2016 年 39 巻 9 号 p. 1564-1567
2016 年 39 巻 9 号 p. 1564-1567
The aim of the present study was to investigate whether pretreatment with the Japanese herbal medicine, “Juzen-taiho-to” (JTX), had an ameliorative effect on carbon tetrachloride (CCl4)-induced hepatotoxicity through anorexia prevention. Mice injected with CCl4 exhibited severe anorexia. Moreover, CCl4 increased the plasma levels of hepatic injury markers (i.e., alanine aminotransferase and aspartate aminotransferase), lipid peroxidation, and hepatic Ca2+ levels. Pretreatment with JTX recovered the CCl4-induced anorexia. In addition, JTX pretreatment decreased CCl4-induced plasma levels of hepatic injury markers. Increased Ca2+ is a known indicator of the final progression to hepatocyte death, and CCl4-induced hepatotoxicity is mainly caused by oxidative stress. The present study indicated CCl4-induced lipid peroxidation and hepatic Ca2+ content decreased with JTX pretreatment. Our results suggest that JTX has potential to protect of CCl4-induced anorexia, and the modulation of oxidative stress.
The liver is one of the most important organs in the human body and has multiple functions such as detoxification, protein synthesis, and the production of chemicals necessary for digestion. In addition, the liver is the most vulnerable organ, sensitive to toxic chemical agents.1) Acute liver injury is the rapid development of hepatocellular dysfunction with poor prognosis. It frequently results from drug administration, viral infection, and toxic or hepatic ischemic-reperfusion injuries.2)
Carbon tetrachloride (CCl4) belongs to a class of chemicals widely used as grain fumigants, anthelmintics, and intermediates of the synthesis of haloalkane chlorofluorocarbons.3) CCl4 is one of the most extensively studied hepatotoxicants, since its exposure mimics human hepatotoxicity. CCl4-induced hepatotoxicity is a multifactorial process.4–6) The first step is the metabolic activation of CCl4 by cytochrome P450 2E1 (CYP2E1), resulting in trichloromethyl and trichloromethyl peroxy radicals. The second step is radical binding: the free radicals react with the sulfhydryl groups of antioxidant enzymes (e.g., glutathione and protein thiols). The third step involves the overexpression of these free radicals leading to several deleterious effects such as enhanced membrane lipid peroxidation and covalent macromolecule binding. The fourth step is ATP depletion and intracellular Ca2+ elevation. Through these steps, necrosis is induced, which is classified as the fifth step. Multiple compounds such as esculentoside A, a saponin isolated from the root of Phytolacca esculenta, have shown protective effects against CCl4-induced hepatotoxicity. In addition, associated protective mechanisms possibly act on each of the five steps.7–10)
The Japanese herbal medicine “Juzen-taiho-to” (JTX) is a Kampo medicinal prescription, and has been used historically in Japan for the treatment of cancers, rheumatoid arthritis, and atopic dermatitis.11) JTX is consisting of ten medicinal herbs in the indicated amounts (g per day in human intake); Astragali Radix (3.0), Cinnamomi Cortex (3.0), Rehmanniae Radix (3.0), Paeoniae Radix (3.0), Cnidii Rhizoma (3.0), Atractylodis Lanceae Rhizoma (3.0), Angelicae Radix (3.0), Ginseng Radix (3.0), Poria Cocos (3.0) and Glycyrrhizae Radix (1.5). In addition to these therapies, JTX has traditionally been used to treat anorexia.12) Since CCl4 is an anorexic agent, we hypothesized that JTX suppressed CCl4-induced toxicity by preventing anorexia.
Therefore, in the present study, we investigated whether pretreatment with JTX was sufficient to attenuate CCl4-induced anorexia. Moreover, we further studied whether pretreatment with JTX decreased CCl4-induced hepatic injury.
Male ddY mice were purchased from Japan SLC (Shizuoka, Japan). Following arrival at our facility, mice were maintained under standard conditions of controlled temperature (24±1°C), humidity (55±5%), and light (12 : 12 h light/dark cycles) with free access to water and food. Experimental treatments were performed using seven-week-old animals. Following the experiment, any surviving mice were sacrificed using pentobarbital. All experiments were approved by the Institutional Animal Care and Experiment Committee of Kinjo Gakuin University (No. 129).
Experimental ProtocolMice were divided into four groups. Twenty-four hours before the CCl4 injection, animals of Groups-2 (JTX group) and -4 (JTX+CCl4 group) were intraperitoneally (i.p.) administered a JTX extract powder (Tsumura, Tokyo, Japan) dissolved to saline (10% JTX solution) at 1 g/kg (10 mL/kg). This dose was according to Anjiki et al.13) Animals in Groups-1 (control group) and -3 (CCl4 group) were injected i.p. with equivalent volumes of the saline vehicle. Twenty-four-hours after the JTX or saline injection, both Groups-3 and -4 were administered 1.6 g/kg (at 10 mL/kg) CCl4 i.p. Groups-1 and -2 were injected i.p. with equivalent volumes of olive oil. Twenty-four hours after the CCl4 or olive oil injection, mice from each group were euthanized and bled for plasma analysis. The food intake after CCl4 or olive oil administration was calculated per day. The resulting plasma samples were stored at −80°C before conducting alanine aminotransferase (ALT) and aspartate aminotransferase (AST) assays. The livers were harvested from each animal, and separate samples from each liver were stored at −80°C.
Measurement of ALT and ASTPlasma ALT and AST activities were measured using the Wako Transaminase CII Test (Wako Pure Chemical Industries, Ltd., Osaka, Japan), according to the manufacturer’s instructions and as previously described.14,15)
Measurement of Hepatic Ca (Ca2+) ConcentrationLiver specimens (0.2–0.3 g each) were digested in 0.5 mL of concentrated nitric acid (60%: Kanto Chemical, Tokyo, Japan) in glass test tubes. The digested samples were held at 80°C for 1 h, with a subsequent gradual increase in temperature (10°C/h) until the sample temperature reached 130°C. The samples were held at 130°C until the specimens became transparent and were diluted to a 5 mL final volume with distilled water and used to determine the Ca2+ concentration by atomic absorbance using a Z-2300 spectrophotometer (Hitachi, Tokyo, Japan).
Measurement of Malondialdehyde (MDA) Levels in LiverTotal MDA levels in the liver were examined with a colorimetric thiobarbituric acid reactive substances (TBARS) microplate assay kit (FR40, Oxford Biochemical Research, Oxford, MI, U.S.A.), according to the manufacturer’s protocol and as previously described.16)
Determination of Metallothionein (MT) LevelsMT protein levels were determined by the Cd saturation–hemolysate method (Cd–hem method), as previously described.15)
Statistical AnalysisAll data from the control and treatment groups were obtained from the same numbers of replicated experiments. All experiments were performed independently at least twice. Statistical significance between two groups was estimated using two tailed Student’s t-test. Multiple comparisions were analyzed using one-way ANOVA with post-hoc Tukey–Kramer’s test. All statistical analyses were performed using SPSS 19.0 software (Chicago, IL, U.S.A.). Values of p<0.05 were considered statistically significant.
First, we measured food intake and found that CCl4 decreased food intake by >75% than that of the control (from 5.32 to 1.18 g, Fig. 1), which was consistent with previously reported data.7) In contrast to the CCl4-injected group, pretreatment with JTX partially recovered food intake (from 1.18 to 2.96 g). In addition, saline- or JTX-injected groups showed the same level (5.32, 5.00 g, respectively) although dosage was about 8 times higher compared to the adult dose in human. Several reports indicated relation anorexia and hepatic injury.17,18) These mentioned anorexia is sign of hepatic injury. These data suggested that JTX improved CCl4-induced anorexia presumably by protecting liver injury.

Mice were injected i.p. with 10% JTX solution (1 g/kg). Twenty-four hours after pretreatment, mice were injected i.p. with 1.6 g/kg CCl4. Amount of food intake during 24 h was determined after i.p. injection with CCl4.
Together with the food intake measurement, we analyzed plasma ALT and AST activities (Fig. 2), which are markers of liver injury and dysfunction. The control and JTX groups showed a normal level of ALT (Fig. 2A) and AST (Fig. 2B) activities. Administration of CCl4 led to an increase in ALT and AST plasma concentrations, whereas pretreatment with JTX suppressed ALT and AST activities by 60 and 44%, respectively.
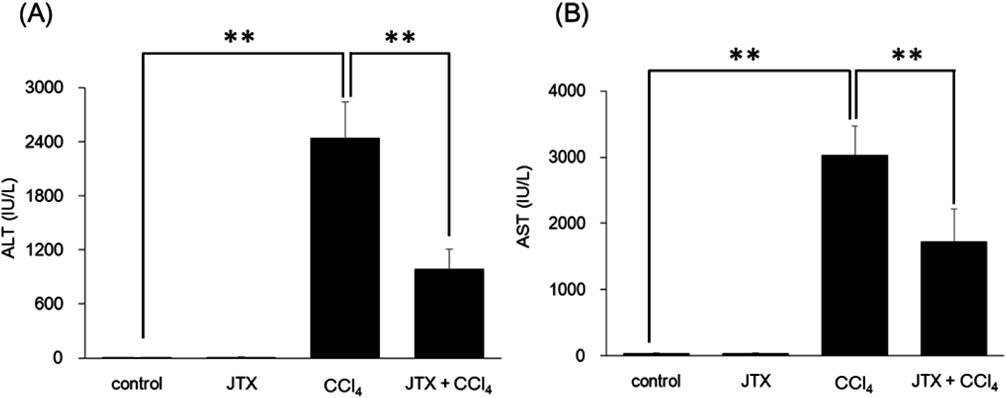
Mice were injected i.p. with 10% JTX solution (1 g/kg). Twenty-four hours after pretreatment, mice were injected i.p. with 1.6 g/kg CCl4. ALT (A) and AST (B) levels in liver was determined 24 h after i.p. injection with CCl4. Data are presented as the mean±S.D. of five or six mice. ** Significant difference between compared values (p<0.01).
To further investigate the protective effect of JTX against CCl4, we calculated hepatic Ca2+ content, since exposure to CCl4 elevates hepatocellular Ca2+ levels.19) In the present study, CCl4 injection upregulated hepatic Ca2+, whereas pretreatment with JTX decreased it (Fig. 3). These data suggest that JTX inhibits CCl4-induced hepatotoxicity.

Mice were injected i.p. with 10% JTX solution (1 g/kg). Twenty-four hours after pretreatment, mice were injected i.p. with 1.6 g/kg CCl4. Ca concentration in liver was determined 24 h after i.p. injection with CCl4. Data are presented as the mean±S.D. of five or six mice. ** Significant difference between compared values (p<0.01).
Several studies suggest that a possible molecular mechanism involved in CCl4 hepatotoxicity is the disruption of the delicate oxidant/antioxidant balance, which can lead to liver injury via oxidative damage.4,20) Moreover, CCl4 is a prototypical lipid peroxidant that induces early lipid peroxidation in the liver.8) As a marker of lipid peroxidation, MDA concentration was measured in all animal groups. CCl4 treatment significantly increased MDA levels, whereas pretreatment with JTX eliminated CCl4-induced MDA upregulation (Fig. 4), suggesting that JTX itself and/or JTX-induced gene products may have an antioxidant effect. One potential mechanism is the induction of MT. MT has antioxidant properties against reactive oxygen species (ROS) and free radical scavenging abilities.21) MT expression is upregulated in many organs by various metals such as Zn2+ and Cd2+,22) and by non-metallic compounds.23) We previously reported that Zn2+-induced MT expression has a protective effect on CCl4-induced hepato- and nephrotoxicity.15,16) In addition, Anjiki et al. reported JTX induced MT in the liver and small intestines.13) Our current study demonstrate i.p. injection of JTX increased hepatic MT levels more than 200 µg/g liver (Fig. 5). Itoh et al. reported Echnoside A and Sakuraso-saponin induced MT (200 µg/g liver) and protection is observed against CCl4-induced hepatotoxicity.24) Since MT induction level is same by Itoh et al., these data support our hypothesis.

Mice were injected i.p. with 10% JTX solution (1 g/kg). Twenty-four hours after pretreatment, mice were injected i.p. with 1.6 g/kg CCl4. MDA levels in liver was determined 24 h after i.p. injection with CCl4. Data are presented as the mean±S.D. of five or six mice. ** Significant difference between compared values (p<0.01).

Mice were injected i.p. with 10% JTX solution (1 g/kg). Twenty-four hours after pretreatment, the liver samples were collected followed by determining the MT protein levels. Data are presented as the mean±S.D. of six mice. ** Significant difference between compared values (p<0.01).
CCl4 is widely used to investigate hepatic injury associated with oxidative stress and free radicals. The ROS induced by CCl4 causes direct tissue damage, and initiates inflammation through the activation of various cytokines.9) Several studies have shown how some compounds show protective effects against CCl4-induced hepatotoxicity.7–10) Some of these compounds attenuated oxidative stress and the inflammatory response.10) As JTX modulates the immune system, such as Toll-like receptor 4 signaling pathways,25,26) it might also suppress the inflammatory response activated by CCl4.
Since JTX is composed of ten different medical herbs, it is unknown whether our present results are caused by one or more compounds. Further investigation is necessary to elucidate the active component of JTX. In addition, a detailed protective mechanism of JTX against CCl4-induced hepatotoxicity is still needed, as our current study is purely descriptive. Moreover, further study is needed to indicate hepatoprotective action of JTX in lesser concentration than current concentration. These issues are currently being investigated and the manuscripts are in preparation.
In conclusion, we have demonstrated that pretreatment with JTX suppresses CCl4-induced anorexia and hepatic injuries. We hypothesize that the hepatoprotective effect of JTX is attributed to its antioxidant role. To our knowledge, this is the first evidence suggesting that JTX protects against CCl4-induced acute hepatotoxicity. Although further investigation is needed to clarify the active component within JTX, these findings are expected to improve our understanding of the protective effect of this Japanese herbal medicine against radical-induced organ injury and disease.
The authors thank Dr. Kenichi Saeki and Dr. Nobuyuki Fukuishi (Kinjo Gakuin University, Japan) for his kind suggestions.
The authors declare no conflict of interest.
The online version of this article contains supplementary materials.