
|
|
|
|
P(B&T) |
| 10 |
|
–10 |
–12 |
–42 |
|
1,857 |
–23 |
–7 |
|
|
|
|
P(H), D, LN |
Bold numerals indicate the greatest increase in each case.
*Apparent primary lymph node gastrinoma near the uncinate process of pancreas. **Lymph node metastasis of duodenal neuroendocrine tumor resected ten years earlier.
Abbreviations: IRG, immunoreactive gastrin; GDA, gastroduodenal artery; SMA, superior mesenteric artery; SpA-pro, proximal splenic artery; SpA-dis, distal splenic artery; PSPDA, posterior superior pancreaticoduodenal artery; IPDA, inferior pancreaticoduodenal artery; DPA, dorsal pancreatic artery; PHA, proper hepatic artery; RHA, right hepatic artery; LHA, left hepatic artery; RHA-ant, anterior branch of right hepatic artery; RHA-post, posterior branch of right hepatic artery; D, duodenum; P(H), pancreas head; P(B&T), pancreas body and tail; L, liver; LN, lymph node
Table 3b
Comparison of IRG levels between definite feeding arteries and definite non-feeding arteries
|
Definite feeding arteries (n = 10) |
Definite non-feeding arteries (n =
34) |
| IRGmax/basal IRG |
4.21 (1.58–17.16) |
1.29 (1.00–2.46) |
| ΔIRGmax |
6,334 (1,060–133,900) |
470 (0–16,400) |
Data are presented as median with range.
Abbreviations: IRG, immunoreactive gastrin
Discussion
This single-institution study evaluated the clinical significance of SACI by comparing the SACI responses with the postoperative definitive diagnosis. Notably, we observed false positivity after the selective stimulation of arteries in a substantial number of patients with gastrinomas. Conversely, the evaluation by SACI was negative in one artery in a patient with gastrinomas located at two sites. Furthermore, apparent increases in serum IRG levels by GDA stimulation were observed in most of the patients without gastrinomas. These results suggest that interpretation of SACI responses should be reconsidered.
In 1972, Isenberg et al. discovered the paradoxical rise in serum gastrin levels after intravenous injection of secretin in patients with ZES [20]. Based on this observation unique to gastrinoma, Imamura et al. proposed the concept underlying SASI [14] and the criteria for localization of gastrinomas that could not be visualized by conventional imaging studies more than three decades ago [1, 15, 16]. At the time, total gastrectomy was usually performed in patients with ZES [16], which led to the resolution of most of the physiological gastrin secretion, thereby enhancing gastrin response after stimulation of the artery feeding the gastrinoma. However, with efficient PPIs, total or partial gastrectomy is no longer indicated [21]. Additionally, given that high extracellular Ca2+ levels induce gastrin release from human gastrinoma cells that express calcium receptors [22], calcium gluconate has been adopted as the superior secretagogue as an alternative to secretin for SASI. However, unlike secretin, increased Ca2+ concentrations lead to gastrin release from antral gastrin-producing cells [23]. SASI is based on the observation that the secretagogue stimulates the release of gastrin from gastrinoma but not normal gastrin-secreting cells in the gastric antral region and duodenal bulb mucosa. Therefore, several concerns remain regarding SACI. Whether SACI can determine the absence of gastrinoma is unclear. To date, few studies compared SASI using stimulation with secretin and calcium gluconate [12, 16].
No case series to date determined serum IRG responses to SACI in patients without gastrinomas. In the current study, the SACI response was positive in GDA in the majority of patients without gastrinomas as well, which indicates the low specificity of SACI in gastrinoma. We speculate that calcium gluconate might have led to physiological gastrin release from antral gastrin-producing cells, which should be taken into consideration while interpreting the GDA response to SACI.
The response to SACI was positive not only in the arteries feeding the definitive gastrinoma locations but also other arteries in seven of the ten patients with gastrinomas. In these patients, we confirmed the remission of ZES and a significant reduction in serum IRG levels after surgery. Therefore, it is unlikely that the occult gastrinomas remained after surgical resection and SACI had actually detected the location of these lesions. Previous studies reported that SASI using secretin could lead to an increase in gastrin at multiple sites; anastomoses among the feeding arteries were considered to underlie the false-positive localization [24]. Regarding SACI for insulinoma localization, Thompson et al. reported a positive response in one, two, and three arterial distributions in 73.8%, 21.4%, and 4.8% in a cohort of 42 patients, respectively [25]. They also observed significant variations and overlaps in regional pancreatic perfusion and suggested that major pancreatic arteries may perfuse overlapping distributions, resulting in a positive response after calcium gluconate injection into different arteries in the presence of a single insulinoma.
In the current study, in one case, SACI failed to indicate a positive response in one of the feeding arteries (posterior superior pancreaticoduodenal artery) of multiple gastrinomas although the tumor cells reacted to the anti-gastrin serum (case 5). Imamura et al. reported one patient with gastrinoma which was confirmed to lack gastrin secretion in culture medium even though the tumor specimen was positive for gastrin by histopathological assessment [14]. Therefore, in the current case, it was possible that SACI failed to detect the gastrin-producing gastrinoma in the pancreas head because the tumor was functionally silent.
In the current study, SACI using the criteria proposed by Imamura et al. was not effective in localizing gastrinomas. The criteria for SASI using secretin, proposed by Thom et al., include the ratio of post-stimulation serum IRG level to baseline serum IRG level [18]. In their case series, the authors calculated mean response ratio of true-negative arteries stimulated by secretin and defined the positive response as a ratio greater than two standard deviations above the mean (>1.48 at 30 seconds or >2.09 at 60 seconds). Indeed, some studies considered the SACI response as positive if the increase in serum gastrin levels were twofold or greater within 30–120 seconds after the calcium gluconate injection [12, 19]. In the current study, we could not propose the optimal diagnostic criteria by adjusting the cut-off ratio of the maximum post-stimulation IRG level to the baseline IRG level due to a considerable overlap; the minimum ratio of true-positive arteries was 1.58 (Case 4), which is smaller than the values of some false-positive arteries in gastrinoma group (shown in Tables 2a and 3b). However, the greatest ΔIRG30sec at least indicated the true localization of gastrinomas in all patients with ZES. Overt gastrinomas appear to exhibit greater and earlier response to stimulation by calcium gluconate injection. On the other hand, regarding SACI for insulinoma localization, several studies addressed a similar issue. Although a positive response to the calcium gluconate injection is usually defined as the doubling of the insulin concentration from the baseline, these studies demonstrated that false-positive cases were not rare using the criteria and recommended defining the peak insulin cut-off for improved interpretation of response to SACI [26, 27]. Other authors suggested that the artery with the greatest response should be considered as the dominant artery [28]. In addition, changes in the timing of the post-injection evaluation [29] or the dose of calcium gluconate [30] were proposed for improvement in retrospective studies that assessed the accuracy of SACI for localization. In the current study, we found that the artery with the greatest response corresponded with the true localization of the gastrinoma in patients with ZES. However, considering those various mentions for insulinoma localization using SACI in previous studies, further multimodal approaches should be evaluated to elucidate the most appropriate criteria for SACI to localize gastrinomas.
This retrospective study has several limitations. First, most of the patients with gastrinomas could not discontinue PPIs prior to SACI because of severe ZES. PPIs cause hypergastrinemia in most normal subjects, have an extended duration of action of up to one week, and can confound diagnosis [2]. Therefore, failure to discontinue PPIs might have affected the SACI outcomes in the current study. However, recent guidelines no longer recommend abrupt PPI discontinuation even for the diagnostic workup of gastrinomas because acid peptic complications can develop rapidly [2]. In addition, one case series reported the successful localization of gastrinomas by SACI in patients who remained on PPI or H2 receptor antagonist therapy [19]. Second, although we confirmed a significant reduction in serum IRG levels and subsequent improvement of ZES after surgery in all patients with gastrinomas, the absence of invisible gastrinomas except the resected lesions could not be proven conclusively. Third, the number of eligible patients was not large enough to avoid sample bias. Therefore, generalization of our findings may be limited. However, the current study included not only patients with gastrinomas but also those without abnormal gastrin secretion as the control group, which is a strength of the current study assessing the specificity of SACI.
In summary, our results suggest that arteries with the greatest increase in serum IRG levels could be the tumor-feeding arteries, which can be utilized for the definitive localization of gastrinomas in patients with clearly established diagnosis of ZES. Conversely, distinguishing multiple gastrin-secreting lesions from non-functioning lesions by SACI seems to be difficult. The criteria proposed since the introduction of SASI using secretin did not exhibit high diagnostic accuracy in the current study patients who underwent SACI. Indications for SACI and its results should be evaluated with caution because of the changes in the background of gastrinoma treatment.
Disclosure
The authors declare that they have no conflicts of interest.
References
- 1 Imamura M, Komoto I, Ota S (2006) Changing treatment strategy for gastrinoma in patients with Zollinger-Ellison syndrome. World J Surg 30: 1–11.
- 2 Falconi M, Eriksson B, Kaltsas G, Bartsch DK, Capdevila J, et al. (2016) ENETS consensus guidelines update for the management of patients with functional pancreatic neuroendocrine tumors and non-functional pancreatic neuroendocrine tumors. Neuroendocrinology 103: 153–171.
- 3 Norton JA, Fraker DL, Alexander HR, Venzon DJ, Doppman JL, et al. (1999) Surgery to cure the Zollinger-Ellison syndrome. N Engl J Med 341: 635–644.
- 4 Alexander HR, Fraker DL, Norton JA, Bartlett DL, Tio L, et al. (1998) Prospective study of somatostatin receptor scintigraphy and its effect on operative outcome in patients with Zollinger-Ellison syndrome. Ann Surg 228: 228–238.
- 5 Imamura M, Komoto I, Ota S, Hiratsuka T, Kosugi S, et al. (2011) Biochemically curative surgery for gastrinoma in multiple endocrine neoplasia type 1 patients. World J Gastroenterol 17: 1343–1353.
- 6 Okada K, Sudo T, Miyamoto K, Yokoyama Y, Sakashita Y, et al. (2016) The selective arterial calcium injection test is a valid diagnostic method for invisible gastrinoma with duodenal ulcer stenosis: a case report. Hiroshima J Med Sci 65: 13–17.
- 7 Puli SR, Kalva N, Bechtold ML, Pamulaparthy SR, Cashman MD, et al. (2013) Diagnostic accuracy of endoscopic ultrasound in pancreatic neuroendocrine tumors: a systematic review and meta analysis. World J Gastroenterol 19: 3678–3684.
- 8 Ramage JK, Ahmed A, Ardill J, Bax N, Breen DJ, et al. (2012) Guidelines for the management of gastroenteropancreatic neuroendocrine (including carcinoid) tumours (NETs). Gut 61: 6–32.
- 9 Khashab MA, Yong E, Lennon AM, Shin EJ, Amateau S, et al. (2011) EUS is still superior to multidetector computerized tomography for detection of pancreatic neuroendocrine tumors. Gastrointest Endosc 73: 691–696.
- 10 Hussain JS, Srinivasa RN, Hage A, Pampati R, Chick JFB (2018) Bringing SASI back: single session selective arterial secretin injection and transarterial embolization of intrahepatic pancreatic neuroendocrine metastasis in a MEN-1 patient. Radiol Case Rep 13: 333–335.
- 11 Imamura M, Nakamoto Y, Uose S, Komoto I, Awane M, et al. (2015) Diagnosis of functioning pancreaticoduodenal neuroendocrine tumors. J Hepatobiliary Pancreat Sci 22: 602–609.
- 12 Turner JJ, Wren AM, Jackson JE, Thakker RV, Meeran K (2002) Localization of gastrinomas by selective intra-arterial calcium injection. Clin Endocrinol (Oxf) 57: 821–825.
- 13 Doppman JL, Miller DL, Chang R, Maton PN, London JF, et al. (1990) Gastrinomas: localization by means of selective intraarterial injection of secretin. Radiology 174: 25–29.
- 14 Imamura M, Takahashi K, Adachi H, Minematsu S, Shimada Y, et al. (1987) Usefulness of selective arterial secretin injection test for localization of gastrinoma in the Zollinger-Ellison syndrome. Ann Surg 205: 230–239.
- 15 Imamura M, Takahashi K, Isobe Y, Hattori Y, Satomura K, et al. (1989) Curative resection of multiple gastrinomas aided by selective arterial secretin injection test and intraoperative secretin test. Ann Surg 210: 710–718.
- 16 Wada M, Komoto I, Doi R, Imamura M (2002) Intravenous calcium injection test is a novel complementary procedure in differential diagnosis for gastrinoma. World J Surg 26: 1291–1296.
- 17 Hagi T, Hosoda Y, Komoto I, Uemoto S, Hijioka S, et al. (2017) A primary hepatic gastrinoma accompanied by hyperplasia of multi-nodular Brunner’s glands. Surg Case Rep 3: 118.
- 18 Thom AK, Norton JA, Doppman JL, Miller DL, Chang R, et al. (1992) Prospective study of the use of intraarterial secretin injection and portal venous sampling to localize duodenal gastrinomas. Surgery 112: 1002–1008.
- 19 Dhillo WS, Jayasena CN, Jackson JE, Lynn JA, Bloom SR, et al. (2005) Localization of gastrinomas by selective intra-arterial calcium injection in patients on proton pump inhibitor or H2 receptor antagonist therapy. Eur J Gastroenterol Hepatol 17: 429–433.
- 20 Isenberg JI, Walsh JH, Passaro E Jr, Moore EW, Grossman MI (1972) Unusual effect of secretin on serum gastrin, serum calcium, and gastric acid secretion in a patient with suspected Zollinger-Ellison syndrome. Gastroenterology 62: 626–631.
- 21 Jensen RT, Cadiot G, Brandi ML, de Herder WW, Kaltsas G, et al. (2012) ENETS consensus guidelines for the management of patients with digestive neuroendocrine neoplasms: functional pancreatic endocrine tumor syndromes. Neuroendocrinology 95: 98–119.
- 22 Itami A, Kato M, Komoto I, Doi R, Hosotani R, et al. (2001) Human gastrinoma cells express calcium-sensing receptor. Life Sci 70: 119–129.
- 23 Ray JM, Squires PE, Curtis SB, Meloche MR, Buchan AM (1997) Expression of the calcium-sensing receptor on human antral gastrin cells in culture. J Clin Invest 99: 2328–2333.
- 24 Tonelli F, Fratini G, Nesi G, Tommasi MS, Batignani G, et al. (2006) Pancreatectomy in multiple endocrine neoplasia type 1-related gastrinomas and pancreatic endocrine neoplasias. Ann Surg 244: 61–70.
- 25 Thompson SM, Vella A, Service FJ, Grant CS, Thompson GB, et al. (2015) Impact of variant pancreatic arterial anatomy and overlap in regional perfusion on the interpretation of selective arterial calcium stimulation with hepatic venous sampling for preoperative localization of occult insulinoma. Surgery 158: 162–172.
- 26 Morera J, Guillaume A, Courtheoux P, Palazzo L, Rod A, et al. (2016) Preoperative localization of an insulinoma: selective arterial calcium stimulation test performance. J Endocrinol Invest 39: 455–463.
- 27 Hayashi T, Honda H, Yasumori K, Kawashima A, Kaneko K, et al. (1995) Selective intra-arterial injection of calcium for localization of insulinomas: proposed new criteria. Nihon Igaku Hoshasen Gakkai Zasshi 55: 952–956 (In Japanese).
- 28 Guettier JM, Kam A, Chang R, Skarulis MC, Cochran C, et al. (2009) Localization of insulinomas to regions of the pancreas by intraarterial calcium stimulation: the NIH experience. J Clin Endocrinol Metab 94: 1074–1080.
- 29 Ueda K, Ito T, Kawabe K, Lee L, Fujiyama T, et al. (2017) Should the selective arterial secretagogue injection test for insulinoma localization be evaluated at 60 or 120 seconds? Intern Med 56: 2985–2991.
- 30 Braatvedt G, Jennison E, Holdaway IM (2014) Comparison of two low-dose calcium infusion schedules for localization of insulinomas by selective pancreatic arterial injection with hepatic venous sampling for insulin. Clin Endocrinol (Oxf) 80: 80–84.



