-
69 巻 (2021) 12 号 p. 1170-1178Development of Novel Methodology Using Diazo Compounds and Metal Catalysts もっと読む編集者のコメント
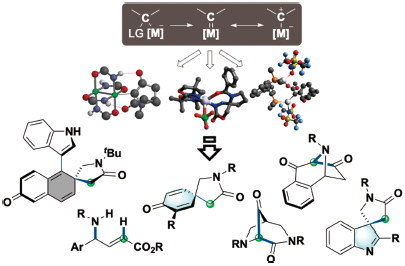
The author focuses on expanding metal-carbene chemistry via developing novel reactions, mechanistic analysis based on computational predictions, and synthetic demonstrations for pharmaceutically essential molecules. The findings include that silver-carbenes possess unusual chemical properties, leading to unique chemoselectivities and regioselectivities with stereocontrol. By utilizing the exceptional reactivities, the enantioselective reactions involving silver-carbenes were achieved in studies regarding arene dearomatization. A novel type of insertion reactions was also developed using a designed Rh2(NHCOtBu)4 catalyst, assembling a series of nitrogen-bridged heterocycles. The author believes that the obtained mechanistic profiles could serve as bases for designing novel reactions and advanced catalysis for pharmaceuticals.
-
69 巻 (2021) 12 号 p. 1195-1199Synthesis and Evaluation of Quinone Derivatives for Activity against Trypanosome cruzi もっと読む編集者のコメント
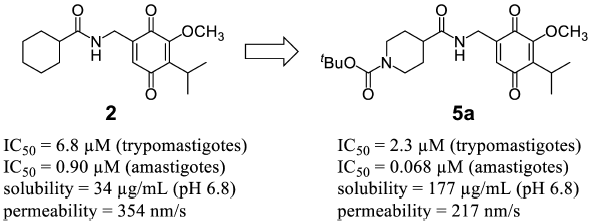
Chagas disease is a neglected tropical disease caused by the protozoan parasite Trypanosoma cruzi and an important health problem in rural areas of Latin American countries. Safe drugs with high clinical efficacy have been urgently needed. Authors synthesized quinone compounds and evaluated their in vitro antitrypanosomal activity against trypomastigotes and amastigotes of Trypanosoma cruzi. In addition, solubility and membrane permeability were measured to confirm whether compounds with promising antiprotozoal activity had physicochemical properties compatible with oral administration. Among the synthesized compounds, 5a showed adequate antiprotozoal activity, solubility, and high membrane permeability.
-
編集者のコメント
The authors rationally designed a series of curvature-sensing peptides, which could selectively detect bacterial extracellular vesicles (EVs) in cultured media. The most sensitive peptide nFAAV5, in which the N-terminal region of the previosly reported curvature-sensing peptide FAAV was modified, was applicable for quantification of changes in the amount of EVs even in the presence of the EV-secretory bacterial cells. Therefore, the novel peptide is useful to EV studies as a potent EV-sensing peptide.
-
編集者のコメント
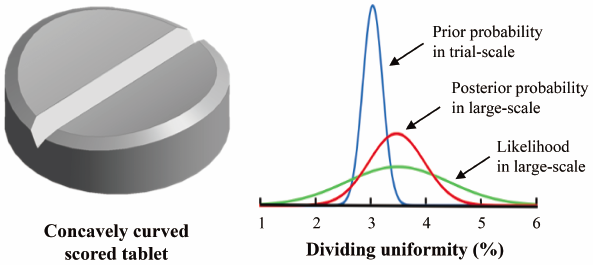
In view of self-medication, it is valuable to develop patient-friendly scored tablets that possess dividing uniformity. Preparation conditions for a concavely curved scored tablet were optimized, employing a design of experiment and a response surface method incorporating a thin-plate spline interpolation, and a bootstrap resampling technique. To make it possible to scaleup the optimal solution estimated on a trial-scale, a Bayesian estimation was applied. Credible ranges of critical responses in large-scale manufacturing were successfully estimated as a posterior probability from the trial-scale experiment as a prior probability.
-
69 巻 (2021) 11 号 p. 1104-1109Bivalent β-Carbolines Inhibit Colorectal Cancer Growth through Inducing Autophagy もっと読む編集者のコメント
Autophagy plays diverse functional roles in various stages of tumor, and numerous drugs targeting autophagy in in-vitro and in-vivo models for colorectal cancer (CRC) have been reported. In search for novel and effective drugs with minimal cytotoxicity to reduce the mortality rate of CRC, the authors designed and synthesized a series of alkyl diamine linked bivalent β-carbolines, and compound 8 was found to be the most potent antitumor agent against human colon carcinoma cell lines with the IC50 value of less than 5 μM. The mechanism of action study revealed that compound 8 reduced growth of colon cancer cells by inducing autophagy.
-
69 巻 (2021) 11 号 p. 1110-1122Discovery of DS68702229 as a Potent, Orally Available NAMPT (Nicotinamide Phosphoribosyltransferase) Activator もっと読む編集者のコメント
Nicotinamide phosphoribosyltransferase (NAMPT) catalyzes the rate-limiting step of the NAD+ salvage pathway. Since boosting NAD+ has positive effects on metabolic regulation, activation of NAMPT is an attractive therapeutic approach for the treatment of various diseases, including type 2 diabetes and obesity. Optimization of our previous lead compound led to identification of DS68702229, a potent NAMPT activator with good oral exposure. Oral administration of DS68702229 to high-fat diet-induced obese mice elicited NAD+ level increase in various tissues, ultimately leading to continuous and significant body weight reduction. These observations indicate that DS68702229 is a promising anti-obesity drug candidate.
-
69 巻 (2021) 10 号 p. 931-944Site-Selective Molecular Transformation: Acylation of Hydroxy Groups and C–H Amination もっと読む編集者のコメント
Control of site selectivity, selectivity among several identical functional groups, in molecular transformation has become a fundamental issue in synthetic organic chemistry owing to the potential utility for selective modification of biologically active multi-functionalized molecules. In the review article, the authors’ research related to site selectivity in two types of transformations, namely, the acylation of hydroxy groups and C-H amination, is summarized. In catalyst-controlled site-selective transformation, the catalysts choose the reaction point where they feel most comfortable.
-
編集者のコメント
A variety of topics have been reviewed from the view point of analytical chemistry. The topics include: self-reproducing vesicles led by molecular transformation, characterization and physiological roles of oxidized phospholipids, advantages of small fish as a carcinogenesis model, separation techniques in supercritical fluid chromatography, drug-antibody ratio determination of antibody-drug conjugates, and membrane-based rapid test reagents for point of care testing. All these reviews will provide indispensable information based on analytical chemistry that supports and evolves pharmaceutical sciences as well as biology and medicine.
-
69 巻 (2021) 10 号 p. 1005-1009Nitroxyl Radical/Copper-Catalyzed Electrooxidation of Alcohols and Amines at Low Potentials もっと読む編集者のコメント
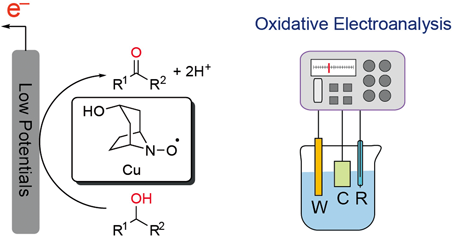
Accurate quantitative and qualitative analysis of biological and clinical samples is important, and rapid and simple analytical methods are demanded. The authors investigate an electrochemical detection of alcohols and amines by cyclic voltammetry at low potential using nortropine N-oxyl (NNO) and a copper salt as catalysts. As a result, although the concentration that could be quantified with good accuracy was limited in both the detection of alcohols and the detection of amines, a correlation was observed between the current and the concentration.
-
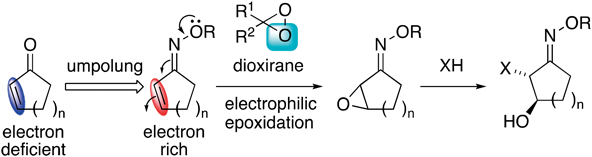
69 巻 (2021) 10 号 p. 1010-1016Electrophilic Epoxidation of α,β-Unsaturated Oximes with Dioxiranes and Ring Opening of the Epoxides もっと読む編集者のコメント
α,β-Unsaturated carbonyl compounds undergo nucleophilic reaction at electron-deficient carbon-carbon double bonds, as well as cycloaddition with dienes or 1,3-dipoles having a high HOMO energy levels. This character of the enones and enals can be transformed from electron-deficient to electron-rich by conversion to the corresponding oximes because of the electron-donating ability of an electron pair (umpolung). This paper describes that α,β-unsaturated oximes underwent electrophilic epoxidation with in-situ-generated dimethyldioxirane to give the corresponding epoxides in good yields. Nucleophilic ring-opening reactions of the epoxides afforded α-substituted products. Shi asymmetric epoxidation of the oximes proceeded with moderate asymmetric selectivity.
-
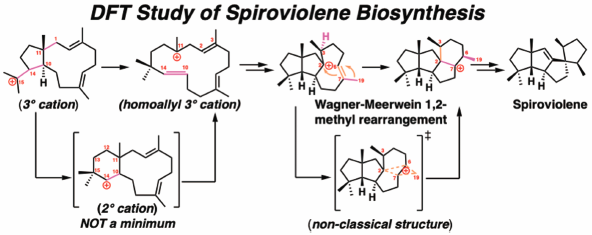
69 巻 (2021) 10 号 p. 1034-1038Theoretical Study on the Mechanism of Spirocyclization in Spiroviolene Biosynthesis もっと読む編集者のコメント
Authors uses computational chemistry to scrutinize the biosynthetic mechanism of a diterpenoid, spiroviolene, which features a 5/5/5/5 tetracyclic system with a spirocyclic skeleton. Terpenes/terpenoids are biosynthesized via multiple stable carbocation intermediates, such as tertiary, allyl, and cyclopropylcarbinyl cations. However, formation of secondary carbocations have been proposed in the spiroviolene biosynthesis in earlier reports. On the basis of DFT calculations, authors propose a new 16-step carbocation cascade leading to the spiroviolene skeleton. This cascade bypasses the formation of unstable secondary carbocations by breaking the adjacent C–C bond to form a more stable tertiary carbocation and by Wagner-Meerwein 1,2-methyl rearrangement.
-
編集者のコメント
Three hybrid catalysts have been designed based on the reaction mechanism of enzymes. These catalysts using non-covalent intermolecular interactions exhibit remarkable catalytic performance for the activation of α,β-unsaturated amides and carboxylic acids as well as the dynamic kinetic resolution of a racemic electrophile in an SN2-type reaction. These innovative catalytic systems allow the asymmetric synthesis of biologically important molecules such as atorvastatin, beraprost, sitagliptin, and avenaol.
-
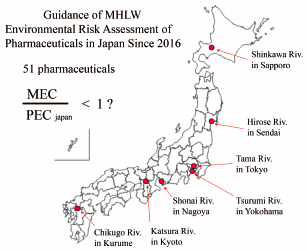
69 巻 (2021) 9 号 p. 840-853Environmental Risk Assessment of Active Human Pharmaceutical Ingredients in Urban Rivers in Japan もっと読む編集者のコメント
In Japan, environmental risk assessments (ERA) for human pharmaceuticals in aquatic environments have barely begun. This paper reports the first study assessing the validity of ERA guidance issued by the Ministry of Health, Labour, and Welfare in 2016 for new medicinal products in the environment, using selected pharmaceuticals already on the market. The accuracy of predicted environmental concentrations was evaluated by measuring environmental concentrations in urban rivers. The results will be useful information applicable to ERA for new medicinal products, and development of future ERA standards in Japan.
-
編集者のコメント
PIM kinases are attractive therapeutic targets for the treatment of multiple hematological malignancies. In search for novel PIM kinase inhibitors, the authors designed and synthesized a series of 5-benzylidene-2-thioxothiazolidin-4-one derivatives with various substitutions at the pyrazine rings. Several compounds exhibited subnanomolar to single-digit nanomolar potencies against all three PIM isoforms and showed anti-proliferative activities against human leukemic EOL-1 and MOLM-16 cell lines. The mechanism of action study of compound 17 clearly revealed that a pan-PIM inhibitor reduced phosphorylation of downstream substrates of PIM kinase such as BAD and 4EBP1 in a dose-dependent manner.
-
編集者のコメント
Understanding the dissociation behavior of cocrystals is an essential task in particular during manufacturing process of the solid dosage forms containing cocrystals as active pharmaceutical ingredients (APIs). The authors have established the promising technique with low-frequency Raman spectroscopy as a monitoring tool for molecular state of APIs during wet granulation, which can detect the dissociation of cocrystals successively and the rate calculated from the spectra enabled to evaluate the stable cocrystal and the formulation with various disintegrants. These findings would help researchers to consider developable pharmaceuticals containing cocrystals.
-
編集者のコメント
An all-carbon quaternary stereocenter is found in many biologically and pharmaceutically active compounds, in spite of being one of the most difficult structures to construct in the field of synthetic organic chemistry. In the present study, we were able to construct all-carbon quaternary stereocenters in an acyclic system by utilizing a face-selective alkylation of enolate intermediates derived from an asymmetric Michael addition reaction of a chiral lithium amide with trisubstituted α,β-unsaturated esters. Moreover, the obtained Michael adduct was able to be converted to the β-amino ester having the all-carbon quaternary stereocenter by an oxidative deprotection.
-
編集者のコメント
The cover figure schematically illustrates the divergent synthesis of various nitrogen-containing heteroaromatic marine natural products by benzyne-mediated cyclization / functionalization cascade reactions. The figure in the center is a slot machine, which represents the flask used in the Cascade reaction, showing that several coins with the structure of nitrogen-containing heteroaromatic complex marine natural products are popping out when three numbers 7 hit at the same time.
-
編集者のコメント
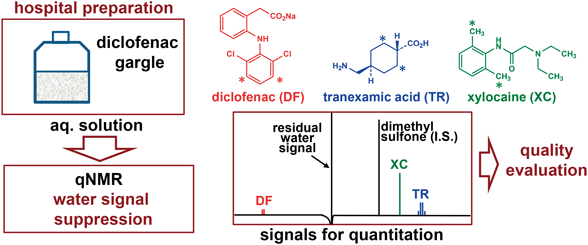
Hospital preparations are frequently prepared in Japanese hospitals, but quality evaluation is not necessarily sufficient. In this article, quantitative 1H NMR spectroscopy (qNMR) was applied to the quality evaluation of diclofenac gargles as an example of a hospital preparation. Aliquots of the gargles were analyzed by qNMR using an internal standard method, where the medicinal ingredients in the preparations were accurately and precisely determined, with the water signal being effectively suppressed by water suppression enhanced through T1 effects (WET). The quality and stability of the gargles were elucidated based on the results. As such, qNMR with WET is potentially useful for the quality control of various aqueous preparations because of its simplicity and convenience.
-
編集者のコメント
The authors have developed the novel composite ordered-mixed (OM) particles containing fine drug-particles. The composite OM particles were found to be double-structured spherical particles consisting of finely-milled particles layered around the core particles. It was also found that they have excellent characteristics from both pharmaceutical and micromeritic perspectives and could overcome manufacturing problems of solid dosage forms. Such unique OM particles could be precisely designed by ultra cryo-milling in liquid nitrogen and subsequent mechanical powder processing which would be a potential platform approach to develop the oral dosages (tablet, capsule, granule) with improved dissolution property of poorly water-soluble drugs.
-
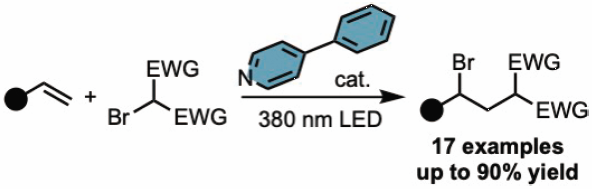
69 巻 (2021) 8 号 p. 796-801Photoinduced Atom Transfer Radical Addition Reaction of Olefins with α-Bromo Carbonyls もっと読む編集者のコメント
The authors developed ATRA reactions of olefins with bromomalonate ester catalyzed by an in situ-formed halogen-bonding complex. The use of 4-phenylpyridine as the halogen-bonding acceptor, the desired reaction proceeded well under external irradiation of 380 nm LED light to furnish the corresponding ATRA reaction product. The substrate activation through the halogen bonding from the CT complex is a novel activation method that can be used for challenging synthetic routes since halogen bond acceptors such as pyridine are relatively inexpensive and readily available. This approach offers a new strategy for other classes of compounds for photocatalysis, including alkyl halides with amines or phosphine.