2016 年 39 巻 9 号 p. 1514-1522
2016 年 39 巻 9 号 p. 1514-1522
In this study, solid lipid nanoparticle (SLN) suspensions were prepared using a base of hard fat with or without ethylcellulose (EC) and polyvinyl alcohols (PVA) and polysorbate (Tween) 60 surfactants. Commercially available PVAs vary in their degree of saponification and polymerization, and the appropriate PVAs to form SLNs from hard fat with or without EC were investigated. A relatively low-saponification-degree PVA was required to reproducibly form SLN suspensions without EC and relatively high-saponification-degree PVAs were suitable for SLNs with EC. The release of morin from SLNs with EC was more sustained than that from SLNs without EC. The maximum plasma concentration (Cmax) of SLNs with and without EC were almost the same, and both were higher than that of a morin suspension. The area under the curve for 0 to 360 min (AUC0–360) of SLNs with EC was increased compared with those of a morin suspension and SLNs without EC. The median diameter of SLNs with EC and a very low-saponification-degree PVA was decreased compared to other formulation, and morin release was more sustained for this formulation. SLNs with EC and a very low-saponification-degree PVA showed higher Cmax and AUC0–360 than SLNs with EC lacking a very low-saponification-degree PVA. The optimized SLNs with EC and a very low-saponification-degree PVA improved bioavailability via increased accessibility to the enterocyte surface by decreased particle size and increased permeation of SLN encapsulated morin through the intestinal membrane by sustained release properties.
Polyphenols are a large and diverse class of compounds that are classified as flavonoids, hydroxycinnamic acids, stilbene derivatives, and polymers.1) Many polyphenols occur naturally in foods, such as beans, cereals, fruits, vegetables, and red wine.2) The biological properties of polyphenols and their role in preventing or ameliorating diseases related to oxidation is attracting increasing interest. Morin (3,5,7,2′,4′-pentahydroxyflavone) is a flavonoid that has been identified in fruits, vegetables, tea, and many Asian medicinal herbs.3) Morin has a number of potentially useful biological activities, including anti-inflammatory, anticancer, antioxidant, antihypertensive, and anticlastogenic responses.4–8)
Unsaturated fatty acids are the most important structural components of biological membranes and help maintain the fluidity of the cellular membrane structure. The peroxidation of unsaturated fatty acids in biological membranes disrupts their structure and function.9,10) Lipid peroxidation has been implicated in the pathogenesis of various diseases, such as coronary heart disease, atherosclerosis, and cancer, as well as the ageing process.11–14)
Morin has been suggested as an inhibitor of lipid peroxidation. However, the absolute bioavailability of morin after a single oral dose is very low (less than 1%), likely because of its low aqueous solubility, P-glycoprotein (P-gp)-mediated efflux, and low intestinal permeability.15–17) Various formulation techniques have been developed to improve the oral bioavailability of morin. However, few studies developing morin formulations have been reported. Encapsulation in solid lipid nanoparticles (SLN) is a technique that can improve oral bioavailability of drugs with poor water solubility.18) SLNs consist of a lipid matrix that is solid at room temperature and dispersed in an aqueous surfactant solution. SLNs trap hydrophobic drugs in the solid matrix, sustain drug release, and prevent premature drug degradation.19)
The present study was undertaken to develop a morin-loaded SLN formulation to improve the oral bioavailability of morin. Hard fat, consisting of triglycerides based on coconut and palm kernel oil, was used as the lipid base. Ethylcellulose (EC) is utilized as a coating agent and a tablet binder that controls drug release from a tablet. We have previously reported that EC dispersed in hard fat constructs a matrix and that a tablet consisting of hard fat, EC and indomethacin sustained drug release.20,21) We investigate the addition of EC to SLN formulations in an attempt to control drug release. Surfactants form the outer shell of the lipid matrix and stabilize colloidal systems. Tween is widely used to stabilize oil–water type emulsions, and SLN formulations using Tween 60 or Tween 80 as emulsifiers have been reported.22–24) SLNs were prepared using Tween 60 or Tween 80 in our preliminary experiment, and the particle size of SLNs containing Tween 60 was smaller than that of SLNs containing Tween 80. Tween 60 was chosen as the emulsifier for subsequent experiments. In this study, a number of polyvinyl alcohols (PVAs) and Tween 60 were used as surfactants. PVAs are nonionic synthetic polymers that exhibit excellent biodegradability. PVAs are used as emulsifiers, colloidal particles stabilizers, adhesives, and coating agents in the textile and paper industries. The biocompatibility, biodegradability, and lack of toxicity favor the broad use of PVAs in medical aplications.25) There are a range of PVAs available that vary in their degree of saponification and polymerization. In this study, the appropriate PVAs for the formation of SLNs from hard fat with or without EC were investigated.
Hou et al. reported that the area under the curve (AUC) and maximum plasma concentration (Cmax) of morin after oral administration of 50 mg/kg to rats were 37- and 27-fold higher, respectively, than those from a dose of 25 mg/kg.26) This increase in bioavailability is attributed to P-gp-mediated efflux. In this study, all the morin doses administered to mice are relatively low (10 mg/kg) to evaluate the effects of formulation without saturating the P-gp-mediated efflux. The preparation of morin-loaded SLNs and their pharmacokinetic parameters after oral administration are evaluated.
Morin was purchased from Kanto Chemical Co., Inc. (Tokyo, Japan). Hard fat (Witocan® H) was supplied by Mitsuba Trading Co. (Tokyo, Japan). PVA was supplied by the Nippon Synthetic Chemical Industry Co., Ltd. (Osaka, Japan). EC45 (about 49% (w/w) ethoxyl basis) and Tween 60 were purchased from Wako Pure Chemical Industries, Ltd. (Osaka, Japan). All other chemicals were obtained commercially at the purest grade available.
Preparation of SLN SuspensionsThe compositions of morin-loaded SLN suspensions are shown in Table 1. Four different PVAs (EG-05P, EG-18P, KL-03, LL-810) were evaluated to establish an appropriate PVA for morin SLN formation. The characteristics of the PVAs are shown in Table 2. The degrees of saponification and viscosity values are those stated by the supplier. The chemical structure of PVA is shown in Fig. 1. Equation 1 was used to calculate the degree of saponification,
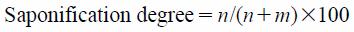 | (1) |
Suspensions of SNLs were prepared as follows. EC and morin were dissolved in ethanol and added to hard fat heated to 60°C. The mixture was agitated and the ethanol was subsequently evaporated from the mixture in vacuo in the presence of desiccating silica gel for 48 h. Tween 60 was added to the dry solid and the mixture was melted and stirred at 50°C. PVA solution (0.4 or 0.8% (w/v)) heated to 50°C was poured into the lipid mixture and stirred at 25000 rpm using a Physcotron® NS-10 homogenizer (Microtect Co., Ltd., Chiba, Japan) for 60 s, followed by sonication using a Smurt NR-50M ultrasonic homogenizer (Microtect Co., Ltd.) for 60 s (H-PVA0.4%, H-PVA0.8%) or 20 s (H-EC-PVA0.4%, H-EC-PVA0.8%). The SLN suspensions were left at 24±1°C for 24 h, centrifuged for 5 min at 1000×g, and the precipitate removed.

| Formulation | Hard fat (mg) | EC45 (mg) | Tween 60 (mg) | Morin (mg) | PVA* (mg) | Water (mL) |
|---|---|---|---|---|---|---|
| H-PVA0.4% | 85.0 | — | 80.0 | 15.0 | 30.0 | 7.5 |
| H-PVA0.8% | 85.0 | — | 80.0 | 15.0 | 60.0 | 7.5 |
| H-EC-PVA0.4% | 83.0 | 2.0 | 80.0 | 15.0 | 30.0 | 7.5 |
| H-EC-PVA0.8% | 83.0 | 2.0 | 80.0 | 15.0 | 60.0 | 7.5 |
* Polyvinyl alcohols used; EG-05P, EG-18P, KL-03.
| PVA | Saponification degree (mol%) | Viscosity (mPa·s) |
|---|---|---|
| EG-05P | 88.0 | 5.5* |
| EG-18P | 87.3 | 18.0* |
| KL-03 | 79.7 | 3.2* |
| LL-810 | 48.7 | 7.7** |
* The viscosity of a 4% (w/v) aqueous solution at 20°C. ** The viscosity of a 10% (w/v) water–methanol (50 : 50) solution at 20°C.
The composition of morin-loaded SLN suspensions containing low-saponification-degree PVA is shown in Table 3. The low-saponification-degree PVA SNL suspensions were prepared as follows. EC, morin and PVA (LL-810) were dissolved in ethanol and added to hard fat heated to 60°C. The mixture was agitated and ethanol evaporated in vacuo in the presence of a desiccating silica gel for 48 h. Tween 60 was added to the dry solid and the mixture was melted and stirred at 50 or 60°C. PVA (EG-05P, 0.8% (w/v)) solution heated to 50 or 60°C was poured into the lipid mixture, stirred at 25000 rpm using a Physcotron® NS-10 homogenizer for 60 s, and sonicated using a Smurt NR-50M ultrasonic homogenizer for 20 or 90 s. The SLN suspensions were gradually cooled by standing at 24±1°C for 24 h or rapidly in ice for 10 min. SLN suspensions were isolated by centrifugation for 5 min (24±1°C samples) or 30 min (ice-cooled samples) at 1000×g, and the precipitate discarded.
| Formulation | Hard fat (mg) | EC45 (mg) | PVA(LL-810)(mg) | Tween 60 (mg) | Morin (mg) | PVA (EG-05P) (mg) | Water (mL) |
|---|---|---|---|---|---|---|---|
| H-EC-PVA(LL-810) | 80.0 | 2.0 | 3.0 | 80.0 | 15.0 | 60.0 | 7.5 |
The ratio of morin in a SLN suspension to the amount of initial morin was used to calculate R%. EE% was determined by calculating the ratio of the morin in the SLN suspension less the free morin to the initial amount of morin. The calculations for R% (2) and EE% (3) are as follows;
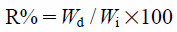 | (2) |
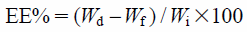 | (3) |
The amount of morin in SLN suspensions was determined using the following method. Methanol was added to a SLN suspension and the mixture was heated to 60°C. After cooling to room temperature, methanol was added to replace any volume lost to evaporation and the mixture was passed through a membrane filter (0.45 µm). The morin concentration in the filtrate was analyzed by HPLC. The amount of free morin was determined by filtering the SLN suspension with an ultrafilter (molecular weight cut-off 10000, pore size 2–3 nm) and analyzing the concentration of morin in the filtrate by HPLC.
Particle Size Distribution and Zeta-Potential AnalysisSLN suspensions were diluted with distilled water and the particle size distribution and the volume median diameter of the samples were measured by a SALD-7100 laser diffraction particle size analyzer (Shimadzu, Kyoto, Japan). SLN suspensions were diluted with Milli-Q water and their zeta potential was determined with a Photal ELS-Zz2 a zeta potential and particle size analyzer (Otsuka Electronics Co., Ltd., Osaka, Japan).
Drug Release PropertiesThe in vitro release of morin from the SLNs was determined by a dialysis membrane method.22,23,27) SLN suspension was dispersed in phosphate buffer (25 mM, pH 6.8) containing 0.1% Tween 80 and transferred to a dialysis bag (molecular weight cut-off 12000–14000, Ieda Trading Corporation, Tokyo, Japan). For comparison, a morin ethanol solution and morin suspension were prepared and dialyzed using the same conditions. To prepare the morin suspension, crude morin was suspended in a 1% sodium carboxymethylcellulose solution. The paddle method of dissolution (as per the Pharmacopoeia of Japan) was used with a NTR-VS6P dissolution tester (Toyama Sangyo Co., Ltd.). The dialysis bag was immersed in phosphate buffer dissolution medium (200 mL, 12.5 mM, pH 6.8 containing 0.2% Tween 80) at 37±0.5°C and the paddle rotated at 100 rpm. At predetermined time intervals, a 1-mL sample of the buffer was taken and replaced with fresh buffer. The morin released from the SLN was quantified by HPLC.
X-ray Powder Diffraction (XRD) AnalysisXRD analysis of freeze-dried SLNs from the suspensions was performed using a 9-kW SmartLab Rigaku diffractometer (Rigaku Corporation, Tokyo, Japan) with a rotating anode at room temperature. The voltage and amperage were set at 45 kV and 200 mA, respectively. Each sample was scanned between 6° and 40° in 2θ with a step size of 0.02° and scan speed of 20°/min.28)
Animal ExperimentsAnimal and Drug AdministrationThe animal protocols used in this study were approved by the issuing committee (the Committee on the Care and Use of Laboratory Animals of Hoshi University), which is accredited by the Ministry of Education, Culture, Sports, Science and Technology of Japan, as conforming with the Guide for the Care and Use of Laboratory Animals (Approval No. 27–048).
Male Jcl : ICR mice were obtained at 5 weeks of age. Mice were housed in temperature-controlled cages under a 12-h light–dark cycle and given free access to water and a normal chow diet for 1 week. Each mouse was fasted for 12 h before drug administration of a single dose of a SLN suspension or morin suspension (10 mg morin/kg) by intragastric gavage. Blood was collected from the postcava under anesthesia at predetermined times (n=3–4 for each time point) after administration and the mice were euthanized after blood sampling. The maximum drug concentration time (Tmax), AUC0–360 and mean residence time (MRT)0–360 values presented were calculated using averages of the measured results.
Plasma Concentration of MorinPlasma samples were isolated from blood by centrifugation at 1000×g for 15 min. The plasma concentrations of morin were determined using HPLC. Plasma samples (200 µL) were dispensed into centrifuge tubes and acetate buffer solution (100 µL, pH 5) and ascorbic acid solution (20 µL, 300 mg/mL) were added, followed by ethyl acetate (300 µL containing ethylparaben 5 µg/mL). The ethyl acetate layer was evaporated under a stream of N2 the sample reconstituted with methanol (100 µL) before HPLC analysis.
HPLC Determination of Morin ConcentrationHPLC was carried out using an LC-6AD pump and a C-R7A chromatopac (Shimadzu) equipped with a Capcell Pak C18 MG II column (4.6×250 mm, Shiseido Co., Ltd., Tokyo, Japan) and a SPD-20AV UV detector (Shimadzu). Chromatography was carried out at 40°C. The injection volume was 20 µL. The mobile phase was acetonitrile–0.2% ortho-phosphoric acid in water (27 : 73, v/v) at a flow rate of 1 mL/min. The detection wavelength was 250 nm. Ethylparaben was used as an internal standard. Calibration curves were obtained by a linear regression analysis of concentrations plotted against peak area.
Statistical AnalysisDifferences in EE% were evaluated by Student’s t-test. Tukey test was performed for differences in zeta potentials. Dunnett’s test was performed to compare the formulations<morin ethanol solution in drug release profiles. A p-value less than 0.05 was considered significantly different.
The R% and EE% of the SLN suspensions are shown in Table 4. R% is the proportion of drug present in the SLN suspensions. The EE% is the proportion of drug incorporated in the SLNs. There was little difference between R% and EE% in the whole SLN suspension, confirming that the majority of the morin was not freely suspended in the supernatant. The level of morin present in the H-PVA0.8% SLN suspensions was higher than the H-EC-PVA0.8% SLN suspensions and this trend was more pronounced between the H-PVA0.4% and H-EC-PVA0.4% preparations. The differences in EE% between H-PVA0.4% and H-PVA0.8% or H-EC-PVA0.4% and H-EC-PVA0.8% were compared and a significant difference was observed between H-EC-PVA0.8%(KL-03) and H-EC-PVA0.4%(KL-03).
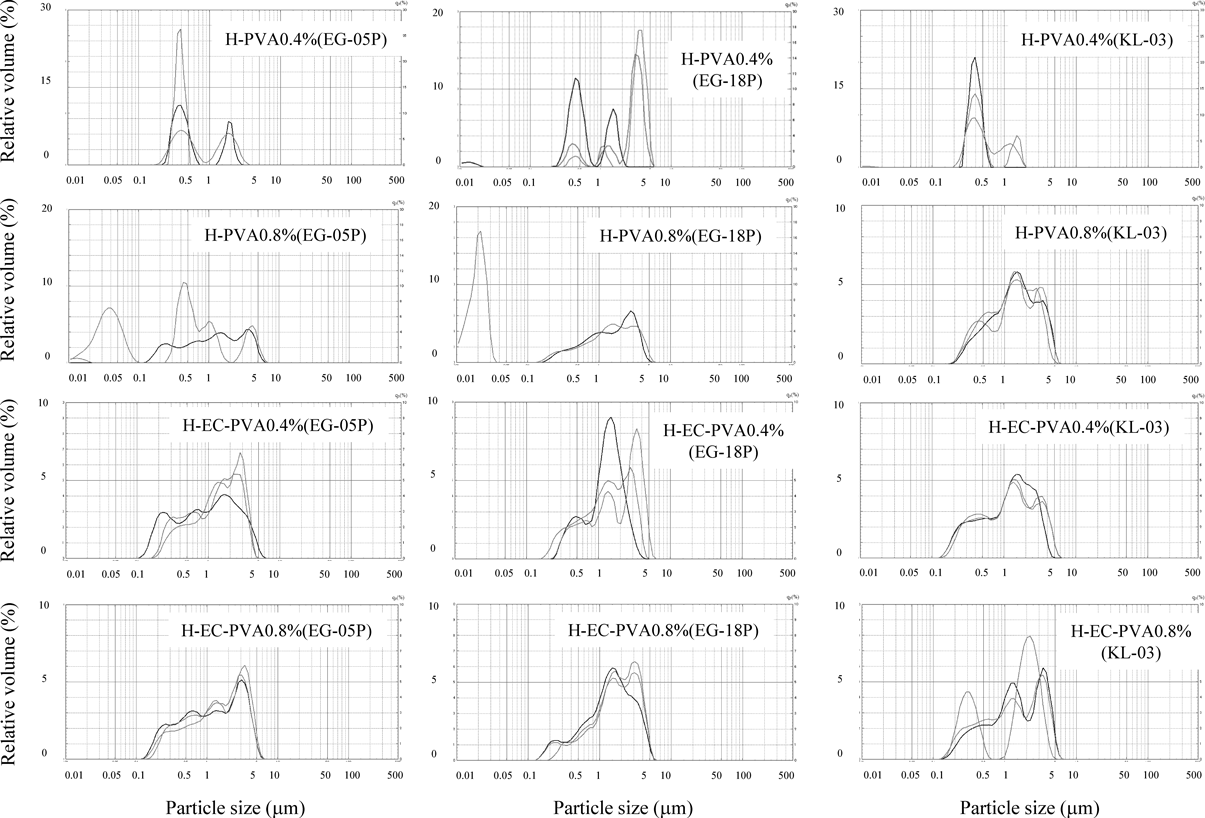
The particle size distributions of the SLNs are shown in Fig. 2. A reproducible particle size distribution was not obtained for H-PVA0.4% using any type of PVA. The use of KL-03 resulted in reproducible particle size distribution for the H-PVA0.8% series of SLNs. The median diameters of particles in the SLN suspensions are shown in Table 4. In SLNs containing EC, reproducible particle size distributions were found for H-EC-PVA0.4%(EG-05P), H-EC-PVA0.4%(KL-03), H-EC-PVA0.8%(EG-05P) and H-EC-PVA0.8%(EG-18P). The most attractive candidate for further development of SLN suspensions prepared including EC was the H-EC-PVA0.8%(EG-05P) formulation because of the reproducible particle size distributions, high EE% and relatively small median diameter. The only significant difference in zeta potentials was between H-PVA0.4%(EG-18P) and H-PVA0.8%(KL-03) (Table 4).
| Formulation | R (%) | EEa) (%) | Median diameter (µm) | ζ Potentialb) (mV) |
|---|---|---|---|---|
| H-PVA0.4%(EG-05P) | 80.5±1.7 | 78.3±2.0 | — | −23.3±2.6 |
| H-PVA0.4%(EG-18P) | 73.1±5.6 | 71.5±5.8 | — | −19.7±1.4 |
| H-PVA0.4%(KL-03) | 77.9±3.2 | 76.1±3.6 | — | −22.9±1.6 |
| H-PVA0.8%(EG-05P) | 77.9±3.5 | 76.9±3.7 | — | −21.6±2.1 |
| H-PVA0.8%(EG-18P) | 75.7±5.9 | 74.3±6.8 | — | −24.2±1.4 |
| H-PVA0.8%(KL-03) | 79.8±7.0 | 79.3±7.4 | 1.377±0.054 | −25.7±1.8 |
| H-EC-PVA0.4%(EG-05P) | 57.2±2.9 | 56.4±3.0 | 1.262±0.208 | −22.4±1.1 |
| H-EC-PVA0.4%(EG-18P) | 56.1±0.4 | 55.5±0.3 | 1.547±0.306 | −23.5±0.5 |
| H-EC-PVA0.4%(KL-03) | 57.1±4.5 | 56.3±4.6 | 1.221±0.004 | −25.2±0.8 |
| H-EC-PVA0.8%(EG-05P) | 66.5±8.4 | 65.8±8.4 | 1.376±0.127 | −22.4±2.5 |
| H-EC-PVA0.8%(EG-18P) | 60.0±4.0 | 59.8±3.7 | 1.626±0.160 | −21.9±0.8 |
| H-EC-PVA0.8%(KL-03) | 69.9±4.0 | 69.3±3.7* | 1.478±0.169 | −25.5±0.9 |
Each value represents the mean±S.D. (n=3). * Significantly different compared with H-EC-PVA0.4%(KL-03) (p<0.05). a) Student’s t-test was performed between H-PVA0.4% and H-PVA0.8% or H-EC-PVA0.4% and H-EC-PVA0.8%. b) Tukey test was performed for differences in zeta potentials. The only significant difference in zeta potentials was between H-PVA0.4%(EG-18P) and H-PVA0.8%(KL-03).
The in vitro release profiles of morin from the SLN suspensions are shown in Fig. 3. The morin ethanol solution represents the permeate profile of morin through the dialysis membrane. The release of morin from the SLN suspensions was sustained compared with the morin ethanol solution. The release of morin from the H-EC-PVA0.8%(EG-05P) suspension was similar to the morin suspension and more sustained than release from the H-PVA0.8%(KL-03) SLNs. Drug release from the H-EC-PVA0.8%(EG-05P) and morin suspensions were significantly lower that the morin ethanol solution for all the measurement points. Morin release from the morin suspension was sustained by rate-limiting dissolution because morin has low solubility.

Each point represents the mean±S.D. (n=3). Dunnett’s test was performed to compare the formulations with release profiles<morin ethanol solution. 0.5 h: H-EC-PVA0.8%(EG-05P) and morin suspension<morin ethanol solution (p<0.05). 1h: H-EC-PVA0.8%(EG-05P) and morin suspension<morin ethanol solution (p<0.01). 2h: H-PVA0.8%(KL-03), H-EC-PVA0.8%(EG-05P) and morin suspension<morin ethanol solution (p<0.05). 4h: H-PVA0.8%(KL-03) and morin suspension<morin ethanol solution (p<0.05). H-EC-PVA0.8%(EG-05P)<morin ethanol solution (p<0.01). 6h: H-EC-PVA0.8%(EG-05P) and morin suspension<morin ethanol solution (p<0.05).
Plasma morin concentration–time curves after oral administration of SLN suspensions are shown in Fig. 4. The pharmacokinetic parameters measured for morin release after oral administration of SLN suspensions are shown in Table 5. The Cmax of H-PVA0.8%(KL-03) and H-EC-PVA0.8%(EG-05P) were almost the same, and these formulations were 2.2 times higher than the morin suspension. However, the AUC0–360 of H-PVA0.8%(KL-03) was less that of the morin suspension, and the AUC0–360 of H-EC-PVA0.8%(EG-05P) was 2.3 times greater than that of the morin suspension. The relative bioavailability of the formulations (Fr) was calculated from the ratio of the AUC0–360 of the formulations to the AUC0–360 of the morin suspension (Table 4). The time that Cmax was observed (Tmax) for the H-EC-PVA0.8%(EG-05P) formulation was longer than the other formulations, and the mean residence time (MRT0–360) of the morin suspension and H-EC-PVA0.8%(EG-05P) formulation were greater than the other formulations.

Each point represents the mean±S.E. (n=3–4).
| Parameters | Administration of morin (10 mg/kg) | |||
|---|---|---|---|---|
| Morin suspension | H-PVA0.8%(KL-03) | H-EC-PVA0.8%(EG-05P) | H-EC-PVA(LL-810, 60°C, 90 s, ice) | |
| Cmax (µg/mL)* | 0.062±0.013 | 0.138±0.025 | 0.134±0.028 | 0.281±0.084 |
| Tmax (min) | 15 | 15 | 30 | 15 |
| AUC0–360 (µg/mL⋅min) | 6.173 | 5.385 | 14.445 | 18.825 |
| MRT0–360 (min) | 144.863 | 59.755 | 134.223 | 96.659 |
| Fr (%) | 100.000 | 87.243 | 234.010 | 304.967 |
* Each value represents the mean±S.E. (n=3–4). MRT: mean residence time, Fr: relative bioavailability, the ratio of the AUC0–360 of formulations to the AUC0–360 of the morin suspension.
The particle size distributions of SLNs containing low-saponification-degree PVA LL-810 are shown in Fig. 5. The H-EC-PVA(LL-810, 50°C, 20 s) SLN formulation was prepared at 50°C and sonicated for 20 s. The median SLN diameters of the low-saponification-degree formulations are shown in Table 6. The median diameter of H-EC-PVA(LL-810, 50°C, 20 s) was almost the same as H-EC-PVA0.8%(EG-05P) (Table 4). In an attempt to decrease the particle size, H-EC-PVA(LL-810, 60°C, 90 s) was prepared at 60°C and sonicated for 90 s. Although the median diameter of this formulation was smaller than that of H-EC-PVA(LL-810, 50°C, 20 s), this treatment resulted in a polydisperse particle size distribution (Fig. 5). This polydispersity was prevented by rapidly cooling the SLN suspension after sonication in ice and centrifugation for 30 min, resulting in further decrease of the median particle size (H-EC-PVA(LL-810, 60°C, 90 s, ice)). Morin was not detected in the supernatant of this formulation. The EE% of the H-EC-PVA(LL-810, 60°C, 90 s, ice) formulation was similar to that of H-EC-PVA0.8%(EG-05P) and the zeta potential of the H-EC-PVA(LL-810, 60°C, 90 s, ice) formulation was slightly more negative.
XRD patterns of freeze-dried SLNs are shown in Fig. 6. Characteristic peaks corresponding to crystalline morin were not observed in the XRD spectra of morin-loaded SLNs with the peaks at 7° and 24° attributed to hard fat. The XRD patterns of SLNs from H-PVA0.8%(KL-03), H-EC-PVA0.8%(EG-05P) and H-EC-PVA(LL-810, 60°C, 90 s, ice) formulations were very similar.
The release profile of morin from the H-EC-PVA(LL-810, 60°C, 90 s, ice) formulation was compared with that of the H-EC-PVA0.8%(EG-05P) formulation (Fig. 7). Morin release from the H-EC-PVA(LL-810, 60°C, 90 s, ice) formulation was more sustained than from the H-EC-PVA0.8%(EG-05P) formulation.



Each point represents the mean±S.D. (n=3).
| Formulation | R (%) | EE (%) | Median diameter (µm) | ζ Potential (mV) |
|---|---|---|---|---|
| H-EC-PVA(LL-810, 50°C, 20 s) | — | — | 1.466±0.078 | — |
| H-EC-PVA(LL-810, 60°C, 90 s) | — | — | 1.118±0.037 | — |
| H-EC-PVA(LL-810, 60°C, 90 s, ice) | 65.3±2.0 | 65.3±2.0 | 0.518±0.014 | −26.2±1.0 |
Each value represents the mean±S.D. (n=3).
The plasma morin concentration–time curve after oral administration of the H-EC-PVA(LL-810, 60°C, 90 s, ice) suspension is shown in Fig. 8. The pharmacokinetic parameters of morin after oral administration of the H-EC-PVA(LL-810, 60°C, 90 s, ice) suspension are shown in Table 5. The Cmax of H-EC-PVA(LL-810, 60°C, 90 s, ice) was 4.5 times greater than the morin suspension. The AUC0–360 from the H-EC-PVA(LL-810, 60°C, 90 s, ice) formulation was three times that of the morin suspension. The MRT0–360 of H-EC-PVA(LL-810, 60°C, 90 s, ice) was shorter than that of the morin suspension.

Each point represents the mean±S.E. (n=3–4).
In this study, SLN suspensions were prepared using hard fat as a base, with or without EC to controlled release. PVAs and Tween 60 were used as surfactants, and the effect of the amount of PVA used on the SLN properties was evaluated. The difference between R% and EE% decreased in SLN formulations containing more PVA; i.e., the morin concentration in the supernatant was lowered by increasing the amount of PVA added. We consider that leakage of morin from SLNs is prevented by increasing the amount of PVA used. For the H-PVA0.4% and H-PVA0.8% formulations, only H-PVA0.8%(KL-03) showed a reproducible particle size distribution (Fig. 2). The H-PVA0.8%(KL-03) formulation was used in subsequent experiments as the optimized high-saponification-degree PVA formulation because of its reproducible particle size distribution and high the EE% value. The EE% was decreased in SLN formulations containing EC. We believe that this is caused by increased precipitation in the preparation of the SLNs. The EE% of EC-SLNs was higher when more PVA was used (H-EC-PVA0.8% vs. H-EC-PVA0.4% series). The EC-SLN formulations with reproducible particle size distributions were H-EC-PVA0.4%(EG-05P), H-EC-PVA0.4%(KL-03), H-EC-PVA0.8%(EG-05P) and H-EC-PVA0.8%(EG-18P). The EE% of the H-EC-PVA0.8%(EG-05P) and H-EC-PVA0.8%(EG-18P) formulations were greater that the corresponding low-PVA formulations (H-EC-PVA0.4%(EG-05P) and H-EC-PVA0.4%(EG-18P)). The median diameter of the H-EC-PVA0.8%(EG-05P) SLNs was smaller than that of the H-EC-PVA0.8%(EG-18P) SLNs. These attributes were deemed favorable and H-EC-PVA0.8%(EG-05P) was used in subsequent optimization experiments. A relatively low-saponification-degree PVA is preferable for SLNs without EC, whereas a relatively high-saponification-degree is more appropriate for SLNs containing EC. Considering the affinity between the PVA and lipid base is necessary to prepare optimal SLNs. Solid lipid has low fluidity compared with a liquid lipid, and is likely to have greater affinity to relatively low-polymerization-degree PVAs. Hard fat contains few hydroxyl groups with most of the hydroxyl groups forming ester bonds with saturated fatty acids. KL-03 (79.7 mol% saponification) has more hydrophobic acetate groups than EG-05P (88.0 mol% saponification). SLNs without EC showed preferential affinity to KL-03, which is likely because of increased affinity between hydrophobic hard fat and the acetate groups of KL-03. The EC used in this study had about 49% (w/w) ethoxyl groups and about 51% of the hydroxyl groups unfunctionalized. The SLNs with EC showed preferential affinity with the relatively high-saponification-degree EG-05P, which is likely caused by affinity between the hydroxyl groups of EG-05P and EC.
Measurement of zeta potential provides a prediction of the storage stability of a colloidal dispersion. In general, a high absolute value of zeta potential (±30 mV or more) is preferably to prevent the coalescence of the dispersed droplets.29,30) However, Abdulkarim et al.31) reported that the highest diffusion in mucus was observed for nanoparticles with a neutral surface charge. The zeta-potential values of the optimized formulations, H-PVA0.8%(KL-03) (−25.7 mV) and H-EC-PVA0.8%(EG-05P) (−22.4 mV), are a compromise to give storage stability and acceptable mucosal permeability. The in vitro release of morin was sustained for longer from the optimized EC containing SLN formulation (H-EC-PVA0.8%(EG-05P)) than from the optimized SLN formulation without EC (H-PVA0.8%(KL-03)). We have previously reported that EC dispersed in hard fat constructs a matrix that sustains drug release.20,21) The formation of a matrix explains the sustained morin release from H-EC-PVA0.8%(EG-05P).
In the in vivo oral administration study, the Cmax of H-PVA0.8%(KL-03) and H-EC-PVA0.8%(EG-05P) were 2.2 times greater than that of the morin suspension. It has been suggested that the affinity and bioadhesion of SLN to the intestinal membrane can improve drug bioavailability.32,33) The increased Cmax of both H-PVA0.8%(KL-03) and H-EC-PVA0.8%(EG-05P) are likely because of improved affinity and bioadhesion to the intestinal membrane. The Tmax after administration of the H-EC-PVA0.8%(EG-05P) formulation was substantially longer than the H-PVA0.8%(KL-03) formulation. After an initial peak, plasma concentration of morin rapidly reduced after dosage with the H-PVA0.8%(KL-03) formulation. The release of morin from the H-PVA0.8%(KL-03) SLNs was likely rapid in the gastrointestinal tract as was the diffusion of the released morin. The plasma concentration of morin remained higher from 45 to 360 min after administration of the H-EC-PVA0.8%(EG-05P) formulation compared with that after administration of the morin suspension. We considered it probable that morin encapsulated in H-EC-PVA0.8%(EG-05P) was retained on the mucosa, and morin was released slowly from the SLN matrix base after 45 min. It is also possible that SLNs encapsulated morin permeated through the intestinal membrane. We considered that the increased bioavailability of morin in H-EC-PVA0.8%(EG-05P) was caused by affinity and bioadhesion of the SLNs to the intestinal membrane and the residence property of SLNs on the mucosa surface.
Smaller-sized SLNs can increase accessibility to the enterocyte surface and improve permeability across the intestinal membrane.32,33) SLNs using only high-saponification-degree PVAs were preparable at a temperature higher than 60°C with longer ultrasonic exposure time. However, particle size distribution was not reproducible and SLNs failed to form in some cases. We consider it unlikely that integrated SLNs were not generated during the longer ultrasonic exposure at 60°C and altering the cooling step would not contribute to generating integrated SLNs. Formulation of SLNs containing EC with a low-saponification-degree PVA could be prepared reproducibly at 60°C with 90 s of ultrasonic exposure. The use of high temperature and long sonication resulted in a polydisperse particle size distribution for the H-EC-PVA(LL-810, 60°C, 90 s) formulation. This was possibly because of aggregation of particles during the cooling process, and H-EC-PVA(LL-810, 60°C, 90 s, ice) was immediately cooled in ice to prevent aggregation. The particle size distribution of the H-EC-PVA(LL-810, 60°C, 90 s, ice) SLNs was monodisperse, and the median diameter was the smallest obtained for the SLNs studied (518 nm). The cooling method used may affect the crystalline form of the formulation. XRD patterns of freeze-dried SLNs were obtained and because no difference was observed between H-EC-PVA(LL-810, 60°C, 90 s, ice), H-PVA0.8%(KL-03) and H-EC-PVA0.8%(EG-05P), we inferred that the cooling method did not affect the crystal form of the SLNs.
The smaller particle size of the H-EC-PVA(LL-810, 60°C, 90 s, ice) formulation resulted in more sustained in vitro morin release compared with that of H-EC-PVA0.8%(EG-05P). PVA has hydrophilic hydroxyl groups and hydrophobic acetate groups, and is commonly used to stabilize emulsions because it allows the formation of relatively small-sized particles with uniform size distribution.34,35) Given that low-saponification-degree PVA has large amounts of hydrophobic acetate groups, it should have a stronger affinity with hard fat. The sustained release from H-EC-PVA(LL-810, 60°C, 90 s, ice) SLNs is caused by the low-saponification-degree PVA stabilizing the surface of the SLNs.
The Cmax of morin after a dose of the H-EC-PVA(LL-810, 60°C, 90 s, ice) formulation was increased to 2.1 times that of a H-EC-PVA0.8%(EG-05P) dose. The Tmax after a dose of H-EC-PVA(LL-810, 60°C, 90 s, ice) was less than that from a dose of H-EC-PVA0.8%(EG-05P). The small particle size of the H-EC-PVA(LL-810, 60°C, 90 s, ice) formulation improves accessibility to the enterocyte surface. Furthermore, the sustained morin release from H-EC-PVA(LL-810, 60°C, 90 s, ice) SLNs is likely caused by permeation of SLN encapsulated morin through the intestinal membrane. The plasma concentration of morin at 45 and 60 min after administration of H-EC-PVA(LL-810, 60°C, 90 s, ice) was higher than that after a dose of H-EC-PVA0.8%(EG-05P). This increase is attributed to retention of the H-EC-PVA(LL-810, 60°C, 90 s, ice) SLNs on the mucosa. The plasma drug concentrations at 120 and 360 min were almost the same for H-EC-PVA(LL-810, 60°C, 90 s, ice) and H-EC-PVA0.8%(EG-05P) and we consider the residence properties on the mucosa surface of these two formulation were the same after 120 min. Hence, H-EC-PVA(LL-810, 60°C, 90 s, ice) has only a moderately increased AUC0–360 compared with H-EC-PVA0.8%(EG-05P).
In this study, all doses of morin were relatively low (10 mg/kg) to minimize saturation of P-gp-mediated efflux. Morin should be discharged by P-gp-mediated efflux after permeating across the intestinal membrane. However, the AUC0–360 of the H-EC-PVA(LL-810, 60°C, 90 s, ice) formulation was three times of that of the morin suspension. This increase of bioavailability is caused by increased affinity and bioadhesion of the SLNs to the intestinal membrane and permeation of morin encapsulated in SLNs through the intestinal membrane.
The optimized H-EC-PVA(LL-810, 60°C, 90 s, ice) formulation improved the bioavailability of morin, which has low aqueous solubility, exhibits P-gp-mediated efflux, and low intestinal permeability. Formulation of SLNs containing EC and a low-saponification-degree PVA could improve the bioavailability of other drugs with low aqueous solubility, P-gp-mediated efflux, and low intestinal permeability.
This work was funded by a Japan Society for the Promotion of Science Grant-in-Aid for Scientific Research (JSPS KAKENHI), Grant number 26460235.
This work was financially supported by the Nippon Synthetic Chemical Industry Co., Ltd., Osaka, Japan.