2016 Volume 22 Issue 1 Pages 153-157
2016 Volume 22 Issue 1 Pages 153-157
We developed a more convenient method for assaying polyamines and re-examined the variation of polyamine contents during each step of the natto production process. The steaming process slightly decreased spermidine in soybeans, and the subsequent fermentation process resulted in 41.1% increase of spermidine and 19.4% reduction of spermine compared with those of steamed soybeans. These results indicated that Bacillus subtilis (natto) produces spermidine during the fermentation process. Further, we determined that spermidine production differs among inoculum strains. It was also suggested that the selection of starter cultures with high spermidine productivity improves polyamine levels in natto.
Both prokaryotes and eukaryotes contain considerable amounts of polyamines, a class of low molecular weight aliphatic polycations. These compounds interact with various biomolecules via electrostatic and covalent interactions, and have diverse effects on cellular functions. The only polyamines synthesized in mammalian cells are spermidine, spermine and putrescine. To date, numerous studies have investigated the biochemistry and physiology of these biogenic polyamines. As a result, it is well known that biogenic polyamines regulate gene expression at the level of transcription and translation (Wallance et al., 2003; Kusano et al., 2008), have anti-inflammatory effects (Soda et al., 2005) via several mechanisms, exemplified by modulating pro-inflammatory cytokines such as tumor necrosis factor (Zhang et al., 1999), and play important roles in regulating ion channels (Pegg, 2009). These discoveries suggest that the homeostasis of biogenic polyamines is essential to mammalian life. Indeed, alterations of physiological polyamine levels have been shown to be associated with carcinogenesis (Minois et al., 2011), cell death (Li et al., 2001) and heritable human diseases, such as Snyder-Robinson syndrome (Pegg, 2009).
On the other hand, animal models have indicated that the biosynthesis and tissue levels of polyamines decline with aging (Nishimura et al., 2006). In addition, increased ingestion of polyamines has been shown to inhibit tumorigenesis induced by 1,2-dimethylhydrazine (Soda et al., 2013) and to stimulate liver maturation in suckling rats (Wery et al., 1996). Taken together, the decrease of polyamine levels may be implicated in some aging-associated pathological conditions, and exogenous polyamines may play an important role in the suppression of such pathogenesis. As such, dietary sources of polyamines have garnered much interest, and the polyamine contents of various foods have been determined (Okamoto et al., 1997; Nishimura et al., 2006; Nishibori et al., 2007). Polyamine-rich foods include green pepper, orange, soybean and Japanese pumpkin as well as some kinds of fermented foods.
Fermented soybean, called natto in Japan, is a traditional food rich in polyamines. Furthermore, the long-term consumption of natto by human volunteers has been reported to increase blood polyamine levels (Soda et al., 2009). Natto is made from soybeans fermented using Bacillus subtilis (natto). Nishibori et al. (2007) measured the polyamine contents of various foods: natto contained 175 nmol/g putrescine, 232 nmol/g spermidine and 19 nmol/g spermine, whereas soybeans contained 194 nmol/g, 728 nmol/g and 181 nmol/g, respectively. These results demonstrate that the individual polyamine contents decrease during the natto fermentation process. However, Otsuka and Hamana (2006) analyzed the polyamine extracts from a glutinous biofilm on the surface of natto, and found that polyamines and their precursor agmatine were produced by B. subtilis (natto). In addition, Kim et al. (2012) reported that B. subtilis isolated from natto produced polyamines. The reason for these discordant results is unclear, but might be attributable to differences in the analytical methods used for the determination of polyamines and/or in the fermentation conditions for natto production.
Overall, these previous findings indicate that there is still considerable room to improve natto production in relation to polyamine enrichment. In Japan, natto is widely recognized as a health-promoting foodstuff because it is a source of iron, calcium, magnesium, protein, dietary fiber and vitamins, including B6, B2 and K (Shimojo, 2013). Therefore, if the polyamine content of natto could be optimized, this foodstuff would be more beneficial, particularly for the elderly. Thus, to obtain fundamental data for reinforcing the polyamine content of natto, we re-examined the variation of polyamine (spermidine, spermine and putrescine) contents during natto fermentation and determined whether inoculum strains of B. subtilis (natto) influence polyamine levels.
Materials Benzoyl chloride was obtained from Wako Pure Chemical Industries (Osaka, Japan). As polyamine standards, spermine tetrahydrochloride, spermidine trihydrochloride and putrescine dihydrochloride were obtained from Sigma-Aldrich Co. (MO, U.S.A.), Acros Organics (Geel, Belgium) and Tokyo Chemical Industries (Tokyo, Japan), respectively. Diethylenetriamine trihydrochloride was kindly provided by Dr. M. Nakajima of our research center.
Soybean cultivar ‘Enrei’ (Glycine max) harvested in 2009 was purchased from a local market.
Bacteria Three strains of Bacillus subtilis (natto) sold for natto production were obtained from Miyagino-nattou Co., Ltd. (Miyagi, Japan), Yuzo Takahashi Laboratory Co. (Yamagata, Japan) and Naruse Fermentation Laboratory Co., Ltd (Tokyo, Japan). Before use, spores were heated to inactive residual vegetative cells by suspending them in hot water (104 cells/mL) at ca. 80°C.
Derivatization of polyamines Polyamines were extracted with trichloroacetic acid from 1 g of freeze-dried samples or 1 mL of bacterial cultures, according to the method of Nishimura et al. (2006). Before the extractions, 1000 nmol of diethylenetriamine trihydrochloride was added to the sample as an internal standard. Each extract was combined, and its 0.2-mL aliquot was freeze-dried.
After 300 µL of acetonitrile (containing 240 mM benzoyl chloride and 33 mM triethylamine) was added to the extract, the solution was stirred for 15 min at 40°C, and then the reaction was stopped by the addition of 1 mL of 0.5 M NaCO3. Following the separation of benzoylated products by liquid-liquid extraction with diethyl ether and subsequent evaporation to desolvation, the residue was dissolved in 1 mL of methanol/water (30/70, v/v). The benzoylated polyamines were captured on a Bond Elut-C18 (100 mg-1 mL; Agilent Technologies, CA, U.S.A.), washed with methanol/water (30/70, v/v) and then recovered with methanol/water (90/10, v/v).
Liquid Chromatography-Mass Spectrometry (LC/MS) Benzoylated polyamines were separated and quantified with a QP-8000 system (Shimadzu, Kyoto, Japan) consisting of an autosampler, high-pressure mixing pumps and a single quadrupole mass spectrometer. HPLC conditions were as follows: a Inertsil ODS-3 column (5 µm, 2.0 × 250 mm, GL Sciences, Tokyo, Japan); mobile phase, (A) methanol/water, (10/90, v/v), (B) methanol; flow rate, 0.2 mL/min; temperature, 40°C; and gradient mode, 60% B for 2 min, 60% – 100% B for 8 min, 100% B for 10 min. MS detection was performed under an atmospheric pressure chemical ionization interface as follows: ion mode, positive; source voltage, 1.5 kV; temperatures of curved desolvation line, 230°C; probe temperature, 400°C and flow rate of nebulizer gas, 2.5 L/min. The monitored ions were m/z 297, 458, 619 and 416 for benzoylated derivatives of putrescine, spermidine, spermine and diethylenetriamine, respectively.
Validation of polyamine determination The accuracy of our analytical method was determined by the recovery of each polyamine in 1 g of freeze-dried steamed soybeans spiked with 500 nmol of polyamines. The recovery rate was calculated as follows:
 |
Changes in polyamine content during natto production Natto was produced on a laboratory scale. Five hundred grams of soybeans were washed with tap water three times and then soaked in water for 24 h at 15°C. The soybeans were steamed for 45 min at 0.1 MPa using a gas-autoclave, and after heat-shocked Bacillus spores were inoculated into steamed soybeans (inoculum rate: 104 cells/g), 45 g of the inoculated soybeans were placed in a polystyrene paper pack and transferred to an incubator. These procedures were performed as soon as possible to prevent the steamed soybeans from cooling. Soybeans were incubated for 21 h at 42°C and 95% humidity, and subsequently chilled at 5°C for one day. For the quantification of polyamines, the soybeans were freeze-dried, milled and stored at −40°C until use.
Polyamine production by pure culture of B. subtilis (natto) in a minimal liquid medium We used modified E9 minimal medium, consisting of E9 medium (Birrer et al., 1994) supplemented with biotin (10−4 g/L), which is an essential growth factor for B. subtilis (natto). Biotin was added to the medium after autoclaving.
The heat-shocked B. subtilis (natto) spores were inoculated into 100 mL of Luria-Bertani broth (inoculum rate: 104 cells/mL) and cultivated overnight at 37°C with continuous shaking. A 0.25-mL aliquot of this pre-culture was added to 25 mL of the modified E9 medium in a 200 mL flask and was then cultivated for 48 h at 37°C with continuous shaking. After cultivation, turbidity at 660 nm was measured for evaluating the growth, and the culture was stored at −40°C until the determination of polyamines.
Quantification of polyamines by LC/MS To evaluate the variation in polyamines produced during natto fermentation, polyamine content must be quantified with high accuracy and reliability. Conventionally, polyamines have been determined by HPLC with fluorescence or a UV detector, following derivatization of the amino groups. Recently, in order to improve the sensitivity and specificity of the conventional methods, Ducros et al. (2009) developed a novel method based on the combination of derivatization with dansyl chloride and LC-tandem mass spectrometry. In this study, as a more convenient method, we determined the polyamines with a single mass spectroscopy alternative to MS/MS, following derivatization with the more cost-effective benzoyl chloride. In addition, benzoylation is sufficient for a shorter incubation time compared to dansylation, which requires an overnight incubation in the dark.
The dose-response relationships were examined in the ranges of 0 – 40 nmol/mL for putrescine and spermine, and 0 – 100 nmol/mL for spermidine (Fig. 1). The results exhibited consistent linearity for each polyamine with a correlation coefficient higher than 0.99. Although the sensitivity was lower than for detection with MS/MS, this method could be applied for the polyamine analysis of foods. Next, the food matrix was evaluated for its influence on the determination of polyamines; steamed soybean did not interfere with the separation of the polyamines (Fig. 2). The recoveries of polyamines added to the soybeans were estimated to be 111.0% for putrescine, 96.2% for spermidine and 116.1% for spermine. These results suggest that our methods could quantify the polyamine contents with high reliability, even in the presence of the food matrix, i.e., the steamed soybeans.
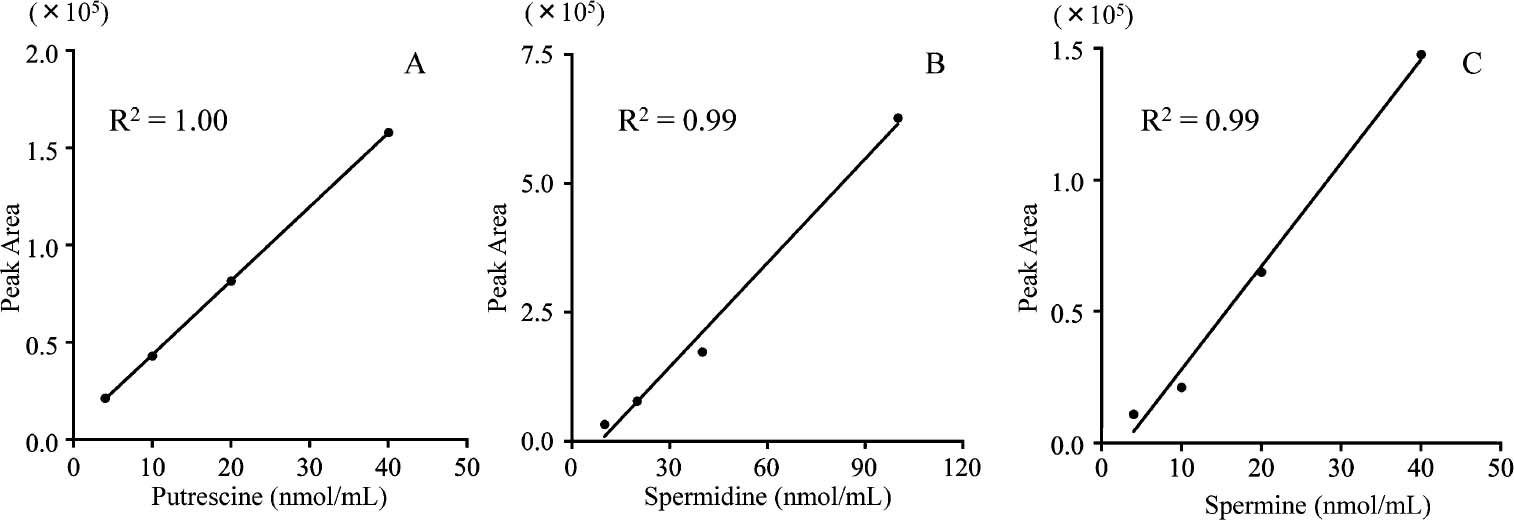
Dose-response relationships for polyamines. (A) putrescine, (B) spermidine and (C) spermine.
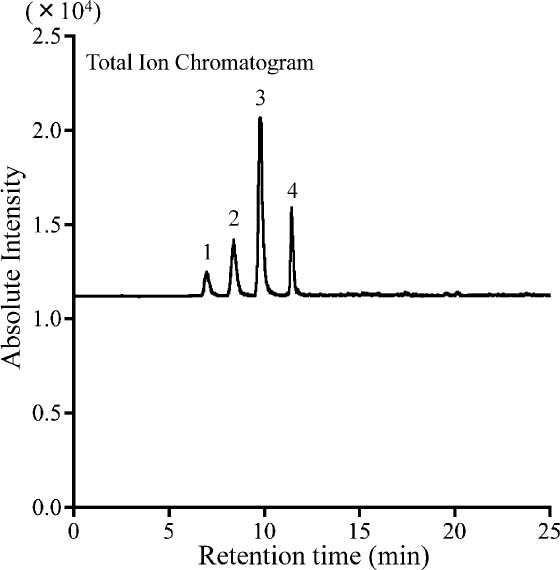
Chromatographic separation of polyamines added to steamed soybeans following benzoylation. The chromatogram was obtained from analysis of 1 g of freeze-dried steamed soybeans spiked with 500 nmol of each polyamine. Peaks: 1, putrescine; 2, diethylenetriamine (as the internal standard); 3, spermidine and 4, spermine.
Changes in polyamine contents during natto production The polyamine contents of soybeans were monitored during natto production (Fig. 3). Raw soybeans contained 383 nmol/g putrescine, 1340 nmol/g spermidine and 272 nmol/g spermine, while those of natto were 411 nmol/g, 1759 nmol/g and 178 nmol/g, respectively. The polyamine levels were sustained until the steaming process, whereas subsequent fermentation resulted in changes of polyamine levels. The spermidine content of natto was 41.1% higher than that of steamed soybean. In contrast, the spermine content decreased by 19.4% during fermentation. Additionally, putrescine was relatively unchanged during natto production. In general, because heating with steam induces the elution of water-soluble materials from soybeans (Takenaka et al., 2009), the loss of some of the highly water-soluble polyamines is predicted during natto production. However, our results showed that the polyamine contents of soybeans could be maintained during the steam process except for a slight decline of spermidine levels. This observation was similar to the finding that heat-treatment decreased spermidine and spermine levels (Verciana-Nogués et al., 1997).
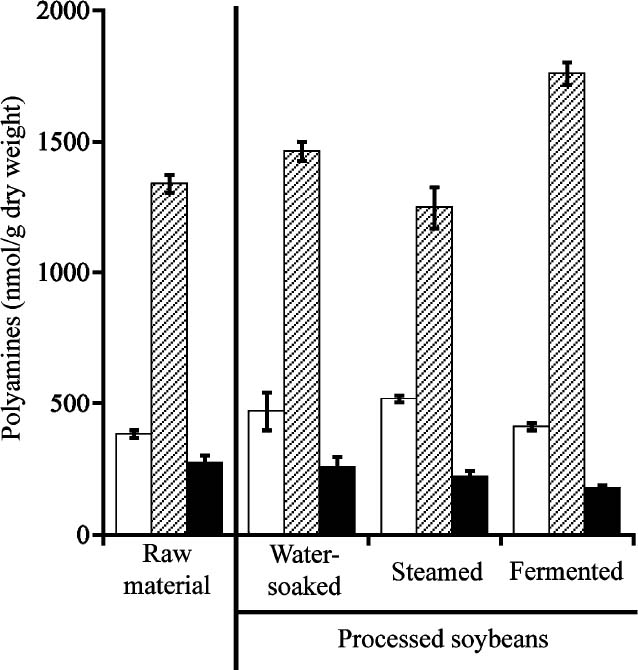
Changes in polyamine contents during natto production. White column, putrescine; shaded column, spermidine; black column, spermine. Data represent the mean values of individual polyamine contents obtained across the triplicate measurements.
The polyamine content of natto has been reported to be lower than that of raw soybeans (Nishibori et al., 2007); therefore, the elevation of spermidine after fermentation is contrary to those results. The reasons for this pattern of results are unclear, but may derive from differences in the analytical conditions. For example, we analyzed fresh samples produced in our laboratory, whereas the previous studies used commercial natto products purchased from local and specialty markets (Okamoto et al., 1997; Nishimura et al., 2006; Nishibori et al., 2007). The polyamines in natto might degrade during storage. Indeed, polyamine-degrading enzymes were identified in B. subtilis (Woolridge et al., 1999; Forouhar et al., 2005). Besides, the increase of spermidine identified by the present study agrees with the findings of Otsuka and Hamana (2006); they demonstrated that polyamines, particularly spermidine and the polyamine precursor agmatine, were produced by B. subtilis (natto) during natto production. Kim et al. (2012) also reported on the bacterial contribution to natto polyamine content.
The effect of starter culture on polyamine contents of natto The potential influence of B. subtilis (natto) starter culture on the polyamine contents of natto was examined using three commercial strains employed in natto production. After the steaming process, the soybeans contained 391 nmol/g putrescine, 1097 nmol/g spermidine and 313 nmol/g spermine. After fermentation, putrescine was relatively unchanged (421 – 453 nmol/g) and spermine decreased (181 – 200 nmol/g), suggesting that the starter culture has little influence. However, the spermidine content increased in accordance with the starter cultures: 1373 nmol/g for strain A, 1361 nmol/g for strain B and 1246 nmol/g for strain C, indicating that the spermidine production of strain C was lower than that of other two strains.
Comparison of spermidine production among starter cultures To examine further the possibility that the spermidine content of natto was affected by the choice of starter culture, polyamine production by B. subtilis (natto) was analyzed in pure culture. Firstly, only spermidine was detected for all three strains. However, the previous study indicated that B. subtilis produced spermidine as well as other polyamines (Kim et al., 2012). Given that distinct cultures were applied to each of the examinations, observational differences might occur due the bacterial production of polyamine. In fact, Kim et al. used culture conditions rich in nutrients and some free amino acids as polyamine precursors, while minimal culture conditions were employed in the present experiment.
Spermidine production was generally higher in shaking cultures compared with static cultures, although bacterial growth was unaffected by shaking conditions except for strain C (Table 1). These results suggest that aerobic conditions are preferable for spermidine production, which may not be directly associated with biomass production. In the pure culture experiments, the spermidine production of strain C was lower than that of the other two strains. Burrell et al. (2010) reported that polyamines are essential for biofilm formation in B. subtilis. Indeed, strains A and B tended to form more substantial biofilms compared with strain C (data not shown). From these observations, it appears that efficient screening for B. subtilis (natto) strains with high spermidine production could be possible via measurement of biofilm formation.
| Static culture | Shaking culture | Static culture | Shaking culture | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Spermidine (nmol/ml) | Cell growth (O.D. 660 nm) | Spermidine (nmol/ml) | Cell growth (O.D. 660 nm) | Spermidine | Cell growth | Spermidine | Cell growth | ||
| Strain A | 65.0±10.4 | 1.88±0.13 | 93.0±6.6 | 1.73±0.02 | A vs. B | * | n.s. | ** | ** |
| Strain B | 87.2±5.7 | 1.78±0.09 | 136.3±13.4 | 1.99±0.06 | A vs. C | ** | ** | ** | n.s. |
| Strain C | 18.6±2.3 | 1.08±0.05 | 33.5±4.4 | 1.83±0.10 | B vs. C | ** | ** | ** | n.s. |
Data were shown as means ± standard deviations (n = 3).
Statistical analysis was performed using the Tukey-Kramer method. *; P < 0.05, **; P < 0.01, n.s.; not significant.
Altogether, our study indicated that the polyamine contents of soybeans are maintained during natto production and could be further augmented by spermidine production of B. subtilis (natto). It was also suggested that the selection of starter cultures with high spermidine production contributes to improved natto polyamine levels, reinforcing the health benefits of this traditional Japanese fermented soy food.
Acknowledgement We thank Dr. S. Ohtsubo for his assistance in the preparation of this manuscript. This work was supported by a grant ‘A-STEP search type AS231Z03549E’ from Japan Science and Technology Agency.