2017 Volume 40 Issue 6 Pages 922-925
2017 Volume 40 Issue 6 Pages 922-925
Ritodrine, a drug for the treatment of threatened premature labor, is a highly selective beta-2 agonist with the major metabolites of sulfate and glucuronide conjugates. This study investigated the continuous evaluation of the concentration of ritodrine conjugates in relation to the clinical course in twin pregnancy. The subjects were 9 twin-pregnancy mothers who delivered after receiving ritodrine treatment between April 2012 and December 2013. Serum ritodrine sulfate and glucuronide conjugates were deconjugated using their specific enzymes. Ritodrine concentration was measured by liquid chromatography-tandem mass spectrometry. The continuous infusion rate of ritodrine was 2.66±0.67 (0.8–3.54) µg/min/kg, and the average concentration of unchanged ritodrine was 118.8±33.2 (63.8–194.0) ng/mL. During the study period between week 32 and week 36 of gestation, the average ratio of unchanged ritodrine concentration and sulfate ritodrine conjugate concentration for weeks 32, 33, 34, 35, and 36 were 1.7, 1.9, 1.5, 1.7, and 1.7 not significant (N.S.), respectively. The average ratio of unchanged ritodrine concentration and glucuronide ritodrine conjugate concentration were 1.8, 2.2, 1.9, 1.8, and 2.1 (N.S.), respectively. No statistical difference was identified in the ratios of unchanged ritodrine concentration and sulfate or glucuronide ritodrine conjugate concentrations. Large individual differences were shown in the concentration of sulfate and glucuronide during the gestational period. No change in the ratio of the formation of ritodrine metabolites was identified as the gestational age progressed.
In Japan, treatment to prevent premature labor with ritodrine, a selective β2 agonist, is allowed as normal medical care until full term birth at the 37th week of gestation.1) Pulmonary edema has been reported as one of the serious side effects of overdoses of ritodrine when used in combination with adrenocorticosteroids in multiple pregnancies.1) Although ritodrine has been shown to provide significant control for births within 48 h, there are reports that it provides no improvement in the perinatal prognosis or newborn mortality.2,3) The use of ritodrine has recently been restricted in Europe because of its maternal side effects.4) However, because fetal development is considered important, ritodrine continues to be used in Japan following a policy of long-term tocolysis for the purpose of extending the gestational age.1)
The most important perinatal problem in multiple pregnancies is premature labor. The risk of premature labor increases with the number of fetuses; the risk is reported to be 5.4 times greater in twin pregnancies compared to singletons.5) Furthermore, there is an effect on intrauterine growth restriction as the number of fetuses increases, with many low birthweight infants among twins.6) The systemic clearance of unchanged ritodrine in twin pregnancy mothers was found to be 0.83–2.34 L/h/kg but large individual differences of up to three-fold were shown in twin pregnancies.7) In addition, 3 of the 14 mothers enrolled in that study showed a negative correlation with regard to gestational age and the clearance of unchanged ritodrine.8) Changes in maternal physiology caused by the continuation of twin pregnancy are considered to be one of the reasons.8) And it was shown that twin pregnancy mothers had a significantly lower systemic clearance of ritodrine than singleton pregnancy.7)
Although there are very few reports regarding the metabolism of ritodrine, including that in single pregnancy, the metabolism process consists mainly of conjugation to sulfate and glucuronide.9) It has been reported that ritodrine conjugates have no pharmacological activity.10) According to a study by Brashear et al. the proportions of ritodrine conjugates in maternal urine collected after giving birth were 45% sulfate and 38% glucuronide.11) Hayashi et al. have also reported that the total of sulfate and glucuronide conjugates was approximately 85% in maternal plasma immediately following birth.12) However, there is little information regarding the presence of ritodrine conjugates continuously during the gestational period. The present study investigated the metabolism of ritodrine throughout the gestational period by enzymatic processing of plasma obtained from twin pregnancy mothers undergoing treatment with ritodrine, and determined the concentrations of unchanged ritodrine and both conjugates separately.
The subjects of this study were 9 mothers pregnant with twins who were diagnosed with threatened premature labor and underwent continuous intravenous infusion of ritodrine hydrochloride from the time of hospitalization until delivery. The patient characteristics are shown in Table 1.
| Category | Mean±S.D. (range) |
|---|---|
| Study period | April 2012–December 2013 |
| Subjects | 9 |
| Dichorionic diamniotic (DD twin) | 6 |
| Monochorionic diamniotic (MD twin) | 3 |
| Serum samples | 39 |
| Age (years) | 35.1±4.5 (29–41) |
| Weight (kg) | 60.8±6.8 (49.1–83.3) |
| Gestational age at birth (weaks) | 34.9±0.9 (33.4–36.0) |
| Ritodrine administration period (d) | 62.4±37.1 (28–145) |
| Rate of ritodrine administration (mg/kg/h) | 0.16±0.04 (0.05–0.21) |
| Number of newborns | 18 |
| Weight of first born (g) | 2268±274 (1938–2834) |
| Weight of second born (g) | 1986±252 (1536–2362) |
Thirty-nine serum samples used were obtained from the remnants of blood samples taken for clinical examination during hospitalization. The blood samples were taken 72 h or later after the continuous infusion of ritodrine hydrochloride, a time at which it was considered to reach steady state, and the samples were stored at −20°C until measurement. Preprocessing of the samples was performed as described by Brashear et al.11) In order to perform enzymatic hydrolysis of the sulfate and glucuronide conjugates, two 50-µL test samples were prepared from the same specimen. The enzymes used were β-glucuronidase from bovine liver (Sigma G-0251, Sigma-Aldrich, St. Louis, MO, U.S.A.) and arylsulfatase from Aerobacter aerogenes (Sigma S-1629, Sigma-Aldrich). The concentration of each of the conjugates was calculated by subtracting the concentration of unchanged ritodrine from the measured overall ritodrine concentration (the sum of the concentration of unchanged ritodrine and the deconjugated ritodrine conjugates) after hydrolysis.
Ritodrine Concentration MeasurementsRitodrine concentration was measured by liquid chromatography-tandem mass spectrometry (LC-MS/MS).13) The internal standard, ritodrine-d4 (10 ng), and 100 µL sodium carbonate buffer 0.1 M (pH 9.75) were added to the 50-µL test sample, to which 1 mL of ethyl acetate was added and mixed. After centrifugation at 2000×g for 10 min, 800 µL of the supernatant was evaporated to dryness at 40°C for 1 h. The residue was dissolved in 100 µL of water, and 5 µL aliquots of resultant solution were injected into the LC-MS/MS system (API 3200 QTRAP system, AB Sciex, Framingham, MA, U.S.A.). The mobile phase used consisted of acetonitrile and water (1 : 1) and (0.1% formic acid), and the flow rate was 0.1 mL/min.
The calibration curve for concentrations of ritodrine from 10–1000 ng/mL was y=10.4x (r=0.999, n=5). The intra-day and inter-day variations were 5.2% (100 ng/mL, n=5) and 7.5% (1000 ng/mL, n=5), respectively.
Statistical AnalysisThe relationships between the concentrations of unchanged, sulfate and glucuronide conjugates of ritodrine in relation to dosage of ritodrine and gestation age were statistically evaluated.
The ratio of the concentrations of ritodrine metabolites and the concentration of unchanged ritodrine were calculated to analyze the relationship to gestational age. Analysis was performed using the Kruskal–Wallis test, and multiple comparisons were conducted with Dunn’s test. A significance level of less than 5% indicated a significant difference.
Ethical ConsiderationsThis study approved by the Ethics Committee of Tenshi Hospital (No. 053). Written consent was obtained from the mothers.
Treatment by continuous intravenous infusion of ritodrine was begun in the 9 study subjects starting on week 26.0±4.8 (14.6–30.6) of the pregnancy [mean±standard deviation (S.D.) (range)], and was continued for 62±35 (28–145) d. As a result, the twin pregnancy mothers all gave birth with no remarkable side effects. The dosage of ritodrine was 0.16±0.04 (0.05–0.21) mg/kg/h, and the average concentration of unchanged ritodrine was 118.8±33.2 (63.8–236.6) ng/mL.
Comparisons of the concentrations of unchanged, sulfate and glucuronide conjugates of ritodrine in relation to dosage of ritodrine from week 32 to week 36 of the pregnancies were evaluated. The average ratios of unchanged ritodrine concentration to ritodrine dosage for weeks 32, 33, 34, 35, and 36 were 14.3, 13.1, 12.6, 13.4, and 23.6 (h·kg·L−1), respectively, with a significant difference between week 33 and week 36 (p<0.05). The average ratios of ritodrine sulfate conjugate concentration to ritodrine dosage were 23.4, 24.7, 19.1, 21.2, and 44.6 (h·kg·L−1, not significant (N.S.)), respectively. However, the average ratios of ritodrine glucuronide conjugate concentration to ritodrine dosage were 25.2, 27.4, 24.1, 24.0, and 50.2 (h·kg·L−1), respectively, with a significant difference between week 33 and week 36 (p<0.05).
The ratio of sulfate ritodrine conjugate concentration and glucuronide ritodrine conjugate concentration to unchanged ritodrine concentration from week 32 to week 36 of the pregnancy is shown in Fig. 1. The average ratio of sulfate ritodrine conjugate concentration and unchanged ritodrine concentration for weeks 32, 33, 34, 35, and 36 were 1.7, 1.9, 1.5, 1.7, and 1.7 (N.S.), respectively. The average ratio of glucuronide ritodrine conjugate concentration and unchanged ritodrine concentration were 1.8, 2.2, 1.9, 1.8, and 2.1 (N.S.), respectively. There was no change shown in the concentration ratio as gestational age progressed.
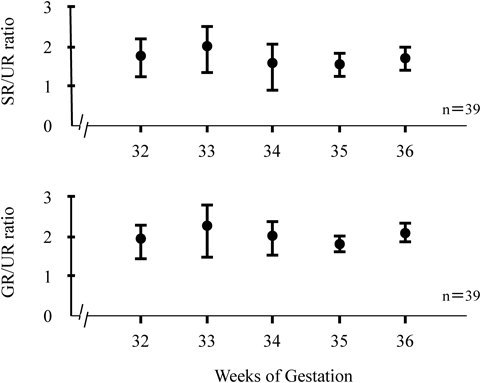
Closed circle represents the mean value, error bars denote standard deviation. UR: Unchanged serum ritodrine concentration (ng/mL), SR: sulfate conjugate ritodrine concentration (ng/mL), GR: glucuronide ritodrine concentration (ng/mL).
The gestational age for premature birth and the survival threshold for neonatal care have been defined as 26 weeks. Most premature infants born around week 33 of pregnancy have been reported to show a favorable prognosis.14,15) Ritodrine, a highly selective β2 agonist, used in Japan for the treatment of threatened premature labor to prolong pregnancy until full term birth at week 37 with long-term continual infusion.1) But, in Europe and the United States, the use of ritodrine to prevent labor is limited to 48 h to avoid the side effects caused by the β2 agonistic effects of ritodrine outside the uterus.15,16) However, because substantial brain development of the fetus occurs during the 2nd and 3rd trimester, neonates born between the gestational ages of 22–25 weeks are highly susceptible to brain damage.14,15) Therefore, ritodrine is used in Japan for long-term tocolysis for the purpose of extending pregnancy.1) Of the 9 twin pregnancy mothers in the present study, ritodrine treatment was started in one mother on the 14th week of pregnancy and on the 25th week in 3 mothers. As a result, as shown in Table 1, the births of the mothers treated with ritodrine were extended to an average of ≥33.4 weeks with no births occurring before the 26th week. There was one low birth weight infant weighing 1536 g, without any complications evident following delivery (Table 1). Concomitant medication in the 9 subjects was as follows: 6 patients underwent continuous administration of sulfate magnesium and 6 patients underwent intramuscular dexamethasone therapy for fetal lung maturity.
Although a number of circulatory system-related side effects associated with ritodrine have been reported,16) there are no reports focusing on ritodrine concentration. While working on a method for the effective use of ritodrine in long-term tocolysis, we developed a procedure to measure the concentration of unchanged ritodrine, and reported a difference of approximately three-fold in the value of the clearance of unchanged ritodrine in some cases.8)
Ritodrine is metabolized mainly to sulfate and glucuronide conjugates, with the ritodrine conjugates having no pharmacological activity.9–12) It was considered that the concentration of unchanged ritodrine varied with the decline or increase in the conjugation reaction, thus influencing the effectiveness on uterine muscle. Therefore, we studied the influence of conjugation concentrations, leading to the large individual differences in the level of systemic clearance of unchanged ritodrine.
Because maternal physiology is greatly altered during pregnancy, both pharmacokinetics and pharmacodynamics of ritodrine are considered to be influenced.17) Especially affected is circulatory blood flow, which is increased by up to 40% in some mothers.18) Thus, Phase 1 of drug metabolism considered to be altered.19,20) In addition, it has been reported that the renal glomerular filtration rate generally increases during pregnancy and that renal clearance of some drugs is increased.19)
The sulfotransferases (SULTs) that mainly play a role in the metabolism of ritodrine are SULT1A1 in the liver and SULT1A3 in the intestine.21) The potency ratio of the conjugation of ritodrine for SULT1A1 compared to SULT1A3 is approximately 1/5 to 1/4, with large individual differences reported for the activity of SULT1A1 in the liver.21,22) It has been reported that because endogenous steroidal hormones are metabolized along the same metabolic pathway, enzyme induction due to increased progesterone during pregnancy and moderation of enzyme activity due to estrogen contribute to increased drug metabolism during pregnancy.23) Furthermore, it has been confirmed that immediately following delivery, nearly the same amount of ritodrine is transferred to the maternal and neonatal blood.10) It has also been reported that sulfate conjugates are excreted into the amniotic fluid during the fetal stage.10) In this study, the ratio of sulfate ritodrine conjugate concentration and unchanged ritodrine concentration was calculated to investigate the relationship to gestational age, but even as full term delivery neared, no change was recognized sulfate ritodrine conjugation ability (Fig. 1).
Uridine 5′-diphosphate (UDP)-glucuronosyltransferase (UGT) activity has been reported to increase during pregnancy and that the systemic clearance of lamotrigine, which undergoes glucuronide conjugation, is also increased during pregnancy.24) In addition, it has been reported that drug clearance is reduced due to a variation of the multidrug resistance-associated protein (MRP) family, belonging to the ATP-binding cassette (ABC) transporters, for which glucuronide conjugates are substrates.25) In the present study, the ratio of the dosage of ritodrine to the concentration of ritodrine glucuronide conjugates at week 36 showed a significant increase in serum concentration over week 33 (27.4 vs. 50.2 (h·kg·L−1, p<0.05). However, the ratio of glucuronide ritodrine conjugate concentration and unchanged ritodrine concentration was calculated to investigate the relationship to gestational age, but even as full term delivery neared, no change was recognized glucuronide ritodrine conjugation ability (Fig. 1).
These results indicate that the changes in maternal physiology affecting the sulfate and glucuronide ritodrine conjugates were not the reason for the variance in the systemic clearance of unchanged ritodrine in this study. Further study is needed to determine the reason for the individual differences in the systemic clearance of unchanged ritodrine.
The present study revealed large individual differences in the formation of sulfate and glucuronic acid conjugates during the gestational period. No change in the ratio of the formation of ritodrine metabolites was identified as gestational age progressed.
The authors declare no conflict of interest.