Abstract
Background:
Giant coronary aneurysm is the most severe sequela in Kawasaki disease, occurring in approximately 0.2% of patients in Japan. Regression is rare, while myocardial infarction (MI) and sudden death are relatively common. Herein, we reviewed patients with giant coronary aneurysm in a 10-year period.
Methods and Results:
A nationwide questionnaire survey was conducted based on a national epidemiological database from 1999 to 2010. We identified 355 giant coronary aneurysm patients, of whom 209 were analyzed. The 5- and 10-year total cardiac event-free rates were 0.72 and 0.68, respectively. Twelve patients died, and MI was observed in 32 patients (18.1%). Five and 6 deaths were due to coronary rupture and MI, respectively. All ruptures occurred within 1 month of onset, while most MI occurred within 18 months. There was no death beyond 2 years. Aneurysm size was significantly related to the occurrence of MI in both the right and left coronary arteries. At the time of writing, 55% of patients had no exercise limitations. And including patients who cannot perform strenuous exercises, 81% of patients were leading ordinary lives.
Conclusions:
Severe cardiac events are likely to occur within 2 years from onset of Kawasaki disease, while no deaths occurred beyond this time. Hence, careful monitoring is needed especially for the first 2 years. Most patients with giant coronary aneurysms can lead ordinary lives with appropriate management.
A nationwide epidemiologic survey on Kawasaki disease (KD), covering almost all hospitals with pediatric wards throughout Japan, has been conducted every 2 years for >30 years.1–3
While the de novo KD occurrence has been increasing from 12,9662
to 26,691,3
advances in the acute treatment of KD in these 14 years have reduced both the frequency and total number of coronary artery aneurysms from 1.97% (256 cases)2
to 0.72% (191 cases).3
The incidence of giant aneurysm, however, the most severe cardiac sequela, has changed from 0.52% (68 cases)2
to 0.18% (47 cases),3
respectively. The occurrence of giant aneurysm does not appear to be declining so much.
Giant coronary aneurysms are defined as having an internal diameter ≥8 mm, and regression is rare. They are associated with risks of coronary stenosis around the aneurysm, myocardial infarction (MI) due to thrombus, and sudden death. While the long-term prognosis of KD with giant aneurysm has been reported in single facilities or limited areas,4–6
these are insufficient to determine the real long-term outcomes of these patients. Therefore, in the present study, we evaluated the long-term prognosis of giant aneurysm patients using nationwide data. Using the detailed database of nationwide KD surveys, we were able to perform a questionnaire survey on giant aneurysms in Japan over a 10-year period.
Methods
Study Design and Patients
We performed a questionnaire survey of all KD patients with giant aneurysms throughout Japan using a database of the 16th–21st KD national surveys (1999–2010). A total of 118,946 de novo KD patients was registered during this period. The questionnaire ware sent to all 275 facilities, involving 415 cases of giant aneurysm. This survey was approved by Nippon Medical School Ethics Committee (No. 23-11-196).
The content of the questionnaire was as follows: sex; date of birth; history of KD and date of onset; diagnosis (complete KD: >5 clinical manifestations, or 4 with coronary artery lesions [CAL]; and incomplete KD, ≤4 clinical manifestations); initial acute-phase medication for KD (within 40 days of onset); and additional therapy including use of steroid, urinastatin, plasma exchange, cyclosporine, and infliximab, with the starting date of each additional treatment; laboratory data at diagnosis; presence or absence of CAL at diagnosis; illness day of CAL formation; maximum size of aneurysm; follow-up duration; latest state of coronary artery by drawing pictures; and presence or absence of coronary thrombosis, stenosis, obstruction, MI, thrombolysis therapy, catheter intervention, and coronary artery bypass graft (CABG).
Statistical Analysis
Continuous variables are described as median (range), or as mean±SD. Categorical variables are described as frequency and proportion. For comparisons between 2 groups, we used the Mann-Whitney U-test or Fisher’s exact test. We defined the composite endpoint of cardiac events as “total cardiac events”, which included cardiac death, MI, thrombolysis therapy, catheter intervention, and CABG. The total cardiac event-free rate was calculated using the Kaplan-Meier method. Factors associated with the total cardiac event-free rate and their odds ratios (OR) were calculated using univariate logistic analysis. We also classified total cardiac events based on location of giant coronary artery aneurysm, right coronary artery (RCA) or left coronary artery (LCA), defined as total RCA cardiac events and total LCA cardiac events, and evaluated them using the Kaplan-Meier method. The Cox proportional hazards model was used to calculate the hazard ratio of maximum diameter of each coronary artery aneurysm adjusted for the number of coronary artery aneurysms, age, sex, and steroid use. The maximum diameter of each coronary artery aneurysm was categorized into 2 groups as ≥8 mm–<10 mm or ≥10 mm, and evaluated in the same manner. JMP10 and SAS 9.4 (SAS Institute, Cary, NC, USA) was used for all analyses.
Results
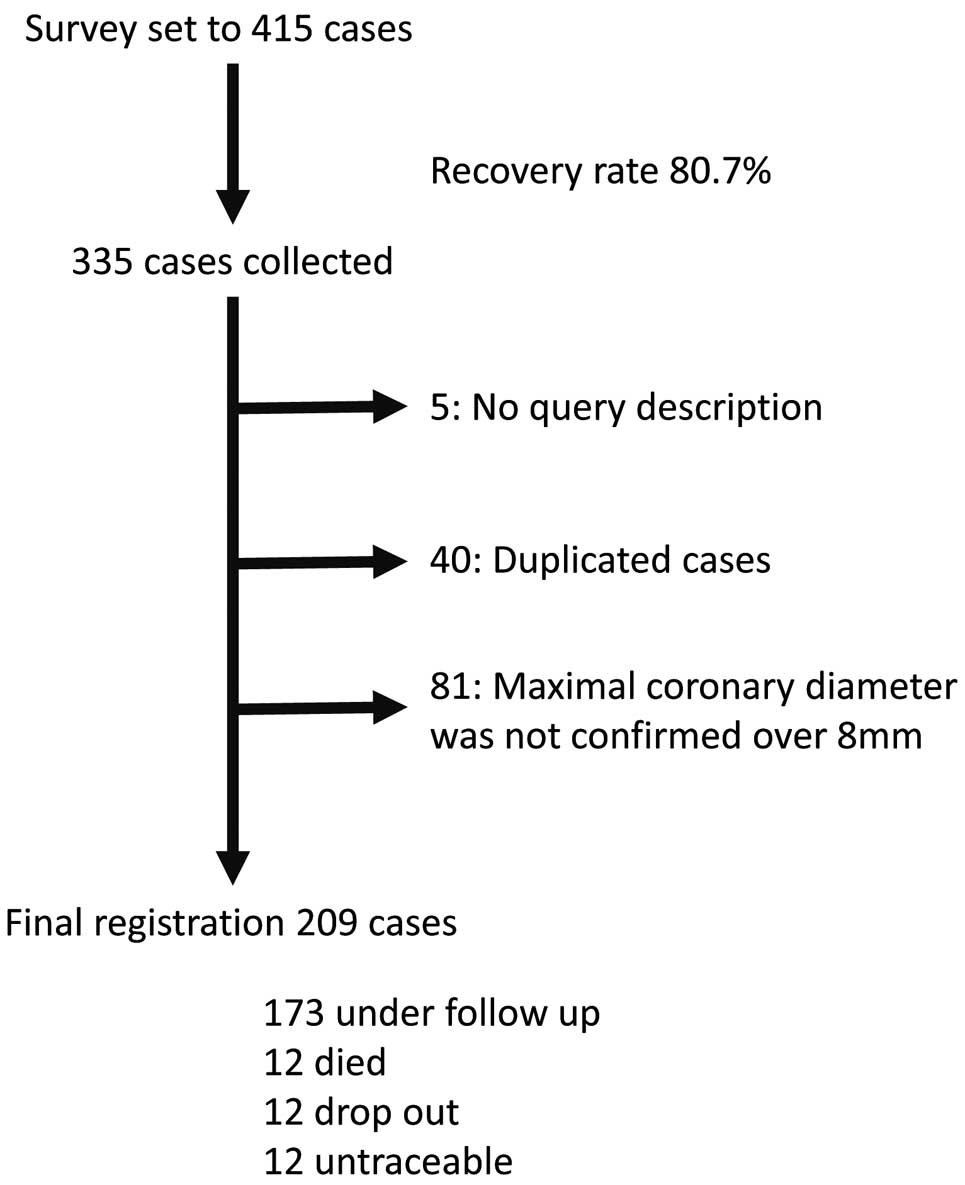
Of the 415 identified giant aneurysm patients, we collected responses for 335 (recovery rate, 80.7%). We excluded 5, 40, and 81 patients due to lack of description, duplication, and maximum coronary dilation not described or <8 mm, respectively. Finally, 209 giant aneurysm patients were included in this survey. Twenty-four patients who declined follow-up or who were untraceable, were followed for at least 2 years from disease onset (Figure 1).
Patient Characteristics
The median age at KD onset was 35 months (range, 1–158 months), and 159/209 patients were male (Figure 2). There were 7 cases of incomplete KD (<3 principal symptoms, or 4 without CAL). Immunoglobulin (IVIG) was used as the initial therapy in 199 patients (96%) at a median of 5 days (range, 2–22 days) from onset. The duration from onset to IVIG treatment was not associated with total cardiac events. In 180 patients, additional treatment was adopted, suggesting that most patients were refractory to the initial IVIG. Additional IVIG and/or steroid therapy was started at a median of 8 days (range, 5–30 days) from onset. Steroids were used in 120 cases. Plasma exchange was performed after a median of 13 days (range, 10–20 days). Included patient data are listed in
Table 1.

Table 1.
Patient Characteristics
| Diagnosis (Complete/Incomplete) |
186/7 |
| Initial therapy |
| IVIG |
199/209 |
| Illness days until initial IVIG |
5.3±3.0, 5 (2–22) |
| Additional treatment |
|
| IVIG (yes/no) |
155/39 |
| Illness days until first additional IVIG |
9.0±4.3, 8 (5–30) |
| Steroids (yes/no) |
120/84 |
| PSL/Pulse/Pulse+PSL/others |
24/45/38/8 |
| Illness days until first additional steroids |
8.8±3.2, 8 (2–22) |
| Urinastatin (yes/no) |
95/108 |
| Illness days until first additional urinastatin |
9.5±4.5, 8 (3–30) |
| Plasma exchange (yes/no) |
7/197 |
| Illness days until first additional plasma exchange |
13.4±3.3, 13 (10–20) |
| Cyclosporine (yes/no) |
18/186 |
| Infliximab (yes/no) |
11/194 |
| Laboratory data at diagnosis |
| WBC count (×1,000/mL), n=195 |
14.5±6.6, 13.5 (4.1–45.3) |
| Hematocrit (%), n=182 |
33.6±3.5, 34.3 (22.2–41.1) |
| Neutrophils (%), n=159 |
78.5±12.8, 82 (34–98) |
| Platelet count (×10,000/μL), n=193 |
33.7±15.0, 30.2 (10.0–97.4) |
| Total bilirubin (mg/dL), n=157 |
1.9±1.9, 0.9 (0.1–8.8) |
| AST (IU/L), n=193 |
155±255, 62 (11–1,900) |
| ALT (IU/L), n=192 |
149±190, 82 (5–1,441) |
| Na (mmol/L), n=182 |
133.1±3.8, 133.0 (122.6–146.0) |
| Alb (g/dL), n=166 |
3.4±0.6, 3.4 (1.6–4.7) |
| CRP (mg/dL), n=194 |
12.1±6.6, 10.6 (0.5–33.1) |
| CAL at diagnosis (yes/no) |
31/161 |
| Illness day at CAL formation |
11.0±4.0, 11 (4–28) |
| Illness day of maximum CAL diameter |
26.3±12.9, 24 (8–81) |
| Maximum RCA diameter (mm) |
9.9±4.4, 9.0 (2.2–41) |
| Maximum LCA diameter (mm) |
10.0±4.5, 9.0 (1.8–33) |
| Giant aneurysm (RCA/LCA/bilateral) |
57/59/93 |
| Maximum giant aneurysm diameter (mm) |
11.7±4.7, 10.2 (8–41) |
| Length of follow-up (months) |
26±13, 95 (1–180) |
| Thrombosis (yes/no) |
67/126 |
| RCA |
44/149 |
| LCA |
41/152 |
| Stenosis (yes/no) |
70/123 |
| RCA |
48/145 |
| LCA |
43/150 |
| Obstruction (yes/no) |
50/143 |
| RCA |
37/156 |
| LCA |
26/167 |
| MI (yes/no) |
32/177 |
| RCA |
16/193 |
| LCA |
18/191 |
| Thrombolysis (yes/no) |
38/171 |
| Catheter intervention (yes/no) |
8/201 |
| Coronary artery bypass surgery (yes/no) |
22/187 |
| Mortality |
12 (5.7) |
| Intervention (thrombolysis, catheter interventions, or CABG), MI, or death (yes/no) |
60/149 |
Data given as mean±SD, median (range) or n (%). Alb, albumin; ALT, alanine aminotransferase; AST, aspartate aminotransferase; CABG, coronary artery bypass graft; CAL, coronary artery lesion; CRP, C-reactive protein; IVIG, i.v. immunoglobulin; LCA, left coronary artery; MI, myocardial infarction; Na, sodium; PSL, prednisolone; RCA, right coronary artery; WBC, white blood cell.
Coronary aneurysms were already formed at the time of KD diagnosis in 31 patients (16.2%). The duration from onset to the first IVIG treatment was significantly greater in patients with vs. without coronary lesions at diagnosis (8.9±5.3 vs. 4.6±1.5 days, P<0.0001). In patients without coronary aneurysms at diagnosis, coronary aneurysms began to form at 11 days (range, 4–28 days) from onset. Giant aneurysms were formed at the RCA, LCA, and both arteries in 57 (27.3%), 59 (28.2%), and 93 patients (44.5%), respectively.
Total Cardiac Events
Total cardiac events included cardiac death, MI, thrombolysis therapy, catheter intervention, and CABG. The total cardiac event-free rate at 1, 5, and 10 years after onset was 0.80, 0.72, and 0.68, respectively (Figure 3A). The majority of cardiac events were ruptures, which were likely to occur within 1 month from onset; MI, likely to occur within 2 years from onset; and CABG, likely to occur after 2 years from onset. The younger the patient was, the more likely they were to develop total cardiac events (34.4±33.2 vs. 45.1±33.6 months, P=0.051) and larger aneurysm size (largest size, including both the RCA and LCA: 14.4±6.4 vs. 10.6±3.2 mm, P<0.0001). The occurrence of total cardiac events was also associated with bilateral giant aneurysms (P<0.0001), coronary stenosis (>75%; P<0.0001), and ischemia (P<0.0001).
Total RCA and LCA Cardiac Events
The number of patients with maximum RCA and LCA aneurysm diameter ≥8 mm was 150 and 152, respectively. And the total cardiac RCA and LCA events were 31 and 41, respectively. The total cardiac event-free rate for RCA aneurysms at 1, 5, and 10 years after onset was 0.87, 0.81, and 0.76, respectively (Figure 3B), and that for LCA aneurysms was 0.82, 0.75, and 0.69, respectively (Figure 3C). The adjusted hazard ratio of maximum RCA aneurysm diameter was 1.097 (95% CI: 1.017–1.185, P=0.0171;
Table 2A), and that for LCA aneurysm was 1.142 (95% CI: 1.079–1.210, P<0.0001;
Table 2B). The maximum diameters of the RCA/LCA aneurysms are given in the Supplementary Results. Total cardiac event were more likely to occur in patients with giant aneurysm ≥10 mm compared with those ≥8 mm and <10 mm in both RCA and LCA (Figures S1,S2
and Tables S1,S2).
Table 2.
Cox Proportional Hazards Model
| Variable |
HR |
95% Wald CI |
| Lower |
Upper |
| (A) Continuous variable: maximum RCA aneurysm diameter |
| Maximum diameter of RCA aneurysm |
1.097 |
1.017 |
1.185 |
| Age (years) |
1.100 |
0.805 |
1.503 |
| Sex (male vs. female) |
0.888 |
0.760 |
1.038 |
| Steroid (yes vs. no) |
2.092 |
0.596 |
7.349 |
| (B) Continuous variable: maximum LCA aneurysm diameter |
| Maximum diameter of LCA aneurysm |
1.142 |
1.079 |
1.210 |
| No. LCA aneurysms |
0.862 |
0.563 |
1.321 |
| Age (years) |
0.883 |
0.764 |
1.021 |
| Sex (male vs. female) |
1.024 |
0.453 |
2.317 |
| Steroid (yes vs. no) |
1.257 |
0.585 |
2.700 |
LCA, left coronary artery; RCA, right coronary artery.
Death was reported in 12 cases (mortality rate, 5.7%;
Figure 4). Ten of 12 deaths (83.3%) occurred within 1 year of KD onset. The causes of death were coronary artery rupture (n=5), MI (n=6), and intracranial hemorrhage (ICH; n=1). All coronary ruptures occurred within 1 month from onset (Table 3). All patients had received IVIG, but it was not effective. All but 1 patient, in whom an aneurysm already existed at the time of the treatment decision, received steroids in addition to IVIG. In all other patients, the aneurysm was not detected at the time of steroid initiation. The courses leading to rupture were similar; despite treatment, the coronary aneurysm continued to grow rapidly until rupture. Most deaths due to MI occurred within 6 months from onset (4/6), and none occurred beyond 2 years from onset. Four of 6 MI deaths occurred at the first MI. One accidental death was reported due to cerebral bleeding. In this case, warfarin and aspirin therapy was continued, and the prothrombin time international normalized ratio (PT-INR) was 3.3 at the time of the cerebral bleeding accident.

Table 3.
Summary of Coronary Artery Rupture Cases
| |
ID no. |
| 1 |
2 |
3 |
4 |
5 |
| Sex |
M |
M |
F |
M |
M |
| Age at KD (months) |
1 |
3 |
18 |
42 |
68 |
| Initial IVIG day |
3 |
5 |
5 |
4 |
3 |
| Aspirin start day |
4 |
3 |
4 |
4 |
5 |
| Additional IVIG |
+ |
+ |
− |
+ |
+ |
| Steroid start day |
4 |
10 |
8 |
6 |
− |
| Steroid treatment method |
2.0 mg/kg/day |
2.1 mg/kg/day |
2.2 mg/kg/day |
Pulse |
− |
| CAL at Diagnosis |
− |
− |
− |
− |
+ |
| CAL start day |
10 |
15 |
10 |
12 |
7 |
| Day CAL became maximum |
23 |
24 |
16 |
12 |
12 |
| Day of rupture |
26 |
24 |
19 |
22 |
24 |
| RCA (mm) |
6.9 |
13.7 |
7.7 |
9 |
10 |
| LMT (mm) |
3 |
2 |
6.7 |
4 |
18 |
| LAD (mm) |
10.6 |
4.5 |
11 |
6 |
|
CAL, coronary artery lesion; IVIG, i.v. immunoglobulin; KD, Kawasaki disease; LAD, left anterior descending coronary artery; LMT left main trunk; RCA, right coronary artery.
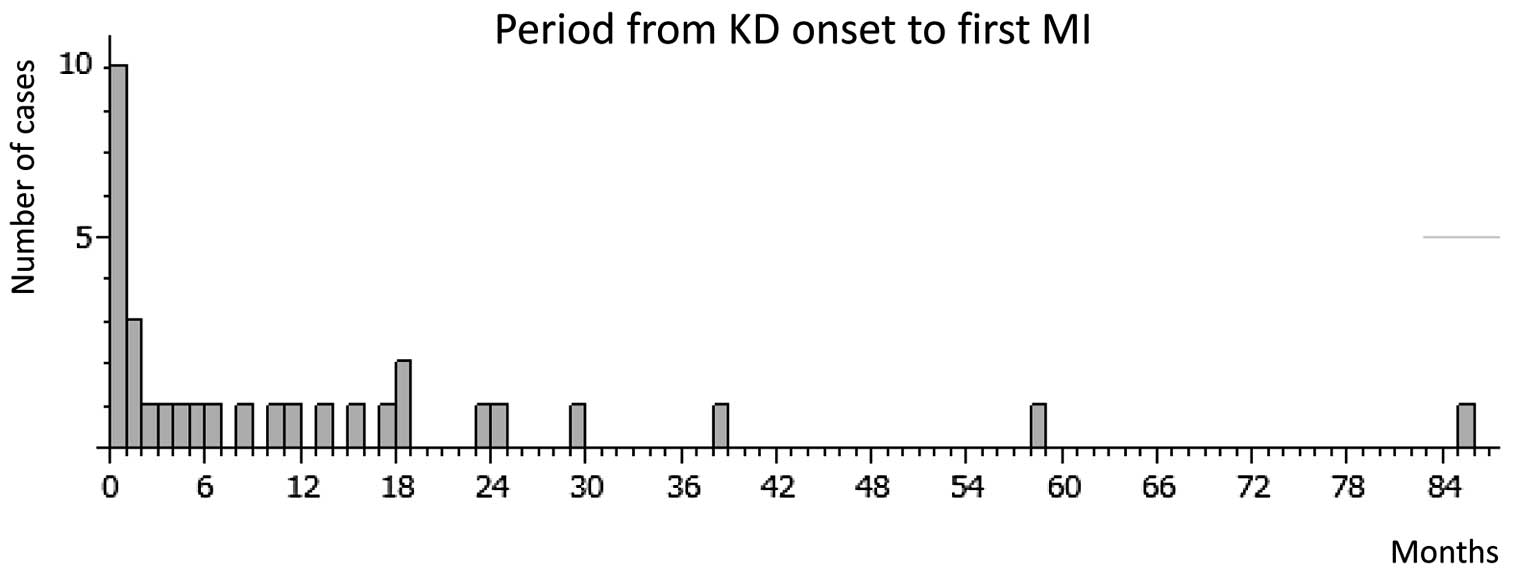
MI occurred in 32 cases (15.8%), with the first MI occurring a median of 5 months (range, 0–85 months) from onset. Except for 1 patient whose MI onset date was unclear, approximately half of the MI (15/31) occurred within 6 months from onset, and 87% (27/31) occurred within 2 years (Figure 5). Steroid therapy were not related to MI, or MI onset period. The affected sites were the RCA, LAD, left circumflex artery, and both RCA and LAD in 14 (43.7%), 13 (40.6%), 3 (9.4%), and 2 patients (6.3%), respectively. In 6 cases, MI occurred twice. MI was significantly more likely to occur in patients with thrombus in the aneurysm (OR, 28.3; 95% CI: 8.2–97.8, P<0.0001) and those with >75% stenosis (OR, 50.3; 95% CI: 11.5–220.3, P<0.0001). Aneurysm diameter was significantly greater in MI patients than in those without (14.7±5.0 vs. 11.2±4.4 mm, P<0.0001).
School Activity
School activity management classification is commonly used for determining school-life activity in Japan. This classification is divided into 5 classes (A–E). Class A patients require hospitalization or home care and are prohibited from attending any school classes. Class B patients are allowed to attend school classes except for exercise. In classes C, D, and E, light, moderate, and strenuous exercises are allowed, respectively. The present patients consisted of 14 class C, 45 class D, and 96 class E patients. Nineteen patients were too young to enter school at the time of this study and hence were ineligible for classification. Notably, no patients were categorized as class A or B; and approximately 96 were class E (55.2%). Including class D, in which only intense exercise is prohibited, 141/174 patients (81.0%) were leading daily lives with high activities.
Therapy Management
Almost all patients were taking aspirin (92.4%), while 17.3% were also prescribed clopidogrel. Warfarin was given to 71.2%. PT-INR was controlled at >2.0 in 58.0% of patients. In cases of MI, the number of patients with PT-INR >2.0 tended to be higher than for non-MI patients (OR, 3.6; 95% CI: 0.98–13.5, P=0.057). Moreover, the school activity classification tended to be more severe in patients treated with warfarin. When we compared class E (no exercise limitation) to classes C and D (mild–moderate exercise limitation), the rate of warfarin use was significantly lower in class E patients (OR, 0.33; 95% CI: 0.15–0.74, P=0.0065).
Thrombolysis
Thirty-eight patients (18.2%) received thrombolysis. The first thrombolysis treatment took place 1.0 month (range, 0–42 months) from KD onset and was performed via i.v. thrombolysis, i.c. thrombolysis, and both methods in 29 (76.3%), 5 (13.2%), and 4 cases (10.5%), respectively. As a thrombolytic agent, urokinase, tissue plasminogen activator, and both were used in 20 (52.6%), 13 (34.2%), and 5 cases (13.2%), respectively. The effectiveness of the initial thrombolysis was 63.2%. With the exception of 1 patient, warfarin and aspirin were used at the time of initial thrombolytic therapy in all cases. There were 2 adverse events: 1 case of mild nasal bleeding; and a very small ICH with no complications.
CABG
Twenty-two patients were reported to have undergone CABG. The median time of CABG was 56 months (range, 3–117 months) from onset. Twelve patients received CABG after MI occurrence, of whom 4 patients received CABG within 3 months after MI onset. Another 10 patients did not have MI. Patients who underwent CABG had significantly higher incidences of MI (OR, 10.0; 95% CI: 3.8–26.1, P<0.0001), stenosis (OR, 52.3; 95% CI: 6.8–399.4, P<0.0001), and thrombolysis (OR, 5.9; 95% CI: 2.3–15.0, P=0.0003), and a significantly lower rate of warfarin treatment (OR, 0.23; 95% CI: 0.09–0.57, P=0.0019). There was no difference, however, in school activity classification between CABG and non-CABG patients.
Steroid Therapy at Acute Phase
Steroids were used in 120 cases. Total cardiac events were higher in the steroid group (34.2%, 41/120 vs. 19.0%, 16/84, P=0.0183). Of the cardiac events, thrombolysis was the most common in the steroid group (22.5%, 27/120 vs. 10.7%, 9/84, P=0.0393). Other total cardiac events, such as MI (16.7%, 20/120 vs. 13.1%, 11/84, P=0.553), death (8.3%, 10/120 vs. 2.4%, 2/84, P=0.1277), catheter intervention (5.0%, 6/120 vs. 2.4%, 2/84, P=0.4747), or CABG (10.0%, 12/120 vs. 8.3%, 7/84, P=0.8086) were not significantly different. Also, we did not detect a significant relationship between aneurysm size and steroid treatment.
Discussion
Giant coronary aneurysm is a severe cardiac sequela that rarely regresses and commonly induces cardiac events including coronary stenosis, cardiac ischemia, MI, and sudden death. Using data from nationwide surveys over a 10-year period, we evaluated the overall outcomes of giant coronary aneurysms in Japan. We found that cardiac death and MI were likely to occur within 2 years from onset, while no death occurred beyond 2 years. Hence, careful monitoring is necessary, especially during the first 2 years from onset. Moreover, we found that most patients with giant coronary aneurysms were leading high activity lives under appropriate management. Taken together, we believe these findings are useful for physicians treating severe KD.
The shape of coronary aneurysms continuously changes. Growth factors are still actively produced in aneurysm tissue, and active remodeling occurs even in the late phase of KD.7
Smaller aneurysms tend to regress, with approximately half appearing normal on coronary angiograms 1–2 years after KD onset.8
Among 75 giant aneurysm patients observed for >20 years, intervention for stenosis was required in 61% of cases, and the accumulated rates of coronary intervention at 5, 15, and 25 years were 28%, 43%, and 59%, respectively.5
In 1 report, intimal proliferation tended to occur when aneurysm size exceeded 4 mm.9
Another study said medium-sized (6–8-mm) aneurysms developed stenosis in 6%, 28%, and 52% of cases after 5, 10, and 15 years of KD onset, respectively, giant aneurysms developed stenosis in 44%, 62%, and 74% of cases, respectively,10
and this trend was confirmed by another group.11
In the present study, total cardiac events were more likely to occur in patients with giant coronary aneurysm ≥10 mm compared with those ≥8 and <10 mm in both the RCA and LCA (Supplementary Results). Thus, the larger the aneurysm size, the higher the cardiac event risk may become.
In the present study, the 10-year survival rate was 94.3% (197/209), and the total cardiac event-free rate was 0.68, similar to previous single-center and local studies.5,6
Hence, although the survival prognosis of giant aneurysm patients is favorable, cardiac events are likely to develop. In previous studies, MI tended to occur within 1 year from onset,12,13
and in the present study, similarly, the first MI occurred a median of 5 months (range, 0–85 months) from KD onset. These results indicate that severe outcomes are likely to occur within several years from KD onset. Twelve deaths were reported in the present study. Except for 1 accidental death, all others were cardiac related. While deaths from coronary rupture were limited to within 1 month from onset, MI deaths mostly occurred within 6 months from onset (4/6), and no death was reported beyond 2 years. Notably, MI itself is a high-risk factor for mortality; MI causes deterioration of left ventricular function, which is the most important risk factor for poor survival prognosis.14
Accordingly, 1 target of KD management is avoidance of MI. In contrast, 5 deaths were due to coronary rupture. The clinical courses of these patients were very similar, with the coronary aneurysm rapidly growing daily until rupture despite various treatments (4/5 patients received steroids in addition to IVIG). In a summary of 11 cases of coronary rupture in KD patients over the past 15 years,15
most were reported in the late 1990 s or early 2000 s, and all ruptures occurred within 1 month from KD onset. Steroids, however, were used in only 2 cases. Steroid use may be unrelated to rupture. The moderate size of ruptured aneurysms in the present study (9–13 mm) suggests that expansion speed is related to rupture risk.
We have previously reported decreased endothelial function16
and reduced shear stress in giant aneurysm patients.17
Shear stress is proportional to bloodstream velocity and inversely proportional to vessel diameter. In giant aneurysms, blood flow becomes turbulent and decreases in velocity. Thus, in addition to increased vessel diameter, the shear stress to the giant aneurysm vessel wall is significantly decreased.17
This reduced shear stress along with endothelial anti-coagulation function and turbulent flow all contribute to thrombus formation in giant aneurysms. A major goal for the long-term control of giant aneurysms is avoidance of thrombus formation. The combination of aspirin, an anti-platelet drug, and warfarin, an anti-coagulant, has been demonstrated to significantly reduce cardiac events compared with aspirin or warfarin monotherapy.18,19
Japanese and US KD guidelines recommend combined use of aspirin and warfarin for giant aneurysms,20,21
and a multicenter survey in Japan showed that combined aspirin and warfarin therapy could prevent cardiac events in 91% of cases at 10 years.12
In the present study, however, the target PT-INR (>2.0) was more frequently achieved in patients with MI compared with patients without MI, which may represent a limitation of aspirin and warfarin therapy for patients with giant aneurysms.
When thrombus formation is uncontrollable or myocardial ischemia develops, thrombolysis, percutaneous coronary intervention or CABG should be considered. Thrombolysis is highly effective within several days after thrombus formation.22
In the present study, the median time to initial thrombolysis was 1 month from KD onset, and its effectiveness was 63.2%. This relatively high effectiveness may be due to early thrombus detection. Given that hemorrhagic complications are much rarer in children than adults,22
it is worth attempting thrombolysis as soon as a thrombus is detected. Furthermore, steroid therapy increased total cardiac events, especially in thrombolysis. We should keep in mind that steroid would increase the risk of thrombosis in acute phase.
CABG may be the last choice to reverse myocardial ischemia. In the patients in whom warfarin therapy could be stopped after CABG, school activity was similar to that in non-CABG patients, suggesting that CABG can improve activity levels in this population.
Finally, the school activity management classification used in the present study is an original system used in Japan. A routine cardiac health check-up system for primary, junior high, and high school has been established. Every student undergoes an initial electrocardiogram screening, and cardiac diseases are diagnosed through the second and third screening examinations. When such a diagnosis is made, a school activity management classification is issued according to cardiac disease severity. In this study, half of the patients were categorized as class E, indicating no exercise limitation. Including class D, in which only intense exercise is prohibited, 81.0% of patients were leading relatively normal daily lives. Taken together, these data suggest that adequate control of giant aneurysms results in high activity levels.
This study has some limitations. It was a nationwide questionnaire survey that covered almost all new-onset KD cases in Japan over 10 years. This follow-up period, however, is shorter than that used in similar previous reports from single facilities or restricted areas. Moreover, the questionnaire was simplified to allow for easy answers and ensure a high recovery rate. Finally, despite our best efforts, 24 patients were untraceable. These untraceable patients were all followed for at least 2 years from onset. Former physicians, however, should always be contacted with regard to significant cardiac events, therefore the influence of lack of data was considered to be minimized. This study reflects the current state of the prognosis of KD with giant aneurysms in both the acute and long-term phases, and hence the data provided herein can help guide physicians treating KD with giant aneurysms.
The present nationwide survey has shown that severe cardiac events associated with giant aneurysms tend to occur in the early phase after KD onset. When patients with giant aneurysms have been managed effectively, they can survive with good life activity. Careful monitoring and observation for the first 2 years from onset, however, are extremely important. In addition, vascular remodeling continues lifelong. Long-term monitoring is therefore crucial and should be emphasized to the parents.
Acknowledgments
Collaborating physicians and institutes are listed in the
Appendix S1.
Conflict of Interest
None.
Supplementary Files
Supplementary File 1
Supplementary Results
Appendix S1
Figure S1.
Total cardiac right coronary artery (RCA) event-free survival.
Figure S2.
Total cardiac left coronary artery (LCA) event-free survival.
Table S1.
Cox proportional hazard model: maximum RCA aneurysm diameter
Table S2.
Cox proportional hazard model: maximum LCA aneurysm diameter
Please find supplementary file(s);
http://dx.doi.org/10.1253/circj.CJ-17-0433
References
- 1.
Yanagawa H, Nakamura Y, Yashiro M, Ojima T, Koyanagi H, Kawasaki T. Update of the epidemiology of Kawasaki disease in Japan: From the results of 1993–94 nationwide survey. J Epidemiol 1996; 6: 148–157.
- 2.
Yanagawa H, Nakamura Y, Yashiro M, Oki I, Hirata S, Zhang T, et al. Incidence survey of Kawasaki disease in 1997 and 1998 in Japan. Pediatrics 2001; 107: E33.
- 3.
Makino N, Nakamura Y, Yashiro M, Ae R, Tsuboi S, Aoyama Y, et al. Descriptive epidemiology of Kawasaki disease in Japan, 2011–2012: From the results of the 22nd nationwide survey. J Epidemiol 2015; 25: 239–245.
- 4.
Tatara K, Kusakawa S. Long-term prognosis of giant coronary aneurysm in Kawasaki disease: An angiographic study. J Pediatr 1987; 111: 705–710.
- 5.
Suda K, Iemura M, Nishiono H, Teramachi Y, Koteda Y, Kishimoto S, et al. Long-term prognosis of patients with Kawasaki disease complicated by giant coronary aneurysms: A single-institution experience. Circulation 2011; 123: 1836–1842.
- 6.
Tsuda E, Hamaoka K, Suzuki H, Sakazaki H, Murakami Y, Nakagawa M, et al. A survey of the 3-decade outcome for patients with giant aneurysms caused by Kawasaki disease. Am Heart J 2014; 167: 249–258.
- 7.
Suzuki A, Miyagawa-Tomita S, Komatsu K, Nishikawa T, Sakomura Y, Horie T, et al. Active remodeling of the coronary arterial lesions in the late phase of Kawasaki disease: Immunohistochemical study. Circulation 2000; 101: 2935–2941.
- 8.
Kato H, Ichinose E, Yoshioka F, Takechi T, Matsunaga S, Suzuki K, et al. Fate of coronary aneurysms in Kawasaki disease: Serial coronary angiography and long-term follow-up study. Am J Cardiol 1982; 49: 1758–1766.
- 9.
Tsuda E, Kamiya T, Kimura K, Ono Y, Echigo S. Coronary artery dilatation exceeding 4.0 mm during acute Kawasaki disease predicts a high probability of subsequent late intima-medial thickening. Pediatr Cardiol 2002; 23: 9–14.
- 10.
Tsuda E, Kamiya T, Ono Y, Kimura K, Kurosaki K, Echigo S. Incidence of stenotic lesions predicted by acute phase changes in coronary arterial diameter during Kawasaki disease. Pediatr Cardiol 2005; 26: 73–79.
- 11.
Mueller F, Knirsch W, Harpes P, Pretre R, Valsangiacomo Buechel E, Kretschmar O. Long-term follow-up of acute changes in coronary artery diameter caused by Kawasaki disease: Risk factors for development of stenotic lesions. Clin Res Cardiol 2009; 98: 501–507.
- 12.
Suda K, Kudo Y, Higaki T, Nomura Y, Miura M, Matsumura M, et al. Multicenter and retrospective case study of warfarin and aspirin combination therapy in patients with giant coronary aneurysms caused by Kawasaki disease. Circ J 2009; 73: 1319–1323.
- 13.
Kato H, Ichinose E, Kawasaki T. Myocardial infarction in Kawasaki disease: Clinical analyses in 195 cases. J Pediatr 1986; 108: 923–927.
- 14.
Tsuda E, Hirata T, Matsuo O, Abe T, Sugiyama H, Yamada O. The 30-year outcome for patients after myocardial infarction due to coronary artery lesions caused by Kawasaki disease. Pediatr Cardiol 2011; 32: 176–182.
- 15.
Miyamoto T, Ikeda K, Ishii Y, Kobayashi T. Rupture of a coronary artery aneurysm in Kawasaki disease: A rare case and review of the literature for the past 15 years. J Thorac Cardiovasc Surg 2014; 147: e67–e69.
- 16.
Fukazawa R, Ikegam E, Watanabe M, Hajikano M, Kamisago M, Katsube Y, et al. Coronary artery aneurysm induced by Kawasaki disease in children show features typical senescence. Circ J 2007; 71: 709–715.
- 17.
Ohkubo T, Fukazawa R, Ikegami E, Ogawa S. Reduced shear stress and disturbed flow may lead to coronary aneurysm and thrombus formations. Pediatr Int 2007; 49: 1–7.
- 18.
Sugahara Y, Ishii M, Muta H, Iemura M, Matsuishi T, Kato H. Warfarin therapy for giant aneurysm prevents myocardial infarction in Kawasaki disease. Pediatr Cardiol 2008; 29: 398–401.
- 19.
Onouchi Z, Hamaoka K, Sakata K, Ozawa S, Shiraishi I, Itoi T, et al. Long-term changes in coronary artery aneurysms in patients with Kawasaki disease: Comparison of therapeutic regimens. Circ J 2005; 69: 265–272.
- 20.
JCS Joint Working Group. Guidelines for diagnosis and management of cardiovascular sequelae in Kawasaki disease (JCS 2013): Digest version. Circ J 2014; 78: 2521–2562.
- 21.
Newburger JW, Takahashi M, Gerber MA, Gewitz MH, Tani LY, Burns JC, et al. Diagnosis, treatment, and long-term management of Kawasaki disease: A statement for health professionals from the Committee on Rheumatic Fever, Endocarditis and Kawasaki Disease, Council on Cardiovascular Disease in the Young, American Heart Association. Circulation 2004; 110: 2747–2771.
- 22.
Harada M, Akimoto K, Ogawa S, Kato H, Nakamura Y, Hamaoka K, et al. National Japanese survey of thrombolytic therapy selection for coronary aneurysm: Intracoronary thrombolysis or intravenous coronary thrombolysis in patients with Kawasaki disease. Pediatr Int 2013; 55: 690–695.