-
69 巻 (2021) 6 号 p. 516-525Development of Catalytic Reactions for Precise Control of Chemoselectivity もっと読む編集者のコメント
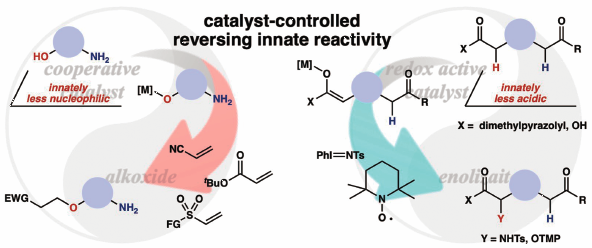
A cooperative catalyst comprising a soft Lewis acid/hard Brønsted base enabled chemoselective activation of a hydroxyl group over an amino group. The present chemoselective catalysis could be applicable for a variety of amino alcohols, including pharmaceuticals, without requiring a tedious protection-deprotection process. Chemoselective enolization and subsequent α-functionalization of carboxylic acid derivatives were also achieved by a redox active catalyst through the radical process, providing unnatural α-amino/hydroxy acid derivatives bearing a complex carbon framework and a diverse set of functionalities. The present chemoselective catalysis described herein offers new opportunities to expand the chemical space for innovative drug discovery research.
-
69 巻 (2021) 6 号 p. 526-528Fluorescent-Oxaboroles: Synthesis and Optical Property by Sugar Recognition もっと読む編集者のコメント
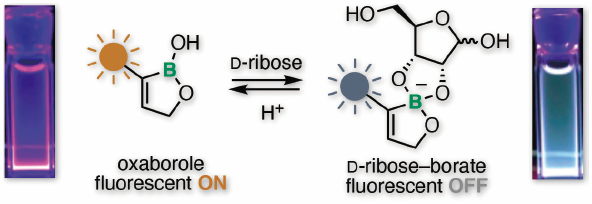
The optical property of fluorescent unit-conjugated aliphatic oxaboroles has been investigated in this featured article. The authors described the synthesis of fluorescent-oxaboroles by originally developed methods and the optical behavior of the boroles with various sugars. The oxaboroles provide good fluorescence quantum yields and selective recognition toward D-ribose and D-ribose-containing molecules. The molecular recognition induced significant fluorescence quenching. The authors also revealed the positive correlation between the LUMO energy of the oxaborole and the relative fluorescence intensity. The property of the boroles showed the possibility of the boron-based NAD sensor probe.
-
編集者のコメント
Quantitative evaluation for bitterness of pediatric medicine is essential for adherence. The authors proposed criteria, change in concentration-dependent potential (CCDP), dose-response slope of the sensor outputs of active pharmaceutical ingredients measured by an artificial taste sensor, which is new and useful bitterness evaluation index for 48 pediatric medicines from the recent edition of the WHO model list of essential medicines for children (7th ed., 2019). CCDP by individual basic bitterness sensor well correlated to various physicochemical factors related hydrophilicity and hydrophobicity. Therefore, CCDP proved to be useful as a bitterness evaluation index of APIs in pediatric medicines.
-
編集者のコメント
Novel 25 derivatives of puberulic acid were synthesized by utilizing the authors’ total synthetic route. According to previous structure-activity relationships information, they focused on the carboxylic acid moiety in puberulic acid and converted it into the corresponding esters, amides and ketones. Antimalarial evaluations in vitro and in vivo of these derivatives were carried out and revealed detailed SAR information. It was clarified that several ester and ketone derivatives conserve strong antimalarial activity similar to that of puberulic acid without any obvious toxicity in oral dose of 15 mg/kg.
-
編集者のコメント
This present review aims to achieve a comprehensive and up-to-date investigation in the phytochemistry and pharmacology of Rosa laevigata Michx.. To date, phytochemical investigation has exposed the presence of triterpenoids, flavonoids, tannins, ligands and polysaccharides in this plant. The crude extracts and the purified compounds have demonstrated various pharmacological effects in vitro and in vivo. In conclusion, the chemical profiles together with the modern pharmacological properties have been adequately summarized. These evidences have revealed this plant to be a valuable source for therapeutic foodstuff and more attention should be paid to a better utilization of this plant.
-
69 巻 (2021) 5 号 p. 444-446Novel Cyclohexyl Meroterpenes Produced by Combinatorial Biosynthesis もっと読む編集者のコメント
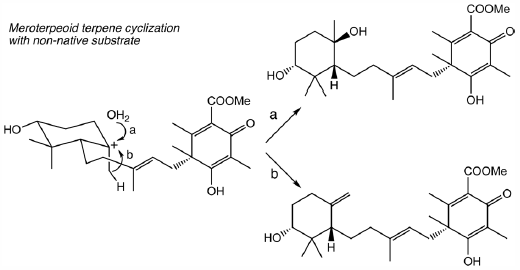
Structurally diverse fungal meroterpenoids are promising drug seed compounds. To obtain unnatural, novel meroterpene scaffolds, the authors tested combinatorial biosynthesis by co-expressing functionally distinct terpene cyclase (TPC) genes, pyr4, ascF, andB, or cdmG, with the biosynthetic genes for the production of a TPC substrate, (10’R)-epoxyfarnesyl-dimethylorsellinic acid-3,5-methyl ester, in Aspergillus oryzae NSAR1 as a heterologous host. As a result, all of the tested TPCs afforded the same two novel mono-cyclization products. This study provides important information on the substrate scope of the TPCs, and will contribute to the production of unnatural, novel molecules for future drug discovery.
-
編集者のコメント
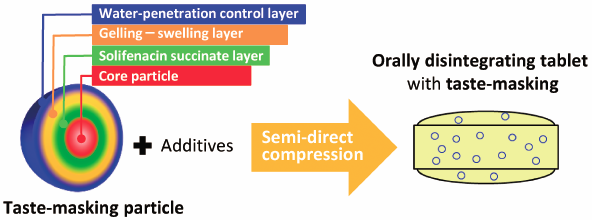
The authors developed solifenacin succinate functional drug particles embedded in a gelling–swelling layer (PEGS) made of a three-layer structure consisting of a core drug layer, a gelling–swelling layer, by which both adequate taste-masking of drug and rapid drug elution were successfully achieved. They also optimized incorporation of above PEGS into orally disintegrating tablets (ODTs), by usage of a fibre-shaped microcrystalline cellulose with poor fluidity, which improved the content uniformity of the ODTs, as the crystal fibres became entangled with the PEGS and other additives. Overall, this taste masking system is so unique and propose new platform for taste masking formulation.
-
編集者のコメント
A methanol extract from the underground part of Calanthe discolor Lindl. (Orchidaceae) demonstrated significant proliferative activity against human hair follicle dermal papilla cells (HFDPC). Through bioassay-guided separation of the extract, a new indole glycoside named 6′-O-b-D-apiofuranosylindican was isolated along with six known compounds including three indole glycosides. Among the isolates, three indole glycoside such as 6′-O-b-D-apiofuranosylindican, glucoindican, and calanthoside showed significant proliferative activity. Furthermore, these active indole glycosides upregulated the expression of VEGF and FGF-7 mRNA and protein in HFDPC, which could be the mechanism of their proliferative activity.
-
編集者のコメント
This manuscript reports aerobic oxidation of primary alcohols into aldehydes with nitroxyl radical/copper catalysis. The reaction method reported herein features that the optimum nitroxyl radical/copper salt catalyst combinations differ depending on the substrate. To express this feature, the catalytic reaction field is represented as a circus tent with three shapes of windows, namely, circle, triangle, and square. Copper plates with different nitroxyl radicals are placed under each of the windows, which indicates different nitroxyl radical/copper salt catalyst combinations. Different alcohol substrates are represented in different forms, namely, sphere, tetrahedron, cube. The sphere, tetrahedral, and cubic alcohols pass through the circular, triangular, and square windows, respectively, to be oxidized into aldehydes.
-
編集者のコメント
Non-canonical amino acid derivatives and the peptides containing such structures are regarded as the attractive scaffold of novel drug candidates. In this featured article, the author describes the development of novel methodologies for the synthesis of them, involving (1) catalytic asymmetric 1,2-addition of N-2-nitrophenylsulfenyl imines, (2) direct chemical transformation such as ring-closing metathesis or asymmetric Friedel–Crafts reaction using hydrophobic anchor-tagged peptidic substrates. The development of (3) novel recyclable organocatalyst by combining the catalytic motif and hydrophobic anchor is also described.
-
編集者のコメント
The authors report novel positive allosteric modulators (PAMs) of M3 muscarinic acetylcholine receptor (mAChR) with N-pyrimidyl/pyridyl-2-thiazolamine scaffold. The structureactivity relationships study was rationalized using conformational analyses based on intramolecular interactions, demonstrating that a unique sulfur-nitrogen nonbonding interaction in the N-pyrimidyl/pyridyl-2-thiazolamine moiety is essential for activity. Newly identified compound 3g showed potent in vitro PAM activity for the M3 mAChR with excellent subtype selectivity. Compound 3g also showed a distinct pharmacological effect on isolated smooth muscle tissue from rat bladder and favorable pharmacokinetics profiles, suggesting its potential as a chemical tool for probing the M3 mAChR in further research.
-
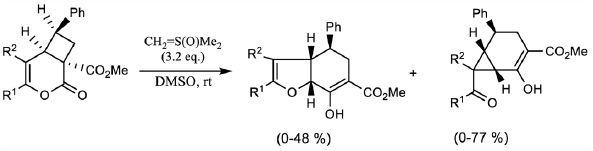
69 巻 (2021) 4 号 p. 391-399Reaction of 3-Oxa-2-oxobicyclo[4.2.0]oct-4-ene-1-carboxylate with Dimethylsulfoxonium Methylide もっと読む編集者のコメント
Cycloalkanes with small ring sizes (such as cyclopropanes and cyclobutanes) constitute a basic structural framework of a wide range of natural products. Cycloalkanes play an important role in organic synthesis as they can take part in various types of reactions. The high reactivities can be attributed to the angle and torsional strains. The authors have been interested in the reactivities of small-ring compounds. The reaction between 3-oxa-2-oxobicyclo[4.2.0]oct-4-ene-1-carboxylate and dimethylsulfoxonium methylide afforded 3a,4,5,7a-Tetrahydro-7-hydroxy-benzofuran-6-carboxylate and/or 2-hydroxybicyclo[4.1.0]hept-2-ene-3-carboxylate. A plausible reaction mechanism has been shown.
-
編集者のコメント
Oral mucositis is one of the most common adverse effects of radiation and chemotherapy in treatments of cancers. A mouthwash containing drugs is often used for the prevention and treatment of oral mucositis. In this study, the authors indicated the disappearance time and palatability in the oral cavities of healthy volunteers in placebo foam formulations. In addition, foam formulations of drugs for use as a prevention and treatment of oral mucositis were prepared. This is the first report in regard to foam formulations containing drugs for oral mucositis, and these formulations could be potentially useful in treatments of cancers.
-
69 巻 (2021) 3 号 p. 237-245Impact of COVID-19 Outbreak on the Long-Range Transport of Common Air Pollutants in KUWAMS もっと読む編集者のコメント
To evaluate the impact of air pollutants long-range transported from the Asian continent on the atmospheric environment in Japan, a background site in the top of Noto peninsula, the Kanazawa University Wajima Air Monitoring Station (KUWAMS), has been used for continuous observation since 2004. Among the numerous reports on air pollution during the COVID-19 pandemic, this article proves for the first time that the implementation of the lockdown policy in China reduced both the air pollution in domestic China and that involved in the long-range transport to KUWAMS, including fine (PM2.5) and ultrafine particulates (PM1.0), Organic Carbon (OC) and Element Carbon (EC).
-
編集者のコメント
The site-selective transformation provides a powerful strategy for synthesizing complex and diverse multi-functionalized molecules. In this article, the first total syntheses of O-3’-acyl α-bisabolol β-D-fucopyranoside natural products and their analogues by using boronic acid-catalyzed site-selective acylation in the final stage is described. This approach allows us to construct compound libraries for the purpose of structure–activity relationship studies. The cover art illuminates key feature of this work by characteristic chemical transformation.
-
編集者のコメント
This paper describes the monitoring analysis of perfluoroalkyl substances (PFAS), 9-chlorohexadecafluoro-3-oxanonane-1-sulfonate (F-53B) and dodecafluoro-3H-4,8-dioxanonanoate (ADONA) in drinking bottle water, tea and juice samples. Liquid chromatography coupled with tandem mass spectrometry and solid phase extraction showed that recovery values were 80.4–118.8% with RSD ≤ 0.6%, and application is that investigation of non-contamination of PFAS, F-53B and ADONA in these drinking samples from Japanese markets. Using the developed method, it suggests that these screening assay of PFAS in various food samples is more widely distributed for human exposure assessment.
-
編集者のコメント
Resin glycosides are well known as the purgative ingredients, which are commonly found in plants belonging to the Convolvulaceae family. The seeds of Ipomoea muricata are used as a laxative and carminative folk medicine. The authors isolated two new genuine resin glycosides with macrolactone structures from the seeds of I. muricata. Their structures were determined based on spectroscopic data including 1D- and 2D-NMR spectra and MS and examination of the component organic and glycosidic acids generated by alkaline hydrolysis of the crude resin glycoside fraction.
-
69 巻 (2021) 2 号 p. 155-177Chemical Research on Antitumor Isoquinoline Marine Natural Products and Related Compounds もっと読む編集者のコメント
The biologically active, naturally occurring 1,2,3,4-tetrahydroisoquinoline-quinone (THIQ) family members isolated from Actinomycetes and marine organisms have been studied thoroughly over the past five decades. Among them, renieramycins and ecteinascidins have attracted interest due to their fantastic structures and meager availability in nature as well as their potent antitumor profiles. This review mainly presents the author’s contributions during 1999–2019 to the field of research supported by the JSPS Asia and Africa Science Platform Program. The author has provided important information for drug discovery through the synthetic supply of natural products and biological evaluation of various derivatives.
-
編集者のコメント
In this study, authors used statistical methods to examine whether the 13C-NMR spectral data from 11 methoxyflavones could be used to correlate their structural features and biological activities. The 13C-NMR spectral data for the 15-carbon flavonoid skeleton in eleven methoxyflavones isolated from Kaempferia parviflora (Zingiberaceae) were processed by principal component analysis (PCA). Based on the PCA score plots, the methoxyflavones were categorized into three groups according to their structural features. Pearson’s correlation analysis revealed that a structural design which lowers the 13C-NMR chemical shift at C-10 would be important for the development of cytotoxic compounds. This approach offers a new strategy for directing structure/activity relationship research.
-
編集者のコメント
Adenylation (A) domain functions as a gatekeeper, by selecting building blocks from a pool of 20 proteinogenic amino acids, non-proteinogenic amino acids, and aryl acids. Therefore, the A-domain is an attractive target for the reprogramming of non-ribosomal peptide synthetase machinery for the production of non-native peptides. Author determined the substrate profiles of a reprogrammed A-domain toward naturally occurring aminobenzoic acid substrates, synthesized sulfamoyloxy-linked acyl-AMP analogues, and conducted binding and modeling studies of them toward the reprogrammed A-domain. The study outcomes would help a better understanding of the unique substrate binding site of the reprogrammed A-domain, facilitating reprogramming of the A-domain.