2022 Volume 45 Issue 9 Pages 1373-1377
2022 Volume 45 Issue 9 Pages 1373-1377
This study aimed to identify the components of proton pump inhibitors (PPIs) or potassium-competitive acid blocker (PCAB) that lead to cardiovascular events in individuals of working age. We analyzed large claims data of individuals who were administered PPIs or PCAB. We enrolled working-age individuals administered PPI or PCAB without cardiovascular history with a 12-month screening and 12-month observation period and determined the proportion of cardiovascular events and the predictive factors of cardiovascular events in this population. Among the eligible individuals, 0.5% (456/91098) had cardiovascular events during the 12-month observation period. Predictive factors for cardiovascular events were age for +1 year (p < 0.0001), male sex (p < 0.0001), hypertension (p = 0.0056), and diabetes mellitus (p < 0.0001). The cardiovascular disease risk was higher in working-age individuals administered lansoprazole than in those administered other drugs (vs. rabeprazole; p = 0.0002, vs. omeprazole; p = 0.0046, vs. vonoprazan; p < 0.0001, and vs. esomeprazole; p < 0.0001). We identified the risk for cardiovascular events in individuals being treated with lansoprazole. Lansoprazole is known for its higher CYP2C19 inhibition activity compared with other PPIs or PCAB. A possible mechanism by which lansoprazole may lead to cardiovascular events is inhibiting the generation of epoxyeicosatrienoic acids from arachidonic acids, an intrinsic cardioprotective activator via CYP2C19 inhibition. Thus, we recommend avoiding administering lansoprazole to working-age individuals require PPIs or PCAB.
Globally, cardiovascular-related deaths account for one-third of the total deaths. The number of cases of cardiovascular disease (CVD) have increased from 271 million in 1990 to 523 million in 2019. The number of deaths had also increased from 12.1 to 18.6 million during this period, despite of advancement for health care.1) The onset of CVD is generally common among older individuals; however, the number of CVD cases in the younger population have been increasing recently. Approximately 39% of the CVD related deaths were of those aged below 70 years.2) The productivity-loss due to the death of individual aged 45–64 years (working age) caused by CVD in Australia was 755 million dollars in 2015; this increased to 1.082 billion dollars in 2030 in terms of loss in gross domestic product.3) This is an important issue globally, especially in individuals of working age.
Proton pump inhibitors (PPIs) and potassium-competitive acid blocker (PCAB) are frequently prescribed for reflux esophagitis and gastric ulcers. Adverse events associated with PPIs and PCAB are reported to be approximately 10%. In general, the adverse events caused by PPIs and PCAB are known to be reversible, such as headache, diarrhea, nausea, elevation of liver enzymes, pancytopenia, and allergic reactions.4–12) However, irreversible adverse events, such as cardiovascular events, pneumonia, chronic kidney disease, dementia, osteoporotic fractures, and upper gastrointestinal cancer, have also been reported recently.13–24) To prevent PPIs and PCAB toxicity, to detect the risk factor is important issue.
Recently, PPIs have been associated with all causes of death including CVD.24) However, CVD toxicity due to the exposure to PPIs, including PCAB, which is a similar drug category, has not been well investigated. In addition, the relationship between the components of PPI and PCAB and cardiovascular event risk has not been assessed. Here, we aimed to identify the components of PPI or PCAB that lead to cardiovascular events in individuals of working age in a 1-year retrospective assessment.
The JMDC (Japan Medical Data Center, Tokyo, Japan) claim database consists of anonymized data of over 10 million people (inpatient, outpatient, and pharmacy claims) aged ≤74 years from approximately over 90% of the medical facilities in Japan. All individuals in the JMDC database have “social insurance,” which includes working individuals and their families. As of April 2021, the database represented 10% of the Japanese population.25)
Case Identification and DefinitionThis retrospective study included approximately 370000 individuals who were prescribed PPIs and PCAB (Anatomical Therapeutic Chemical (ATC) code for A02BC01, omeprazole; A02BC03, lansoprazole; A02BC04, rabeprazole; A02BC05, esomeprazole; A02BC08, vonoprazan), excluding a combination drug for PPI and aspirin. Among these patients, a retrospective cohort was set for 12 months of screening and observation period, and 142116 individuals remained. Of these individuals, those with missing data regarding the prescription date of PPIs or PCAB (n = 2061) and family members (n = 40127) and those with a history of cardiovascular-related diagnosis (International Classification of Diseases-10 codes I20-25 and I60-69) during the 12-month screening period (n = 8830) were excluded from this study. Finally, 91098 individuals were included in the study (Fig. 1).
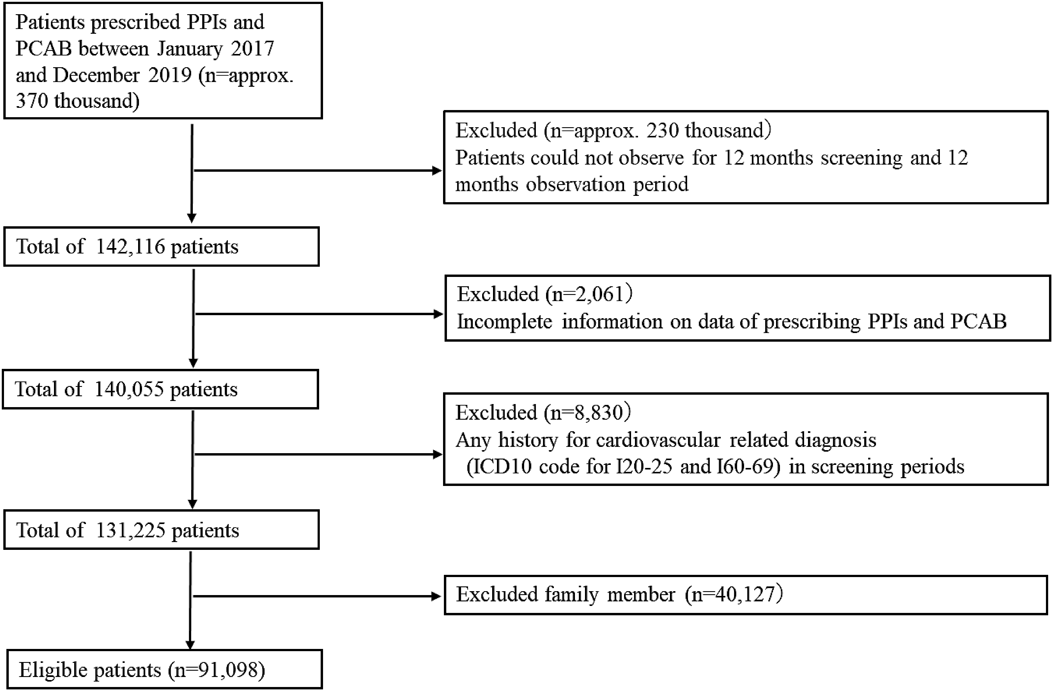
PCAB, potassium-competitive acid blockers; PPI, proton pump inhibitors.
Cardiovascular events were defined as surgery and endovascular treatment using billing codes for surgical procedures assigned by the Ministry of Health, Labour and Welfare. We identified percutaneous coronary intervention (PCI) using the following treatment codes: “K546 (percutaneous coronary angioplasty),” “K547 (percutaneous coronary atherectomy),” “K548 (percutaneous coronary angioplasty [with special catheter]),” “K549 (percutaneous coronary stenting),” “K550 (intracoronary thrombolysis),” “K550-2 (percutaneous coronary artery thrombectomy),” or “K551 (coronary angioplasty [thromboendarterectomy]).” Coronary artery bypass grafting (CABG) was identified using the codes “K552 (coronary artery and aortic bypass grafting)” or “K552-2 (coronary artery and aortic bypass grafting [without using artificial heart–lung]). Brain surgery and endovascular treatments were identified using the codes “K175 (cerebral aneurysm encapsulation),” “K176 (cerebral aneurysm inflow blood vessel clipping [using craniotomy]),” “K177 (cerebral aneurysm cervical clipping),” “K178 (cerebrovascular surgery),” “K178-2 (percutaneous cerebral angioplasty),” “K178-3 (percutaneous selective cerebral thrombosis/embolization lysis),” “K178-4 (percutaneous cerebral thrombosis recovery),” “K178-5 (percutaneous cerebrovascular stent placement),” “K164 (intracranial hematoma removal [using craniotomy]),” “K164-3 (cerebrovascular embolism [thrombus] removal),” “K164-4 (stereotactic intracerebral hematoma removal),” and “K164-5 (endoscopic intracerebral hematoma removal)” and the use of tissue plasminogen activators (ATC code for B01AD04, urokinase; B01AD02, alteplase; D05412, monteplase).
Ethics DeclarationCommercially available JMDC database used in this study is anonymized processed information based on Japan’s Personal Information Protection Law, and individual informed consent is not required for provision and use. In addition, according to the ethical guidelines for clinical research in Japan, research using anonymized processed information is not required to be reviewed by an ethical review committee.
Study EndpointsThe data of eligible individuals were used to assess the primary and secondary endpoints. The primary endpoint was the proportion of cardiovascular events in individuals of working age who started PPIs or PCAB during the 12-month observation period. The secondary endpoint was a predictive factor for cardiovascular events in individuals of working age who started PPIs or PCAB.
Statistical AnalysisIn this study of 91098 individuals, the predictive factors for cardiovascular events included baseline parameters, such as age (elderly are at higher risk for CVD), sex (males are at higher risk for CVD), comorbidity for hypertension, diabetes mellitus, dyslipidemia, and chronic kidney disease; PPIs; and PCAB. We followed standard methods to estimate the sample sizes for multiple logistic regression. At least 10 outcomes were required for each independent variable to avoid overfitting.
Data are expressed as medians with ranges or mean ± standard deviations. Data analysis was performed using JMP 15® (SAS Institute Inc., Cary, NC, U.S.A.).
In our study, the most frequently prescribed PPI or PCAB was vonoprazan (30.6%) for eligible individuals (male/female: 67262/23836, 45.5 ± 11.6 years). Consequently, the prescription rates of esomeprazole, lansoprazole, rabeprazole, and omeprazole were 29.9, 20.9, 13.1, and 5.5%, respectively (Table 1). The following comorbidities were present: hypertension in 13301 (14.6%) individuals; dyslipidemia, 12142 (13.3%); diabetes mellitus, 7026 (7.7%); and chronic kidney disease, 554 (0.6%). Among the eligible individuals, 0.5% (456/91098) for whom PPI or PCAB was administered experienced cardiovascular events within the 12-months observation period (Table 1).
| Total | |
|---|---|
| Age, years [S.D.] | 45.5 ± 11.6 |
| Sex, n (%) | |
| Female | 23836 (26.2) |
| Male | 67262 (73.8) |
| Comorbidity, n (%) | |
| Hypertension | 13301 (14.6) |
| Dyslipidemia | 12142 (13.3) |
| Diabetes mellitus | 7026 (7.7) |
| Chronic kidney disease | 554 (0.6) |
| Administration period, n (%) | |
| Within 8 weeks | 69276 (76.0) |
| Over 6 months | 8953 (9.8) |
| Cardiovascular events, n (%) | |
| Medical practice for the event | 456 (0.5) |
| Administration of t-PA | 14 (0.02) |
| Coronary intervention, surgery | 397 (0.44) |
| Brain endovascular treatment, surgery | 48 (0.05) |
| PPI, n (%) | |
| Rabeprazole | 11927 (13.1) |
| Lansoprazole | 19075 (20.9) |
| Vonoprazan | 27869 (30.6) |
| Esomeprazole | 27253 (29.9) |
| Omeprazole | 4974 (5.5) |
PPI, proton pump inhibitors S.D., standard deviation.
Exposure to PPI or PCAB components within 8 weeks after their administration for managing reflux esophagitis was noted in 76.0% (n = 69276) of individuals. PPI or PCAB use over 6 months was observed in 9.8% (n = 8953) of individuals.
Secondary Endpoint: Predictive Factors of Cardiovascular Events in Individuals Administered PPIs or PCABLogistic regression analysis revealed the following odds ratios for various factors: age for +1 year (1.07 [1.06–1.08], p < 0.0001), male sex (3.41 [2.35–4.95], p < 0.0001), hypertension (1.37 [1.01–1.71], p = 0.0056), and diabetes mellitus (2.06 [1.62–2.62], p < 0.0001). The Cardiovascular events risk was higher in working-age individuals administered lansoprazole than administered other drugs (vs. rabeprazole: 0.53 [0.38–0.74], p = 0.0002; vs. omeprazole: 0.54 [0.35–0.83], p = 0.0046; vs. vonoprazan: 0.59 [0.46–0.75], p < 0.0001; and vs. esomeprazole: 0.58 [0.46–0.75], p < 0.0001) (Table 2).
| With cardiovascular events (n = 456) | Without cardiovascular events (n = 90642) | Adjusted odds ratio (95% CI) | p-Value | |
|---|---|---|---|---|
| Age, year [S.D.], [odds for +1year] | 55.8 ± 7.8 | 45.5 ± 11.6 | 1.07 (1.06–1.08) | <0.0001 |
| Sex | ||||
| Female | 30 | 23806 | Reference | |
| Male | 426 | 66836 | 3.41 (2.35–4.95) | <0.0001 |
| Comorbidity | ||||
| Hypertension | 176 | 13125 | 1.37(1.01–1.71) | 0.0056 |
| Diabetes mellitus | 127 | 6899 | 2.06 (1.62–2.62) | <0.0001 |
| Dyslipidemia | 140 | 12002 | 1.06 (0.84–1.35) | 0.6065 |
| Chronic kidney disease | 12 | 542 | 1.52 (0.84–2.76) | 0.1674 |
| Components for PPI or PCAB | ||||
| Lansoprazole | 147 | 18928 | Reference | |
| Rabeprazole | 46 | 11881 | 0.53 (0.38–0.74) | 0.0002 |
| Omeprazole | 25 | 4949 | 0.54 (0.35–0.83) | 0.0046 |
| Vonoprazan | 123 | 27746 | 0.59 (0.46–0.75) | <0.0001 |
| Esomeprazole | 115 | 27138 | 0.58 (0.46–0.75) | <0.0001 |
CI, confidence interval; PPI, proton pump inhibitors; PCAB, potassium-competitive acid blocker; S.D., standard deviation.
In this study, we found a risk of cardiovascular events in 0.5% of individuals administered PPI or PCAB. The predictive factors for cardiovascular events in working-age individuals administered PPI or PCAB during the 12-month observation period were exposure to lansoprazole, aging, male sex, hypertension, and diabetes mellitus (Table 2).
Medical practice bills estimated from by national medical care expenditure in Japan reached approximately 31.3 trillion Japanese yen (about 276 billion dollars) in 2018, and 20% of these expenses were related to CVDs.26) Cardiovascular diseases must be prevented especially in working-age individuals in Japan because medical costs are covered by the universal public health insurance system that is compensated by workers tax.
PPIs have been reported to impair endothelial function and accelerate endothelial senescence—one of the mechanisms of PPI use-induced cardiovascular events as observed in this study on working-age individuals as well.27–31) This suggests that one of the reasons for cardiovascular events observed in our study working-age individuals within 12 months. In addition, lower levels of epoxyeicosatrienoic acid (EET), a cardioprotective activator, could also potentially confer cardiovascular risks. EET, produced from arachidonic acid, was produced via CYP2C including CYP2C19 metabolism in the human body. However, the presence of CYP2C19 inhibitors results in lower levels of EET, which in turn increases the potential risk of cardiovascular events.32–35) Despite our study participants not having any history of cardiovascular events at least 12 months prior to study inclusion, we observed cardiovascular risks on use of lansoprazole in multiple logistic regression analysis. Lansoprazole is known to have higher CYP2C19 inhibition activity than other PPIs.36–39) In our study, the risk of cardiovascular events observed owing to lansoprazole use may reasonably be explained by this inhibition of CYP2C19 activity, thus influencing cardiovascular risks.
Our study found the predictive factors for cardiovascular events in working-age individuals administered PPI or PCAB. In particular, lansoprazole posed a higher risk of cardiovascular events than other drugs. We recommend avoiding lansoprazole in cases requiring PPI or PCAB administration.
All authors satisfy the ICMJE recommended four criteria. In particular, KM and AW built the study. KN and NK contributed to extracting raw data from the JMDC Inc. database. KM and AW drafted the manuscript. KM, AW, and YK collected the raw data. KM performed statistical analyses. TU managed ethical handling. KT and TS completed the study. All authors contributed to the discussion of the manuscript. All authors have agreed to publish this manuscript.
KM, TS (Showa University), and JMDC Inc. collaborate on this project according to the collaborative research agreement. JMDC Inc. provided large claims data. KN and NK are employees of JMDC Inc. All the authors, including KN and NK, discussed and planned the present research; JMDC Inc. did not intervene in data implementation according to the analysed results. KM received honorarium fees for presentations from JMDC.Inc. TS received honorarium for presentation at Daiichi Sankyo Company, Ltd., NIPRO CO., LTD., Nichi-Iko Pharmaceutical Co., Ltd., Pfizer Co., Ltd., Sandoz Co., Ltd., Mylan Co., Ltd., and Meiji Seika Pharma Co., Ltd. As a potential conflict of interest, the Department of Hospital Pharmaceutics, School of Pharmacy, Showa University received a research grant from Daiichi Sankyo Company, Ltd. The other authors declare no conflict of interest associated with this manuscript.