Polycythemia vera is a myeloproliferative neoplasm that can cause opposite complications: thrombosis and bleeding. Since arterial or venous thrombosis is often fatal, prevention of thrombogenesis is the main treatment measure for polycythemia vera, both in daily life and perioperatively. However, reports of bleeding are less frequent than reports of thrombosis. Herein, we report a case of polycythemia vera in which significant postoperative bleeding was a perioperative complication.
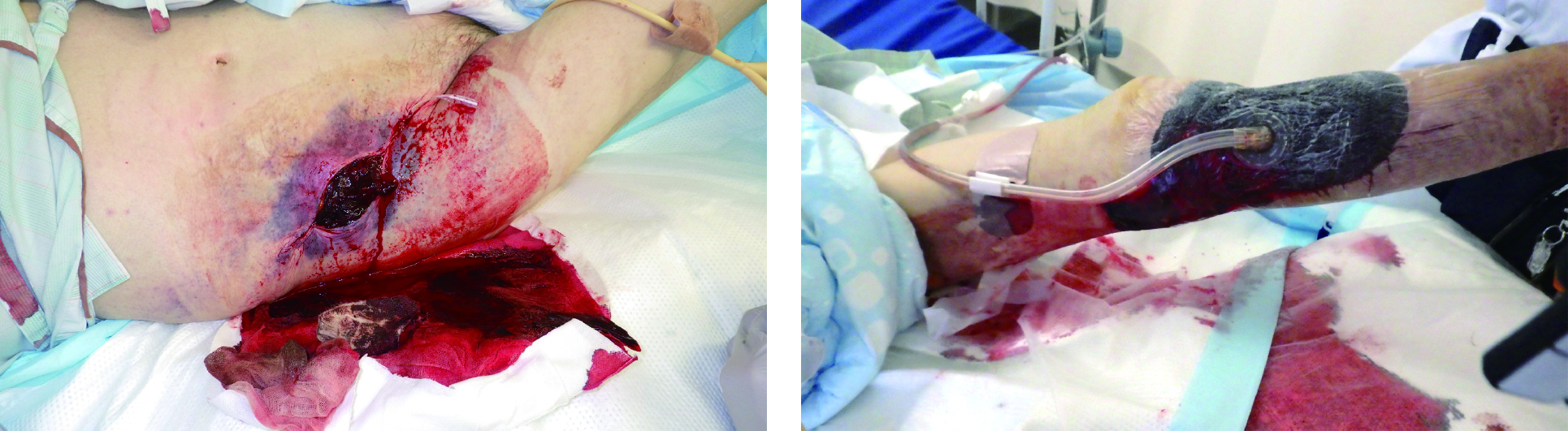
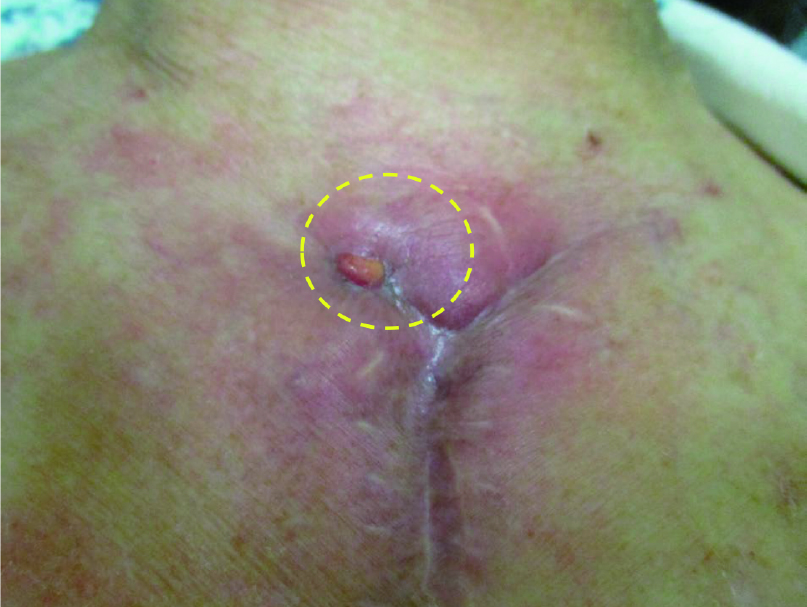
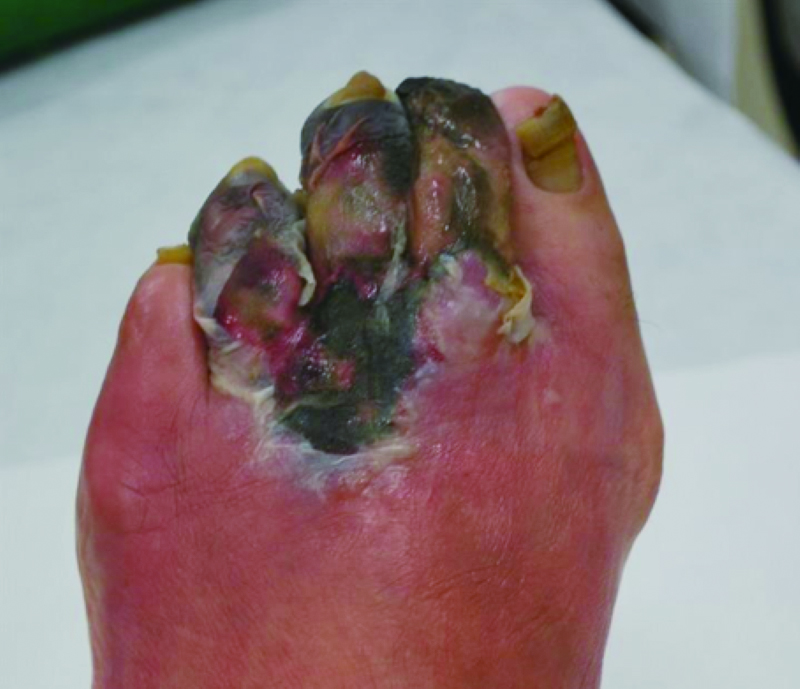
A 72-year-old woman with polycythemia vera had a subcutaneous hematoma after bruising her left lower leg, and was admitted to another hospital. As skin necrosis gradually appeared, the patient was transferred to our hospital. Following debridement and wound bed preparation, we applied a split-thickness skin graft using the right inguinal region as the donor site. After surgery, significant bleeding was observed at the donor and recipient sites, and this persisted until the next morning, which required transfusion of four units of blood. Hemostasis was difficult but finally achieved with strong and firm pressure. The skin graft survived, and the troublesome wound healed.
Regarding the perioperative complications of polycythemia vera, not only thrombosis, but also bleeding should be considered. Strict control of patients' hematocrit and platelet count with phlebotomy and/or hydroxyurea as cytoreduction therapy is essential.
After the surgery, significant bleeding was observed at the donor site (a) and recipient site (b). This required a transfusion of four units of blood.
View full abstract