2019 Volume 67 Issue 5 Pages 501-504
2019 Volume 67 Issue 5 Pages 501-504
An aqueous solution of equimolecular amounts of 2-chloropyrimidine and (−)-epigallocatechin-3-O-gallate (EGCg) afforded a colorless block crystal, which was determined to be a 2 : 2 complex of 2-chloropyrimidine and EGCg by X-ray crystallographic analysis. The 2 : 2 complex was formed by the cooperative effect of three intermolecular interactions, π–π and CH–π interactions, and intermolecular hydrogen bonds. Upon formation of the 2 : 2 complex, a 2-chloropyrimidine molecule was captured by a hydrophobic space formed by the three aromatic rings of A, B, and B′ rings of two EGCg molecules. The molecular capture abilities of various heterocyclic compounds using EGCg were evaluated by ratio of the heterocyclic compounds included in the precipitates of complex of EGCg to the heterocyclic compounds used. The amount of the heterocyclic compounds was measured by an integrated value of corresponding proton signals in the quantitative 1H-NMR spectrum.
Tea is commonly prepared by pouring hot or boiling water over leaves of the tea plant, Camellia sinensis, Theaceae, which includes caffeine, tannins, vitamins, and theanine.1) When a hot tea beverage cools down, it becomes turbid and brown-white particles settle out. This phenomenon is called a “creaming-down.” Since creaming-down triggers deformation of the original appearance and color of tea, it is one of the most serious problems in making tea beverages.
Previously, Ina and colleagues reported that creaming-down eventually occurs when an aqueous caffeine solution is poured into an aqueous solution of (−)-epigallocatechin-3-O-gallate (EGCg), which is the most abundant of the tea catechins.2)
Therefore, we attempted crystallization of the precipitate of creaming-down made from an aqueous solution of caffeine and EGCg, and as a result obtained a crystal which was determined to be a 2 : 2 complex of caffeine and EGCg by X-ray crystallographic analysis3) (Fig. 1a).
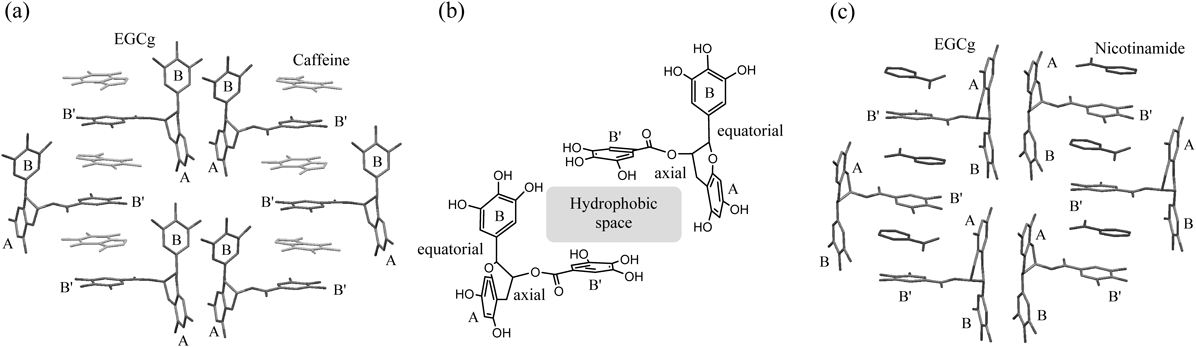
(a) 2 : 2 complex of caffeine and EGCg, (b) Hydrophobic space formed by two EGCg molecules, (c) 2 : 2 complex of nicotinamide and EGCg.
As shown in Fig. 1b, the caffeine moieties of the 2 : 2 complex were located in the space surrounding the bottom and top walls of the B′ rings of EGCg moieties and left and right walls of the A and B rings of EGCg moieties. As a result, caffeine molecules were captured by the hydrophobic space formed by the three aromatic rings of A, B, B′ rings of the EGCg in the 2 : 2 complex. Water molecules existed outside the space formed by the three A, B, B′ aromatic rings of EGCg and were not observed in the space, suggesting that the space had high hydrophobicity. It was therefore thought that the precipitate of creaming-down derived from the aqueous solution of caffeine and EGCg occurred due to its high hydrophobicity.
Nicotinamide, which is also a tea ingredient, has an aromatic ring for formation of π–π interactions with the B′ ring of EGCg. Next, we obtained the precipitate of the creaming-down made from an aqueous solution of nicotinamide and EGCg, and then obtained its crystal, which was determined to be a 2 : 2 complex of nicotinamide and EGCg, similar to a 2 : 2 complex of caffeine and EGCg, by X-ray crystallographic analysis4) (Fig. 1c). As a result, caffeine and nicotinamide molecules were captured by the hydrophobic space formed by the three aromatic rings of A, B, B′ rings of the EGCg in the 2 : 2 complex.
This time, we studied complex of precipitates made from aqueous solution of various heterocyclic compounds, which are not tea ingredient, and EGCg. Among them, the stereochemical structure of the complex of 2-chloropyrimidine and EGCg, which succeeded in crystallization, was investigated in detail by X-ray crystal structure analysis.
The capture of heterocyclic compounds using precipitates of the complex of EGCg was also investigated, and then the molecular capture abilities of the heterocyclic compounds were evaluated by ratio of the heterocyclic compounds included in the precipitates to the heterocyclic compounds used. The amount of the heterocyclic compounds was measured by an integrated value of corresponding proton signals in the quantitative 1H-NMR spectrum.
2-Chloropyrimidine (1.25 mg, 1.09 × 10−2 mmol) and EGCg (5.00 mg, 1.09 × 10−2 mmol) were dissolved in distilled water (70 µL) by heating at 90°C for 5 min. The aqueous solution was left at room temperature for 1 h, and then left at 10°C for about 1 d to obtain a colorless block crystal. This crystal was formed by 2-chloropyrimidine and EGCg at a molar ratio of 1 : 1, using measurement of the integrated value of 1H-NMR signals.
X-Ray Crystal Structure Analysis of the 2 : 2 Complex of 2-Chloropyrimidine and EGCgA single crystal of a 2 : 2 complex of 2-chloropyrimidine and EGCg was determined using X-ray crystallographic analysis at 293 K. X-Ray intensity data of 28059 reflections (of which 10363 were unique) were collected on a Rigaku RAXIS RAPID imaging plate area detector with graphite monochromated Cu-Kα radiation (λ = 1.54187 Å). The structure was solved by direct methods using SIR20081 and expanded using Fourier techniques.5) The non-hydrogen atoms were refined anisotropically. Hydrogen atoms were refined using the riding model. All calculations were performed using the CrystalStructure6) crystallographic software package except for refinement, which was performed using SHELXL97.7) These crystallographic data have been deposited with the Cambridge Crystallographic Data Center as supplementary publication no. 1878888.
Quantitative 1H-NMR Experiments1H-NMR spectra were recorded at room temperature on a JEOL JMN-LA500 (Tokyo, Japan) operating at 500 MHz. Quantitative 1H-NMR was performed with the following optimized parameters: probe temperature, 25°C; spinning, off; number of scans, eight; spectral width, 20 ppm; relaxation delay, 64 s; pulse angle, 90°; internal standard, sodium 2,2-dimethyl-2-silapentane-5-sulfonate (DSS) (Wako Pure Chemical Industries, Ltd., Osaka, Japan). Dimethyl sulfoxide (DMSO)-d6 (99.9 atom % D; Wako Pure Chemical Industries, Ltd.) was used as a solvent and DSS was used as an internal standard.
Preparation of a Sticky Precipitate Formed by Heterocyclic Compounds and EGCgHeterocyclic compounds (1.09 × 10−2 mmol) and EGCg (1.09 × 10−2 mmol) were dissolved in H2O (70 µL) at 90°C, and left at room temperature for 1 h and then 4°C for 1 d to obtain a supernatant liquid and sticky precipitate. After removing the supernatant liquid, the sticky precipitate was evaporated under reduced pressure to create a residue.
The residue was dissolved in DMSO-d6 (520 µL) containing DSS (1.44 × 10−3 mmol). The contents of the resulting DMSO-d6 solutions were measured by the integrated value of the proton signal of heterocyclic compounds not overlapping with other proton signal, singlet (2H) of H2′,6′ of EGCg, and singlet (9H) of the methyl group of DSS as an internal standard in quantitative 1H-NMR spectrum. The molecular capture abilities of various heterocyclic compounds using EGCg were evaluated by ratio of the heterocyclic compounds included in the residues to the heterocyclic compounds (1.09 × 10−2 mmol) used.
An aqueous solution of an equimolecular amount of 2-chloropyrimidine, which is not a tea ingredient, and tea gallated catechin EGCg afforded a precipitate as a colorless block crystal. This crystal contained 2-chloropyrimidine and EGCg at a molar ratio of 1 : 1, based on measurement of the integrated value of corresponding proton signals in 1H-NMR signals.
A single crystal was determined using X-ray crystallographic analysis. Based on an ORTEP drawing, this crystal contained two crystallographically different EGCgs (EGCg A and EGCg B) and two 2-chloropyrimidines, and these formed a 2 : 2 complex which the aromatic ring of the 2-chloropyrimidine and the B′ rings of each EGCg faced (Fig. 2a). Also, one unit cell contained four units of the 2 : 2 complex of 2-chloropyrimidine and EGCg and 24 H2O molecules as crystal solvent (Fig. 2b).
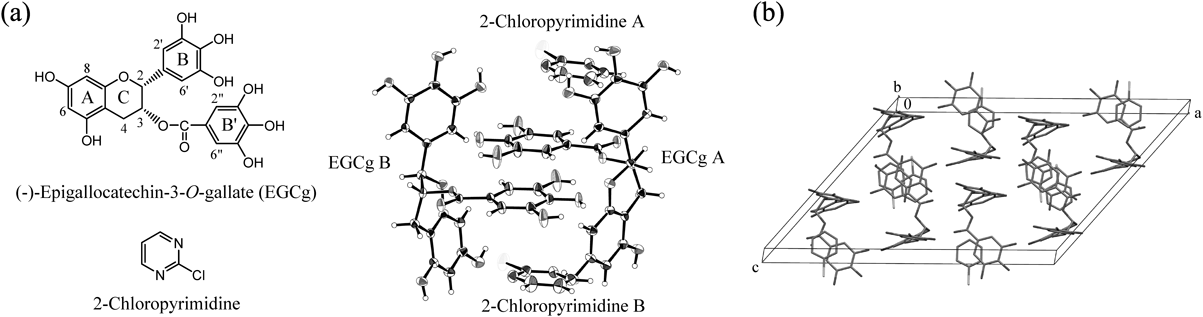
Crystal solvent is omitted for clarity. (a) ORTEP drawing with thermal ellipsoid at a 30% probability level, (b) One unit cell.
The torsion angles of the EGCg moieties (EGCg A and B) in the 2 : 2 complex of 2-chloropyrimidine and EGCg showed that the B rings of EGCgs A and B were both in equatorial positions and the B′ rings of EGCgs A and B were both in axial positions with respect to the C rings of EGCg (Table 1).
| Torsion angle | A | B |
|---|---|---|
| ∠C1′-C2-C3-O | 67.7(4)° | 66.5(4)° |
| ∠H2-C2-C3-O | 67.3° | 66.4° |
The layer structure of the 2 : 2 complex of 2-chloropyrimidine and EGCg was formed from two layers (Fig. 3). Namely, one layer had 2-chloropyrimidine molecules stacked between the B′ rings of EGCg A and the other had 2-chloropyrimidine molecules stacked between the B′ rings of EGCg B, while 2-chloropyrimidine molecules were located in almost the middle of the two B′ rings of EGCg A or B. These two layers existed parallel to the b axis.
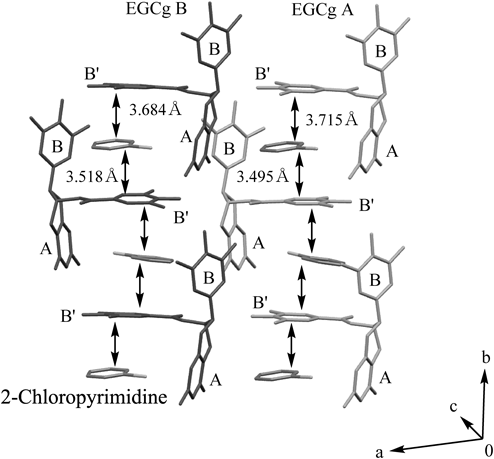
Gray and light gray molecules are EGCg A and B. Hydrogen atoms and crystal solvent are omitted for clarity.
In the 2 : 2 complex of 2-chloropyrimidine and EGCg, intermolecular interactions forming between 2-chloropyrimidine and EGCg moieties were determined (Fig. 4). π–π interactions formed between a six-membered ring of 2-chloropyrimidine and the B′ ring of EGCg, B rings of EGCg, and A rings of EGCg respectively. Also, CH–π interactions formed between the H4 of 2-chloropyrimidine and B rings of EGCg A, B, respectively. Two OH…N intermolecular hydrogen bonds were observed between EGCg and 2-chloropyrimidine.

(a) Side view (hydrogen atoms of EGCg are omitted for clarity). (b) Top view. Gray dotted and black both arrows indicate π–π interactions and CH–π interactions, respectively. Gray dotted lines indicate O–H…N hydrogen bonds.
It was found that 2-chloropyrimidine formed a 2 : 2 complex with EGCg in the same manner as the 2 : 2 complex of caffeine, nicotinamide and EGCg. As a result, 2-chloropyrimidine molecules were captured by the hydrophobic space formed by the three aromatic rings of A, B, B′ rings of the EGCg as shown in Fig. 5, and precipitated as the 2 : 2 complex from the aqueous solution of an equimolecular amount of 2-chloropyrimidine and EGCg due to its high hydrophobicity.

Hydrogen atoms and crystal solvent are omitted for clarity.
An aqueous solution of an equimolecular amount of various heterocyclic compounds (Table 2) other than 2-chloropyrimidine, which is not a tea ingredient, and EGCg also afforded sticky precipitates containing the heterocyclic compounds and EGCg at a molar ratio of 1 : 1, based on measurement of the integrated value of 1H-NMR signals. The precipitates were thought to be 2 : 2 complexes of the heterocyclic compounds and EGCg such as the 2 : 2 complex of 2-chloropyrimidine and EGCg, and the heterocyclic compounds were captured in the hydrophobic space formed by the three aromatic rings of A, B, B′ rings of EGCg.
| Heterocyclic compounds | A (%) | B |
|---|---|---|
| Pyridine | 48.73 | 1.00 |
| 2-Aminopyridine | 73.34 | 1.51 |
| 3-Aminopyridine | 66.96 | 1.37 |
| 4-Aminopyridine | 87.81 | 1.80 |
| 2-Methylpyridine | 71.69 | 1.47 |
| 3-Methylpyridine | 83.00 | 1.70 |
| 4-Methylpyridine | 77.50 | 1.59 |
| 2-Ethylpyridine | 46.13 | 0.95 |
| 4-Ethylpyridine | 43.15 | 0.89 |
| 3-Methoxypyridine | 78.67 | 1.61 |
| 4-Methoxypyridine | 92.63 | 1.90 |
| 2-Hydroxypyridine | 0.00 | 0.00 |
| 3-Hydroxypyridine | 34.73 | 0.71 |
| 4-Hydroxypyridine | 0.00 | 0.00 |
| 3-Pyridineacetonitrile | 85.44 | 1.75 |
| 3-Pyridinemethanol | 27.19 | 0.56 |
| 3-Picolylamine | 82.59 | 1.69 |
| 2-Cyanopyridine | 20.09 | 0.43 |
| 3-Cyanopyridine | 44.90 | 0.92 |
| 4-Cyanopyridine | 51.14 | 1.05 |
| 2-Nitropyridine | 49.70 | 1.02 |
| 3-Nitropyridine | 45.56 | 0.93 |
| 4-Nitropyridine | 76.67 | 1.57 |
| 2-Pyridinecarboxamide | 0.00 | 0.00 |
| 3-Pyridinecarboxamide | 44.41 | 0.91 |
| 4-Pyridinecarboxamide | 82.65 | 1.70 |
| 2-Pyridinecarboxylic acid | 52.76 | 1.08 |
| 3-Pyridinecarboxylic acid | 73.24 | 1.50 |
| Isoniazid | 66.23 | 1.36 |
| Nicotinohydrazide | 57.47 | 1.18 |
| 2-Methylpyrazine | 67.07 | 1.38 |
| Chloropyrazine | 84.07 | 1.73 |
| 3-Amino-4-hydroxypyridine | 5.28 | 0.10 |
A: Mole number of each heterocyclic compound in a crude precipitate/total mole number. B: A relative ratio when pyridine is set to 1.000.
Therefore, we investigated how much the heterocyclic compound could be captured in precipitate of the 2 : 2 complex from aqueous solution of the heterocyclic compound and EGCg. The molecular capture abilities of various heterocyclic compounds using EGCg were evaluated by ratio of the heterocyclic compounds included in the precipitates to the heterocyclic compounds used (Table 2). The amount of the heterocyclic compounds was measured by an integrated value of corresponding proton signals in the quantitative 1H-NMR spectrum in DMSO-d6. In Table 2, a heterocyclic compound having A (%) of 0% means that an aqueous solution of the heterocyclic compound and EGCg did not give a precipitate.
It was thought that the molecular capture ability depended not only on the structure of the heterocyclic compound but also on such as physical factors of the degree of polymerization, entropy, solubility, etc. of the 2 : 2 complex of EGCg and heterocyclic compound. In particular, the molecular capture ability of a heterocyclic compound having a hydroxyl group was low due to its high hydrophilicity.
The 2 : 2 complexes dissolved in organic solvent, and readily decomposed into the constituent EGCg and the heterocyclic compound, which can be easily isolated by column chromatography. Therefore, the molecular capture ability is considered to be an important indicator of how much the heterocyclic compound can be isolated using EGCg from aqueous solution.
The authors declare no conflict of interest.
The online version of this article contains supplementary materials.