2014 年 79 巻 1 号 p. 77-84
2014 年 79 巻 1 号 p. 77-84
Background: Defibrillation testing (DT) is considered a standard procedure during implantable cardioverter-defibrillator (ICD) implantation. However, little is known about the factors that are significantly related to patients with high defibrillation threshold (DFT) using the present triad system.
Methods and Results: We examined 286 consecutive patients who underwent ICD implantation with a transvenous dual-coil lead and DT from December 2000 to December 2011. We defined patients who required 25 J or more by the implanted device as the high DFT group, and those who required less than 25 J as the normal DFT group. For each patient, assessment parameters included underlying disease, comorbidities, NYHA functional class, drugs, and echocardiographic measures. The high DFT group consisted of 12 patients (4.2%). Multivariate analysis identified 3 independent predictors for high DFT: atrial fibrillation (odds ratio (OR) 4.85, 95% confidence interval (CI) 1.24–22.33, P=0.023), hypertension (OR 4.01, 95% CI 1.08–15.96, P=0.039), thickness of interventricular septum (IVS) >12 mm (OR 4.82, 95% CI 1.17–20.31, P=0.030).
Conclusions: Atrial fibrillation, hypertension and IVS hypertrophy were significantly associated with high DFT. Identification of such patients could help to lower the risk of complications with DT. (Circ J 2015; 79: 77–84)
The implantable cardioverter-defibrillator (ICD) has become standard treatment for patients at risk of sudden cardiac death.1–4 Its use has been shown to reduce the mortality in primary and secondary prevention recipients.5,6 Therefore, it is expected that the number of implantations will increase.
Historically, effective defibrillation testing (DT) has been considered part of the standard procedure at ICD implantation to ensure adequate sensing of ventricular fibrillation (VF) and ability of the device to terminate VF. However, there is ongoing debate about the risk and benefit of DT, because of the risk of complications related to DT,7,8 the development of higher energy devices that can deliver more than 30 J, and limited clinical relevance for DT.9–12 Therefore, in this modern era, it is clinically important to identify predictors of patients with high defibrillation threshold (DFT). Nevertheless, only a few studies have reported on predictors of high DFT using ICD with the latest technology.13–15 Thus we conducted a retrospective survey in order to determine the patients at risk of high DFT with the transvenous dual-coil lead system.
To assess the prevalence and predictors of high DFT, we performed a retrospective review of 316 consecutive patients who underwent initial ICD implantation at Hokkaido University Hospital from December 2000 to December 2011; 30 patients were excluded: 10 patients using a single-coil lead and 20 patients without DT, including insufficient anticoagulation for atrial fibrillation (AF; 12 patients), noninducible VF (1 patient), left ventricular (LV) thrombosis (4 patients), recent cerebral infarction (1 patient), air embolism during ICD implantation (1 patient), and prolonged operation time (1 patient).
A dual-coil transvenous defibrillator lead was usually implanted at the right ventricular (RV) apex. At the end of the surgical procedure, DT was performed twice. VF was induced with low energy T-wave shock. The energy of the first DT was 20 J. If not successful, the energy level was raised to 25 J and if necessary to the maximal energy of the device (≥30 J). We defined patients who required ≥25 J at least once during the DT as high DFT, and those requiring <25 J twice as normal DFT (Figure 1).

Results of defibrillation testing among recipients of an implantable cardioverter-defibrillator. The delivered energy of the first and second shock was 20 J and 25 J, respectively. When induced ventricular fibrillation (VF) was terminated by the first 20-J shock, the patient was classified as having a normal defibrillation threshold (DFT). Patients who required shock energy ≥25 J for successful defibrillation were classified as high DFT. RVA, right ventricular apex; RVOT, RV outflow tract; SVC, superior vena cava.
The study was approved by the Ethics Committee of Hokkaido University Hospital.
Statistical AnalysisAll data are expressed as mean±standard error (SE). Simple between-group analyses were conducted using Student’s t-test. Categorical variables were compared using Fisher’s exact test. Logistic regression analyses were performed to estimate the predictors of high DFT. Differences with P<0.05 were considered significant. JMP® 10 (SAS Institute Inc, Cary, NC, USA) was used for all statistical analysis.
The present study included 286 patients and their characteristics are shown in Table 1. The number of patients with high DFT was 12 (4.2%). Except for intensive cardiopulmonary resuscitation required by 2 patients (see later), no serious complications occurred with DT during the implantation procedure. The rates of hypertension (HT) and AF were significantly higher in the high DFT group. No significant differences were observed in the pharmacological treatment of the groups (Table 2). However, there was a trend toward higher use of amiodarone and diuretics in the high DFT group.
| Normal DFT (n=274) | High DFT (n=12) | P value | |
|---|---|---|---|
| Age (years) | 58.3±0.9 | 60.5±3.5 | 0.610 |
| Male | 216 (79) | 8 (67) | 0.299 |
| LVEF (%) | 43±1 | 42±4 | 0.857 |
| NYHA ≥II | 151 (55) | 6 (50) | 0.773 |
| Primary prevention | 68 (25) | 2 (17) | 0.736 |
| CRT device | 52 (19) | 4 (33) | 0.260 |
| Single chamber | 84 (31) | 4 (33) | 1.0 |
| Type of HD | |||
| IHD | 84 (31) | 2 (17) | 0.520 |
| DCM | 46 (28) | 3 (25) | 0.409 |
| HCM | 29 (11) | 3 (25) | 0.114 |
| DHCM | 19 (7) | 0 (0) | 1.0 |
| CSA | 17 (6) | 1 (8) | 0.5186 |
| IVF | 37 (14) | 1 (8) | 1.0 |
| HHD | 8 (3) | 1 (8) | 0.302 |
| Sarcoidosis | 7 (3) | 1 (8) | 1.00 |
| Other | 27 (8.4) | 0 (0) | 0.608 |
| Atrial fibrillation | 66 (24) | 8 (67) | 0.003 |
| Paroxysmal/persistent | 47 (71)/19 (29) | 5 (62.5)/3 (37.5) | 0.6880 |
| Diabetes mellitus | 62 (23) | 2 (17) | 1.0 |
| Hypertension | 60 (22) | 7 (58) | 0.008 |
| Dyslipidemia | 108 (39) | 4 (33) | 0.770 |
| Hyperuricemia | 46 (17) | 3 (25) | 0.438 |
| Stroke | 23 (8) | 1 (8) | 1.0 |
Data are given as mean±SE or n (%).
CRT, cardiac resynchronization therapy; CSA, coronary spastic angina; DCM, dilated cardiomyopathy; DFT, defibrillation threshold; DHCM, dilated phase of hypertrophic cardiomyopathy; DT, defibrillation testing; HCM, hypertrophic cardiomyopathy; HD, heart disease; HHD, hypertensive HD; ICD, implantable cardioverter-defibrillator; IHD, ischemic HD; IVF, idiopathic ventricular fibrillation; LVEF, left ventricular ejection fraction; NYHA, New York Heart Association functional classification.
| Normal DFT (n=274) | High DFT (n=12) | P value | |
|---|---|---|---|
| Ia | 7 (3) | 1 (8) | 0.293 |
| Ib | 20 (7) | 2 (17) | 0.230 |
| Ic | 0 (0) | 0 (0) | |
| β-blocker | 195 (71) | 8 (67) | 0.750 |
| Amiodarone | 127 (46) | 9 (75) | 0.075 |
| Calcium antagonists | 42 (15) | 3 (25) | 0.411 |
| ACEI/ARB | 178 (65) | 10 (83) | 0.230 |
| α-blocker | 1 (0.4) | 0 (0) | 0.293 |
| Digitalis | 18 (7) | 2 (17) | 0.201 |
| Statin | 101 (37) | 4 (33) | 1.0 |
| Aldosterone antagonist | 78 (29) | 3 (25) | 1.0 |
| Nitrates | 29 (11) | 1 (8) | 1.0 |
| Diuretics | 131 (48) | 9 (75) | 0.08 |
Data are given as n (%).
Ia, Class Ia antiarrhythmic drugs; Ib, Class Ib antiarrhythmic drugs; Ic, Class Ic antiarrhythmic drugs; ACEI, angiotensin-converting enzyme inhibitor; ARB, angiotensin II receptor blocker. Other abbreviations as in Table 1.
Echocardiographic data are given in Table 3. Interventricular septum thickness (IVST) (P=0.062) and LV posterior wall thickness (LVPWT) (P=0.056) tended to be greater in the high DFT group, but did not reach statistical significance. The ratio of patients with IVST >12 mm (P=0.006) was significantly higher in the high DFT group.
| Normal DFT (n=274) | High DFT (n=12) | P value | |
|---|---|---|---|
| LAD (mm) | 44.4±0.6 | 47.8±2.4 | 0.230 |
| LAD ≥42 mm | 163 (60) | 10 (83) | 0.134 |
| IVST (mm) | 11.7±0.3 | 14.5±1.7 | 0.062 |
| IVST >12 mm | 56 (20) | 7 (58) | 0.006 |
| LVPWT (mm) | 9.6±0.1 | 10.9±0.6 | 0.056 |
| LVPWT >12 mm | 11 (4) | 2 (17) | 0.097 |
| LVDd (mm) | 57.6±0.7 | 56.1±3.4 | 0.682 |
| LVDd ≥55 mm | 142 (52) | 7 (58) | 0.772 |
| LVDs (mm) | 44.8±0.9 | 42.0±3.8 | 0.549 |
Data are given as mean±SE or n (%).
IVST, interventricular septum thickness; LAD, left atrial dimension; LVDd, left ventricular end-diastolic dimension; LVDs, left ventricular end-systolic dimension; LVPWT, left ventricular posterior wall thickness. Other abbreviations as in Table 1.
In order to identify the predictors of high DFT, logistic regression analysis was performed. Presence of AF, HT, use of amiodarone and diuretics, IVST >12 mm, LVPWT >12 mm, and left atrial dimension (LAD) ≥42 were related to high DFT in terms of P-value <0.2 by univariate analysis. Multivariate analysis identified 3 independent predictors for high DFT: AF (odds ratio (OR) 4.85, 95% confidence interval (CI) 1.24–22.33, P=0.023), HT (OR 4.01, 95% CI 1.08–15.96, P=0.039), and IVST >12 mm (OR 4.82, 95% CI 1.17–20.31, P=0.030) (Table 4).
| OR | 95% CI | P value | |
|---|---|---|---|
| Atrial fibrillation | 4.85 | 1.24–22.33 | 0.023 |
| Hypertension | 4.01 | 1.08–15.96 | 0.039 |
| Amiodarone | 1.81 | 0.43–9.39 | 0.428 |
| Diuretics | 3.11 | 0.65–19.24 | 0.181 |
| LAD ≥42 mm | 1.53 | 0.18–10.12 | 0.673 |
| IVST >12 mm | 4.82 | 1.17–20.31 | 0.030 |
| LVPWT >12 mm | 1.17 | 0.17–11.89 | 0.882 |
CI, confidence interval; OR, odds ratio. Other abbreviations as in Tables 1,3.
In the high DFT group, initial energy to terminate VF was 25 J in 5 patients, 30 J in 4 patients, and 35 J in 1 patient. A 35-J shock was unsuccessful in 2 patients who required repeated cardiopulmonary resuscitation because of failure of initial external defibrillation (Figure 1). The 9 patients (3.1%) with a DFT ≤30 J were managed by use of a high-output device and/or change in the shock polarity, thereby obtaining a safety margin of 10 J above the DFT. Among the 3 patients (1.0%) with a DFT ≥35 J, a change in the defibrillator lead position from the RV apex (RVA) to the RV outflow tract (RVOT) reduced the DFT to 25 J in 2 patients. The DFT in another patient with a low shock impedance (35 ohms) in the triad configuration was lowered to 25 J by electrical disconnection of the SVC coil.
The present study has demonstrated that the presence of AF, HT, and IVST >12 mm are independent predictors of high DFT patients, who require ≥25 J to terminate VF during DT with the modern ICD system of an active can and a transvenous dual-coil implantable defibrillator lead using biphasic waveform shock.
Previous studies have reported the characteristics of high DFT patients9,10,13–31 and their predictors included low ejection fraction (EF), increase in LV diameter and mass, NYHA class, and use of amiodarone (Table 5). To our knowledge, our study is the first report of AF and HT as predictors of high DFT. This is because few studies have focused on common diseases such as AF and HT as predictors of high DFT patients and operators are reluctant to perform DT in patients with AF. Even in the recent studies,13,14 these factors were not identified.
| Year | Design | n | ICD system | High DFT, n (%) & definition |
High DFT predictors | Other |
|---|---|---|---|---|---|---|
| 198821 | Single | 33 | Patch | 15 (45%), 5 (15%) ≥20 J, ≥25 J |
Proposal of 10-J safety margin |
|
| 199123 | Single | 125 | Patch | 23 (18%), ≥25 J | Low EF, previous heart surgery | |
| 199217 | Multi | 1,946 | Patch | 90 (4.6%), ≥25 J | ||
| 199526 | Single | 101 | Single-coil | 2 (2%), ≥25 J | LV mass, heart rate at rest | |
| 199719 | Single | 119 | Single-coil | 28 (24%), >20 J | Amiodarone, LV dilation, body size | |
| 200330 | Data from 2 trials |
968 | Dual-coil | 103 (11%), ≥18 J | NYHA class, low EF, Amiodarone, no history of bypass surgery, presenting with VF |
No worse prognosis in high DFT |
| 200528 | Single | 1,045 | 71 (6.2%), <10 J margin |
Younger, nonischemic, low EF, amiodarone, upgrade/replace |
||
| 200531 | Single | 18 | 4 (22%), <10 J margin |
Short VFCL and V-ERP in Brugada vs. structural HD |
Brugada syndrome |
|
| 200516 | Single | 75 | 3 (4%), not terminated by device |
LV wall thickness >45 mm and/or amiodarone |
HCM | |
| 200620 | Single | 121 | 14 (12%), <10 J margin |
QRS duration ≥200 ms | CRT-D, no worse prognosis in high DFT |
|
| 200629 | Ventak CHF/ Contak CD |
478 | 134 (7.1%), >21 J | LVDd >64 mm, procedure time >257 min |
CRT-D | |
| 20089 | SCD-HeFT | 717 | 16 (2.2%), >20 J | |||
| 200815 | Intrinsic RV Study |
1,471 | 59 (3.9%), <10 J margin |
NYHA class, nonischemic | ||
| 200922 | Single | 23 | 13 (57%), ≥15 J | QRS duration | HCM | |
| 201024 | Single | 65 | 8 (12%), <10 J margin | Creatinine >175 μmol/L | CRT-D | |
| 201027 | Single | 23 | Dual-coil | 5 (22%), ≥20 J | LV wall thickness (no correlation between QRS duration and DFT) |
HCM |
| 201018 | Canadian Registry |
229 | 17 (7.0%), 28 (12.2%), <10 J margin, >21 J |
|||
| 201125 | Single | 89 | Dual-coil | 3 (3.4%), >20 J | LVEF, IVS, LV mass and QRS duration were not predictive |
HCM |
| 201210 | SAFE-ICD | 2,120 | 47 (7%), <10 J margin | |||
| 201313 | Single | 2,138 | 48 (2.2%), <10 J margin |
Younger, LVEF <25% (amiodarone use was not predictive) |
||
| 201314 | Single | 436 | Dual-coil | 9 (2.1%), <10 J margin |
Amiodarone, DCM, low EF (by univariate analysis) |
CRT-D, CRT with a defibrillator; EF, ejection fraction; IVS, interventricular septum; LV, left ventricular or ventricle; Multi, multicenter study; Single, single-center study; VF, ventricular fibrillation; VFCL, VF cycle length; V-ERP, effective refractory period of the ventricle. Other abbreviations as in Table 1.
A previous study reported that increased LV wall thickness was a predictor of high DFT.26 It is proposed that an electric field of a certain threshold gradient (≈5V/cm2) must be applied to >90% of the myocardium to terminate VF.32 In larger hearts and those with myocardial fibrosis, the electrical field is likely to diminish, and the conduction velocity of defibrillation wave can be slow, thereby requiring higher energy delivery from the device for successful defibrillation.26,32
It has been proposed that intramural tunnel propagation following ICD shock is the major mechanism of defibrillation failure.33 The present study identified hypertrophy of the septal wall as a predictor of high DFT. Increased mid-myocardial mass could provide a large area for tunnel propagation, thereby leading to defibrillation failure.
Hypertensive heart disease is characterized by cardiac hypertrophy and interstitial fibrosis, and these structural and pathophysiological changes lead to diastolic dysfunction.34 Similarly, a previous study reported that not only persistent but also paroxysmal AF induced myocardial fibrosis and diastolic dysfunction.35 Therefore, HT and AF, common causes of diastolic dysfunction, could be associated with high DFT. Further study is required to test the hypothesis that diastolic dysfunction and ventricular fibrosis are related to high DFT.
In contrast to previous studies,13,14 low EF was not a predictor of high DFT in this study. The rate of patients with preserved EF, such as idiopathic VF, coronary spastic angina, and hypertrophic cardiomyopathy, was relatively high in this study (Table 1). In this regard, the mean LVEF was approximately 43% (Table 1), which was more than 10% higher than in past studies.13,14 The heterogeneous patient population in our study could have negated the possible involvement of LVEF as a predictor of high DFT.
Previous study demonstrated that a high incidence (26%, 4/15 patients) of defibrillation failure was observed postoperatively in patients with a safety margin <10 J at DT during ICD implantation.21 Thus, it was proposed that the DFT should be less than the maximal delivery energy of ICD by ≥10 J (safety margin of 10 J). In this respect, it has been considered necessary to perform DT at ICD implantation and to lower the DFT by modifying the ICD configuration: alteration of location, type, or number of defibrillation electrodes; changing the polarity of shock electrodes; and adjusting the biphasic waveform.36–38
The incidence and predictors of high DFT varies according to several factors, including its definition, the ICD system, and patient characteristics (Table 5). In the earlier era (eg, the 1990 s), the rate of patients with high DFT was relatively high (>10% in most studies). Since then, there have been great advances in ICD technology; for example, the devices have moved from the abdomen to the chest, lead technology has improved, the maximum delivered energy of the ICD has increased, biphasic waveforms have become standard, and there have been advancements in the programmability of waveform characteristics. These advances may have decreased the frequency and changed the clinical profile of patients with high DFT. In fact, recent studies reported a frequency of patients with high DFT among unselected ICD recipients as approximately 2–7%, which is in agreement with our study (4.2%). In contrast, it has been reported that the incidence of high DFT seems to be increased (7–12%) in patients requiring CRT.
There is no doubt that some patients will receive a more effective system after the performing of DT, such as a high-energy device, repositioning of defibrillator leads, addition of electrodes, or reverse polarity, thereby achieving the 10-J safety margin. The results of these interventions after DT in both the recent literature13,14,18 and the present study are summarized in Table 6 and Figure 2. Among 3,089 patients undergoing DT, 85 (2.8%) were identified as high DFT, and 73% (n=62) of them had acceptable DFT (ie, achieved 10-J safety margin) after some interventions (Figure 2A). Polarity reverse and use of a high-energy device were often performed to obtain the appropriate safety margin in these studies (Table 6, Figure 2B).
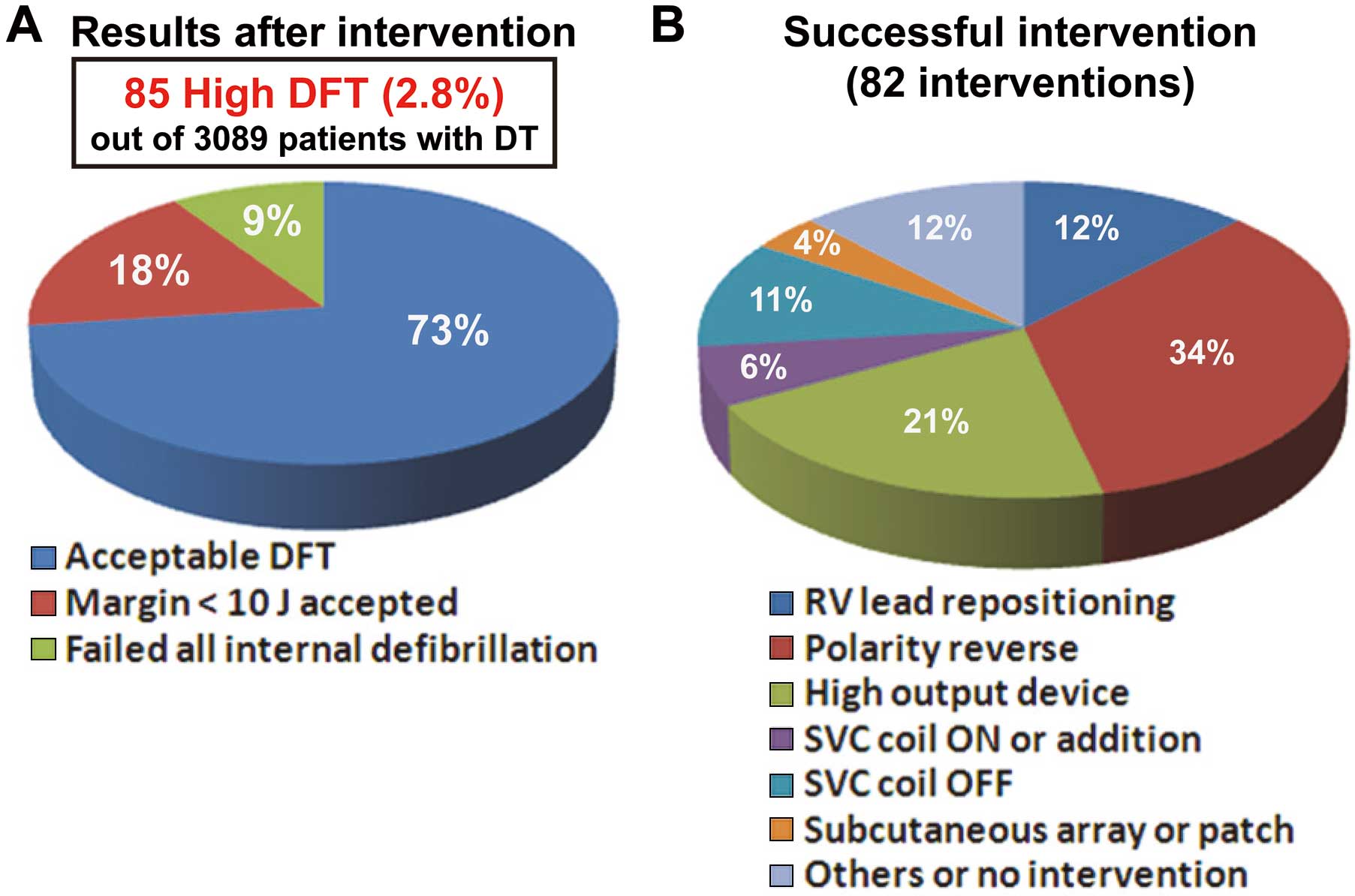
Management of patients with a high defibrillation threshold (DFT) based on combined data from previous reports13,14,18 and the present study. (A) Final results after intervention in patients with high DFT. Acceptable DFT indicates that a safety margin ≥10 J was obtained after some interventions to the ICD system. (B) Details of successful interventions. DT, defibrillation testing; ICD, implantable cardioverter-defibrillator; RV, right ventricular; SVC, superior vena cava.
| Healey et al18 | Vischer et al14 | Lin et al13 | Present study | |
|---|---|---|---|---|
| No. of patients undergoing DT | 229 | 436 | 2,138 | 286 |
| Replacement | 25 (11) | 127 (29) | 409 (19) | 0 (0) |
| High DFT | 16 (7.0) | 9 (2.1) | 48 (2.2) | 12 (4.2) |
| Results after intervention | ||||
| Acceptable DFT | 9 (56) | 9 (100) | 32 (67) | 12 (100) |
| Margin <10 J accepted | 3 (19) | 0 (0) | 12 (25) | 0 (0) |
| Failed all internal defibrillation | 4 (25) | 0 (0) | 4 (8) | 0 (0) |
| Total | 13 | 20 | 32 | 17 |
| RV lead repositioning | 4 (31) | 2 (10) | 2 (6) | 2 (12) |
| Polarity reverse | 3 (23) | 5 (25) | 17 (53) | 3 (18) |
| High-output device | 2 (15) | 1 (5) | 7 (22) | 7 (41) |
| SVC coil ON or addition | 0 (0) | 1 (5) | 4 (13) | 0 (0) |
| SVC coil OFF | 2 (15) | 6 (30) | 0 (0) | 1 (6) |
| Subcutaneous array or patch | 0 (0) | 3 (15) | 0 (0) | 0 (0) |
| Other or no intervention | 2 (15) | 2 (10) | 2 (6) | 4 (24) |
Data are given as n or n (%).
RV, right ventricular; SVC, superior vena cava. Other abbreviations as in Table 1.
Regarding RV lead positioning, it was reported that there were no differences in defibrillation efficacy when the defibrillation lead was placed at the RVOT compared with the RVA.39,40 On the other hand, one study found significantly higher DFT with leads implanted in the RVOT compared with the RVA.41 We experienced 2 cases of extremely high DFT in which VF was not defibrillated by a shock with maximal energy from the ICD and in both cases the patients required prolonged resuscitation. However, changing the location of the defibrillator lead from the RVA to the RVOT dramatically reduced the energy requirement for defibrillation, thereby obtaining the 10-J safety margin (Figure 1). Therefore, in a subgroup of patients, especially with high DFT using a dual-coil lead, repositioning of the defibrillator lead to the RVOT may help lower the DFT effectively.42
A reason why routine DT in the modern era has become controversial could be the long operation time using intravenous anesthesia and the possible complications of DT. The rates of DT-related stroke, prolonged resuscitation, and deaths have been reported to be 0.026%, 0.14% and 0.016%, respectively, in the Canadian experience.7 A subanalysis of the SCD-HeFT trial reported there were no significant differences in mortality and shock efficacy of ICD between a high DFT group and a not-high DFT group.9 Moreover, it was reported that a margin of 4–6 J was adequate to maintain high conversion success with a dual-coil ICD system, and those authors argued against a 10-J safety margin.43 Thus, recent publications question whether DT is worth the risk of the test itself.9–12
On the other hand, the purpose of DT is not only to identify a high DFT patient but also to confirm definite sensing during VF. We should also realize that, in some patients, failure of a defibrillator lead cannot be detected until delivery of a high-energy shock.44 Furthermore, most of the data from recent clinical trials are based on the ICD system with a transvenous dual-coil lead, and the data with a single-coil lead, which seems to be used increasingly, are only available from a small number of patients. Taken together, there is controversy over whether or not to perform DT at defibrillator implantation, and the Shockless IMPLant Evaluation (SIMPLE) trial is being performed to determine if ICD implantation without any DT is non-inferior to implantation with DT.45
Study LimitationsIt was a retrospective study in a single center, and may therefore incorporate important biases, especially, the small number of high DFT patients. Moreover, identification of AF by medical record alone could have underestimated its prevalence and therefore, this result may require further verification. In addition, we merely performed a defibrillation safety margin test without conducting strict DT to measure the actual DFT.
In conclusions, AF, HT, and IVS hypertrophy were significantly associated with high DFT in patients who underwent ICD implantation with a dual-coil lead in routine clinical practice. Patients with these morbidities and who undergo an ICD implantation should receive more effective systems, such as a high-output device and/or a dual-coil lead, unless DT is not performed.
We thank Dr Masayuki Sakurai, Director of Hokko Memorial Hospital, for constant encouragement of this study.