2015 Volume 21 Issue 2 Pages 271-274
2015 Volume 21 Issue 2 Pages 271-274
The physiologically active substances of okara koji, a fermented soybean residue from tofu (bean curd) manufacturing, were examined. First, kojis were made with rice or barley using 14 different koji molds. Next, the acid degree, protease activity, antioxidative activity (DPPH radical scavenging activity), and angiotensin-converting enzyme (ACE) inhibitory activity of the kojis were measured. Four koji molds were selected for high levels of physiologically active substances. Finally, it was confirmed that kojis produced using the selected koji molds showed increased levels of physiologically active substances such as acid proteases and ACE inhibitory substances.
Koji mold (e.g., Aspergillus oryzae) is widely recognized to be a safe microorganism for use in food manufacturing. It has commonly been employed for the production of miso, soy sauce, liquor, vinegar, and amazake, a sweet drink made from fermented rice, and more recently for foods, enzymes, and medicine.
Okara is what remains after extracting the liquid from soybeans during the manufacture of tofu (bean curd). Some okara is destined for use as a livestock feed; however, it is characterized by high moisture levels and, because it is perishable, is typically disposed of as industrial waste (Koide, 1996). Okara is nutritionally equivalent to soybeans and is high in dietary fiber; therefore, its use as a food component is expected (Kagawa, 2009).
In a previous paper, we reported the manufacture of dolphin fish sauce using okara koji (Ochi, 2014). There are a few reports on the antioxidant activity of koji (Matsuo, 1997; Miyake et al., 2007; Matsuda et al., 2000); however, the ACE inhibitory activity of koji remains unclear.
This study undertook a comparative investigation of the physiologically active substances of okara koji, and confirmed that koji made with selected molds increased the levels of physiologically active substances such as acid proteases and ACE inhibitory substances.
Fourteen koji molds were used in this study (Table 1). Of these, 5 koji molds used for shochu (3 Aspergillus kawachii and 2 Aspergillus awamori) and 9 molds used for miso, sake, and soy sauce (8 A. oryzae and 1 Aspergillus sojae) were selected (Table 1). Kojis were prepared as follows: α-rice (or α-barley) and water (35% of the weight of α-rice or α-barley) were mixed and then autoclave-sterilized at 120°C for 20 min. After cooling, the various koji mold spores (5% wt) were individually mixed with each cooled medium and cultured on glass petri dishes (ϕ146 mm × 28 mm) at 37°C in an incubator for ca. 45 hours. After cultivation, the cultures were cooled for 2 hours at room temperature.
| Abbreviation | Commercial names | Koji mold |
|---|---|---|
| K1 | Kawachiikin(white) | Aspergillus Kawachii |
| K2 | Honkakusyouchuukomeyou(white) | Aspergillus Kawachii |
| K3 | Honkakusyouchuumugiyou(white) | Aspergillus Kawachii |
| A1 | Kawachikin(black) | Aspergillus awamori |
| A2 | Kurokojikin | Aspergillus awamori |
| O1 | Kawachikin(yellow) | Aspergillus oryzae |
| O2 | Kikojisyouchukin | Aspergillus oryzae |
| O3 | Hikamiginjyoukin | Aspergillus oryzae |
| O4 | Moromiyou | Aspergillus oryzae |
| O5 | BF1gou kin | Aspergillus oryzae |
| O6 | W-20 | Aspergillus oryzae |
| O7 | Mugimisoyou | Aspergillus oryzae |
| O8 | Suriidaiya | Aspergillus oryzae |
| S1 | Sojae No. 9 | Aspergillus sojae |
 Manufacturer:
Manufacturer:
K1,A1,O1;Kawauchi Genichiro shoten Co,Kagoshima,Japan
K2,K3,A2,O1,O2,O3,O4,O5,O6,O7,O8,S1;
Higuchi Matsunosuke Shoten Co,Osaka,Japan
Four koji molds (K1, A2, O7, and S1; shown in Table 1) were selected based on screening results. Freeze-dried okara was used as the raw material. Water (30% of the weight of okara) was added and the mixtures were then autoclave-sterilized at 120°C for 20 min. The fermentation of okara koji was carried out in the same manner as for rice koji.
The acid degree and acid protease activity were measured according to the National Tax Administration Agency prescribed analysis method (Nishiya, 2003). Measurement of neutral protease activity was conducted according to the standard miso analysis method (Yoshii, 1995).
DPPH radical scavenging activity was measured based on the method of Suda (Suda, 2000). The test sample was filtered through a 0.45 µm filter after 25 mL of an 80% ethanol solution was added to 5 g of koji and stirred for 60 min at room temperature. The antioxidant activity of samples was measured using the DPPH (1,1-diphenyl-2-picrylhydrazyl; Wako Pure Chemical Industries, Osaka, Japan) method and a 96-well microplate reader (Immuno Mini NJ-2300; Biotec, Tokyo, Japan) at an absorbance of 520 nm. The DPPH radical scavenging activity of samples is expressed as µmol Trolox equivalents (TE)/g.
To examine the ACE inhibitory activity in koji, 15 mL of distilled water (pH ranging from 4 to 9, adjusted with dilute NaOH or dilute HCl) was added to 3 g of koji and stirred for 60 min at room temperature. The filtered liquid was heated in boiling water for enzyme inactivation. The ACE activity of samples was evaluated based on the methods of Horiuchi (Horiuchi et al., 1982; Michihata et al., 2004).
Seventy microliters of each sample and 100 µL of ACE solution (0.05 units/mL; Sigma Chemical, St. Louis, MO) were incubated for five min at 37°C. Next, 30 µL of 12.5 mmol/L Bz-Gly-His-Leu (Peptide Institute Co., Osaka, Japan) was added and allowed to react for 30 minutes at 37°C. The reaction was terminated by the addition of 3% metaphosphoric acid, and the liberated hippuric acid in the sample was quantitated using high-speed liquid chromatography. Samples were separated using a cosmosil 5C18-MS-II (4.6 mm × 150 mm Wako Pure Chemical Industries) column and quantitated by photodiode array detection (PDA) at a wavelength of 228 nm.
ACE inhibitory activity was determined using the following equation.
 |
A: Area under the curve (AUC) of hippuric acid of the test sample
B: AUC of hippuric acid of distilled water
C: AUC of hippuric acid when 3% metaphosphoric acid is added to the test solution beforehand
With respect to enzyme production, the results of koji acid degree and acid and neutral protease activity determinations are shown in Table 2. High acid degree was observed in A. kawachii and A. awamori, which are used in the manufacture of shochu, specifically K2, K3, and A2. Acid generation by A. oryzae and A. sojae was not observed. High acid protease activity was observed in rice kojis (K1, K3, and A1) and high values were observed in barley kojis (K1, K2, A1, and O8). Rice koji showed higher neutral protease activity than barley koji; specifically, 11.6- and 8.1-fold higher with K1 and O6, respectively.
| Abbreviation | Rice kojis | Barley kojis | ||||||
|---|---|---|---|---|---|---|---|---|
| Acid degree | Acid protease (U/g dry koji) (10E3) | Neutral protease (U/g dry koji) | DPPH radical scavenging activity (µmol-Trolox equivalents/dry g) | Acid degree | Acid protease (U/g dry koji) (10E3) | Neutral protease (U/g dry koji) | DPPH radical scavenging activity (µmol-Trolox equivalents/dry g) | |
| K1 | 2.25 | 21.2 | 231 | 2.81 | 2.14 | 22.2 | 20 | 0.06 |
| K2 | 4.18 | 16.4 | 111 | 1.71 | 2.47 | 20.4 | 26 | 0.25 |
| K3 | 3.64 | 22.6 | 112 | 1.84 | 3.22 | 13.9 | 10 > | 1.04 |
| A1 | 1.57 | 22.0 | 179 | 2.74 | 0.96 | 27.0 | 71 | 0.12 |
| A2 | 3.00 | 15.1 | 136 | 2.87 | 2.32 | 17.8 | 10 > | 1.25 |
| O1 | 0.06 | 15.3 | 111 | 1.26 | 0.11 | 14.5 | 103 | 0.07 |
| O2 | 0.11 | 14.7 | 126 | 2.51 | 0.43 | 10.2 | 83 | 0.39 |
| O3 | 0.11 | 11.5 | 10 > | 1.81 | 0.11 | 10.1 | 38 | 0.65 |
| O4 | 0.06 | 14.2 | 10 > | 0.73 | 0.11 | 12.9 | 75 | 0.58 |
| O5 | 0.32 | 9.4 | 258 | 1.36 | 0.13 | 15.7 | 149 | 0.70 |
| O6 | 0.02 | 11.4 | 415 | 0.52 | 0.01 | 12.6 | 51 | 0.68 |
| O7 | 0.04 | 12.0 | 216 | 1.15 | 0.05 | 16.2 | 165 | 1.02 |
| O8 | 0.02 | 12.8 | 143 | 0.68 | 0.05 | 20.3 | 133 | 0.84 |
| S1 | 0.04 | 9.9 | 67 | 0.09 | 0.05 | 6.5 | 154 | 0.31 |
| Materials | ND | ND | ND | ND | ND | ND | ND | ND |
ND:Not detected
While functional activities were present in the final koji product, the rice and barley as raw materials did not exhibit antioxidative activity; therefore, it was proposed that the antioxidative activity of koji was acquired during its production. Furthermore, higher antioxidative activity was shown in K1, A1, A2, and O2 in rice koji compared to that in barley koji. Moderate activity was confirmed in K3, A2, and O7 of the barley koji.
The ACE inhibitory activity of koji appeared to be related to the pH of the extraction solution (Fig. 1). The ACE inhibitory activity of O7 showed the highest value at pH4, and decreased with increasing pH. It is considered that the level of many ACE inhibitors was elevated by the action of acid protease at the acidic pH, but not at neutral or alkaline pH. A2 also showed the highest ACE inhibitory activity at pH4; however, the activity did not appear to be strongly dependent on the pH of the extraction solution.
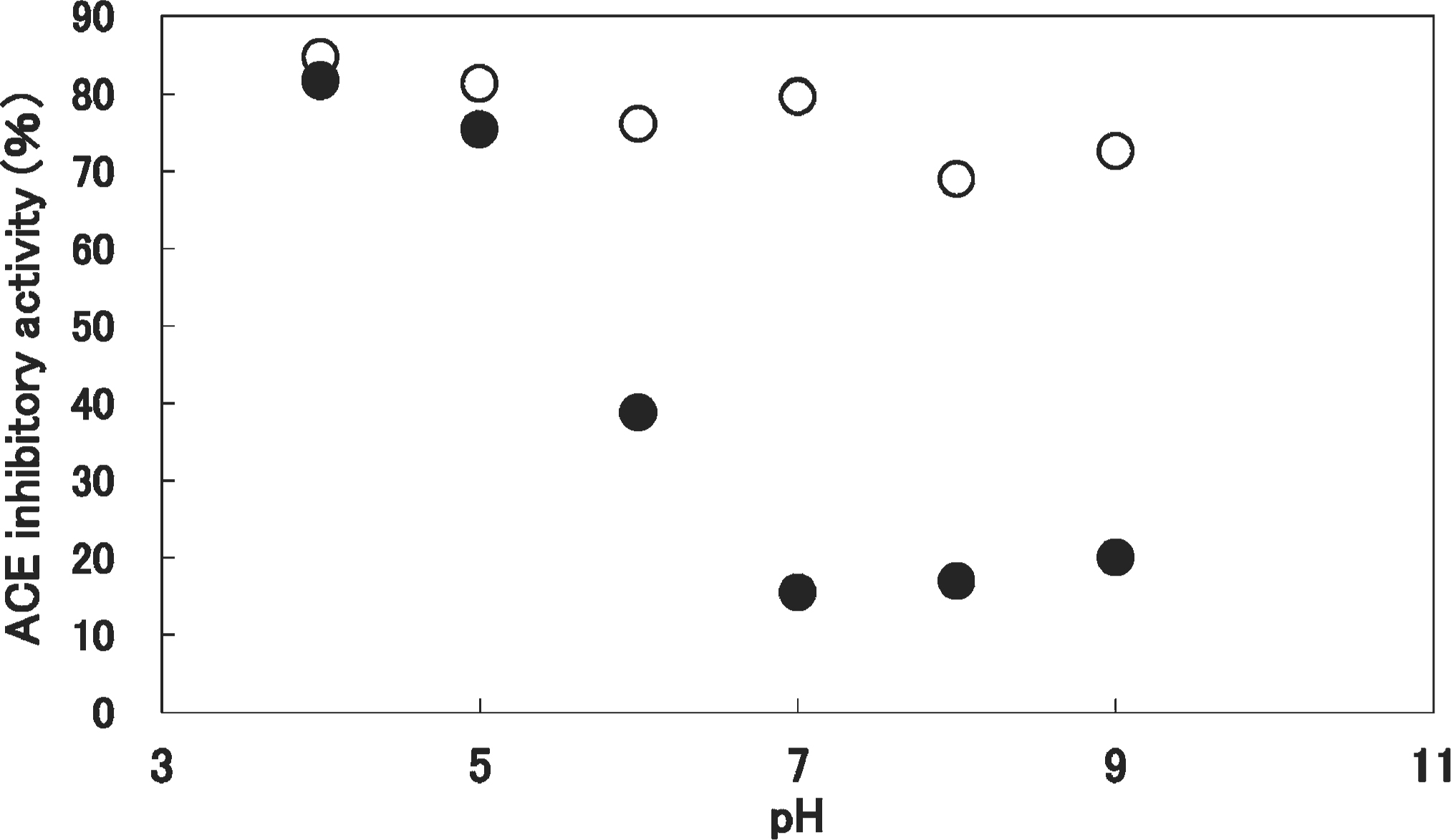
The ACE inhibitory activity of A2 (○) and O7 (•) related to the pH of the extraction solution.
Elevated ACE inhibitory activity was observed for K1, K2, K3, and A1, A2, and S1 in both the rice and barely kojis. In particular, high ACE inhibitory activity of 90% was shown in K2 and A2 of rice koji (Fig. 2).

The ACE inhibitory activity of rice (▪) and barley (□) kojis produced with 14 koji molds.
From the screening results of koji molds, four strains (K1, A2, O7, and S1) were selected for their high enzymatic and physiological activities. The ACE inhibitory activity of these four strains was particularly high, even in the raw rice and barley. Using these mold strains, okara koji was produced, and a similar examination as with rice and barley kojis was performed. As a result, okara koji produced with A2 showed an increased acid degree compared with kojis produced using rice and barley; in contrast, the acid degree of the other samples did not differ (Table 3).
| Abbreviation | Acid degree | Acid protease (U/g dry koji)(10E3) | Neutral protease (U/g dry koji) | DPPH radical scavenging activity (µmol-Trolox corresponding amount /dry g) | ACE inhibitory activity IC50 (mg/ml) |
|---|---|---|---|---|---|
| K1 | 2.3 | 99.2 | 113 | 1.4 | 10.8 |
| A2 | 4.5 | 102.0 | 233 | 3.1 | 3.9 |
| O7 | 0.0 | 7.5 | 516 | 1.4 | 32.6 |
| S1 | 0.9 | 18.7 | 885 | 1.0 | 14.8 |
The acid protease activity of okara koji produced with O7 was decreased and that of S1 was increased two-fold; further, the activity of K1 and A2 was increased five-fold (ca. 100,000 U/g koji, Table 3). It was thought that the proteases in koji were induced by the high protein content.
Moreover, although no antioxidative activity was observed in the okara raw material, increased activity was seen in the koji produced with the four mold strains. ACE inhibitory activity was expressed as IC50, defined as the quantity of sample responsible for 50% inhibition of ACE activity.
A2, which showed high ACE inhibitory activity in rice and barley kojis, also presented high activity. The IC50 value of 3.9 mg/mL is very similar to that observed in miso (5.35 mg/mL, Kawamura 2000), a seasoning made from fermented soybean that exhibits blood pressure-lowering activity. Although ACE inhibitory activity has been reported in soybean albumin (Iwashita et al., 1994), ACE inhibitory activity of okara koji remains to be clarified. This is the first report confirming the high ACE inhibitory activity of okara koji. We propose that the high ACE inhibitory activity of okara koji is due to the induction of proteases by the high protein content of the raw material. It is thought that substances with high ACE inhibitory activity, such as low molecular peptides, were generated because they were resistant to heat denaturation. It is considered that the utilization of okara koji for the production of fermented foods (e.g., miso) can enhanced their physiological activity, especially the ACE inhibitory activity.
Purification, identification, and determination of the amino acid composition of the peptide(s) showing high activity and tolerance to gastric juice will be examined in future studies.