抄録
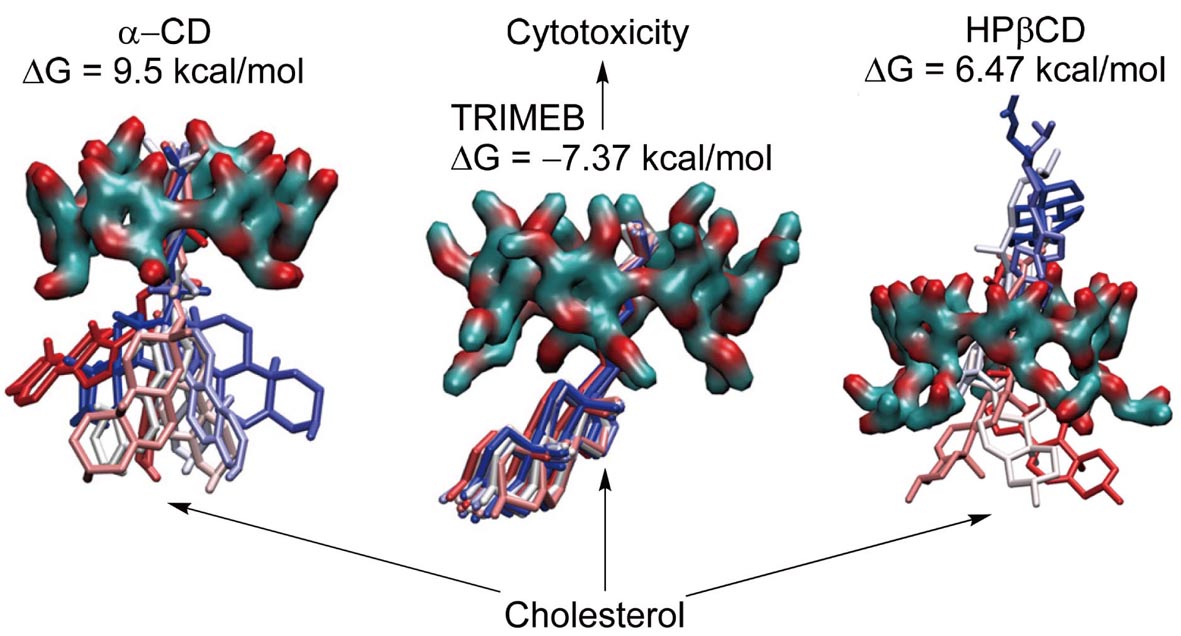
In this study, we investigated the cytotoxic effects of unmodified α-cyclodextrin (α-CD) and modified cyclodextrins, including trimethyl-β-cyclodextrin (TRIMEB) and hydroxypropyl-β-cyclodextrin (HPβCD), on immortalized murine microvascular endothelial (cEND) cells of the blood-brain barrier (BBB). A CellTiter-Glo® viability test, performed on the cEND cells showed significant differences among the different cyclodextrins. After 24 hr of incubation, TRIMEB was the most cytotoxic, and HPβCD was non-toxic. α-CD and TRIMEB exhibited greater cytotoxicity in the Dulbecco’s modified Eagle’s medium than in heat-inactivated human serum indicating protective properties of the human serum. The predicted dynamic toxicity profiles (Td) for α-CD and TRIMEB indicated higher cytotoxicity for these cyclodextrins compared to the reference compound (dimethylsulfoxide). Molecular dynamics simulation of cholesterol binding to the CDs suggested that not just cholesterol but phospholipids extraction might be involved in the cytotoxicity. Overall, the results demonstrate that HPβCD has the potential to be used as a candidate for drug delivery vector development and signify a correlation between the in vitro cytotoxic effect and cholesterol binding of cyclodextrins.